Advanced Copper-Catalyzed Synthesis of N2-Aryl-1,2,4-Triazine Derivatives for Commercial Scale-Up
Introduction to Novel Triazine Synthesis Technology
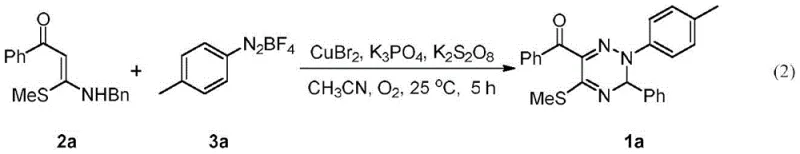
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN111018800B introduces a groundbreaking approach for the synthesis of N2-aryl substituted-1,2,4-triazine derivatives, a class of compounds renowned for their potent antiviral, anticancer, and antibacterial properties. This technology leverages a copper-catalyzed cyclization strategy that merges S,N-substituted internal olefins with aryl diazonium salts under remarkably mild conditions. By shifting away from traditional multi-step sequences that often suffer from low atom economy and harsh reaction parameters, this invention provides a streamlined pathway to access nitrogen-substituted triazines, a structural motif that has historically been challenging to synthesize with high regioselectivity. The significance of this development lies not only in its chemical elegance but also in its potential to accelerate the discovery and production of next-generation therapeutic agents.

Furthermore, the versatility of this synthetic route allows for extensive structural diversification, enabling medicinal chemists to rapidly explore structure-activity relationships (SAR) around the triazine core. The ability to introduce diverse functional groups at the N2 position opens new avenues for optimizing pharmacokinetic profiles and binding affinities in drug candidates. For procurement and supply chain professionals, understanding the underlying chemistry of such patents is crucial, as it directly impacts the feasibility of sourcing high-purity intermediates at a commercial scale. The methodology described in CN111018800B represents a significant leap forward in heterocyclic chemistry, offering a reliable alternative to legacy processes that are often plagued by safety concerns and environmental inefficiencies. As we delve deeper into the technical specifics, it becomes evident that this process is designed with both laboratory precision and industrial practicality in mind.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine skeleton has relied heavily on the condensation of hydrazides with 1,2-dicarbonyl compounds or alpha-diazo beta-ketoesters. While these classical transformations are well-documented in organic synthesis literature, they present substantial drawbacks when applied to modern drug discovery pipelines. A primary limitation is the frequent requirement for elevated reaction temperatures and prolonged heating periods, which can lead to the decomposition of thermally sensitive functional groups and the formation of complex impurity profiles. Additionally, these traditional routes typically yield triazine derivatives substituted at the carbon positions (C-3, C-5, C-6), leaving the nitrogen atoms largely unfunctionalized or requiring tedious multi-step protection and deprotection strategies to achieve N-substitution. The scarcity of reported methods for directly synthesizing N-substituted 1,2,4-triazines has created a bottleneck in accessing this specific chemical space, forcing R&D teams to rely on inefficient, low-yielding pathways that hinder rapid iteration.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111018800B utilizes a direct oxidative cyclization strategy that fundamentally reshapes the synthetic landscape for these heterocycles. By employing S,N-substituted internal olefins as key precursors alongside aryl diazonium salts, the process achieves the formation of the N-N bond and the triazine ring in a single operational step. This approach eliminates the need for pre-functionalized hydrazine derivatives, thereby reducing the overall step count and minimizing waste generation. The reaction proceeds efficiently at temperatures ranging from 25°C to 40°C, which is a dramatic improvement over the high-energy inputs required by conventional protocols. Moreover, the use of earth-abundant copper catalysts instead of precious metals aligns with green chemistry principles, offering a more sustainable and cost-effective solution for large-scale manufacturing. This novel pathway not only solves the regioselectivity issues associated with N-substitution but also provides a robust platform for generating diverse libraries of triazine derivatives with high structural fidelity.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The success of this synthesis hinges on a sophisticated catalytic cycle mediated by copper species, likely involving a single-electron transfer (SET) mechanism. In the presence of an oxidant such as potassium persulfate (K2S2O8), the copper catalyst facilitates the generation of aryl radicals from the diazonium salt precursor. These highly reactive radical species then engage with the electron-rich double bond of the S,N-substituted internal olefin, initiating a cascade of intramolecular cyclization events. The sulfur atom in the substrate plays a critical role, acting as a transient directing group or stabilizing intermediate species before being eliminated or transformed during the aromatization process to yield the final triazine core. The choice of base, such as potassium phosphate (K3PO4), is equally vital, as it helps to neutralize acidic byproducts and maintain the optimal pH environment for the catalytic turnover. Understanding these mechanistic nuances is essential for R&D directors aiming to troubleshoot potential side reactions or optimize the process for specific substrates with sensitive electronic properties.
From an impurity control perspective, the mild reaction conditions inherent to this copper-catalyzed system offer significant advantages. High-temperature processes often promote non-selective radical coupling or polymerization, leading to difficult-to-remove tarry byproducts. However, by maintaining the reaction at ambient to slightly elevated temperatures (25-40°C), the kinetic energy of the system is carefully regulated, favoring the desired cyclization pathway over competing degradation reactions. Furthermore, the use of acetonitrile as the preferred solvent ensures excellent solubility for both organic substrates and inorganic salts, promoting homogeneous reaction conditions that enhance reproducibility. The subsequent workup involving simple solvent removal and silica gel chromatography indicates that the crude reaction mixture is relatively clean, reducing the burden on downstream purification units. This level of control over the reaction trajectory ensures that the final pharmaceutical intermediates meet stringent purity specifications required for regulatory compliance in drug substance manufacturing.
How to Synthesize N2-Aryl-1,2,4-Triazine Derivatives Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and quality. The patent outlines a standardized protocol where the S,N-substituted internal olefin serves as the limiting reagent, typically reacted with a slight excess of the aryl diazonium salt to drive the conversion to completion. The optimization data suggests that a molar ratio of olefin to diazonium salt between 1:1.2 and 1:3.0 is effective, with specific embodiments demonstrating superior results at a 1:2.0 ratio. The catalyst loading is another critical parameter, with copper bromide (CuBr2) showing exceptional performance at loadings as low as 0.3 equivalents relative to the substrate. For a detailed walkthrough of the experimental procedure, including specific quantities and safety precautions, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining S,N-substituted internal olefin, aryl diazonium salt, copper catalyst (e.g., CuBr2), base (e.g., K3PO4), and oxidant (e.g., K2S2O8) in acetonitrile solvent.
- Maintain the reaction under an oxygen or air atmosphere at a mild temperature range of 25°C to 40°C for approximately 5 hours to ensure complete cyclization.
- Upon completion, remove the solvent under reduced pressure and purify the crude product via silica gel column chromatography using a petroleum ether and ethyl acetate eluent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional hydrazide-based routes to this copper-catalyzed olefin cyclization presents compelling economic and logistical benefits. The primary driver for cost reduction lies in the raw material profile; S,N-substituted internal olefins and aryl diazonium salts are generally more accessible and less expensive to produce on a metric ton scale compared to specialized hydrazide precursors. Additionally, the elimination of high-temperature heating steps translates directly into lower energy consumption utilities, significantly reducing the operational expenditure (OPEX) associated with manufacturing. The simplicity of the workup procedure, which avoids complex extraction or crystallization steps in favor of straightforward chromatography or precipitation, further streamlines the production timeline, allowing for faster turnaround times and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The utilization of inexpensive copper salts like CuBr2 or CuCl2 as catalysts removes the dependency on costly precious metals such as palladium or rhodium, which are subject to volatile market pricing and supply constraints. This shift to base metal catalysis ensures a more stable cost structure for the long-term production of these intermediates. Furthermore, the reaction operates efficiently under air or oxygen atmospheres, negating the need for expensive inert gas blanketing systems or specialized pressure vessels, which simplifies the reactor setup and lowers capital expenditure requirements for plant modifications.
- Enhanced Supply Chain Reliability: The broad substrate scope documented in the patent implies that the supply chain is not vulnerable to single-source bottlenecks for specific starting materials. Since the method tolerates a wide array of substituents including halogens, alkyl groups, and electron-withdrawing moieties, procurement teams have the flexibility to source alternative grades of raw materials without compromising the integrity of the final product. This resilience is critical for maintaining continuous supply lines for active pharmaceutical ingredients (APIs), especially in a global market where logistical disruptions can occur unexpectedly. The robustness of the chemistry ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed quality control tests.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-kilogram or ton-scale production is facilitated by the mild thermal profile and the use of common organic solvents like acetonitrile. The absence of hazardous reagents or extreme pressure conditions simplifies the safety assessment and regulatory approval process for new manufacturing facilities. From an environmental standpoint, the atom economy of the cyclization is superior to condensation methods that generate stoichiometric amounts of water or alcohol byproducts, aligning with increasingly strict environmental regulations and corporate sustainability goals. This makes the technology an attractive option for companies looking to minimize their carbon footprint while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and claims presented in CN111018800B, providing a factual basis for decision-making. Whether you are evaluating the feasibility of this route for a specific project or assessing the quality standards of the resulting intermediates, understanding these details is paramount. We encourage technical teams to review these points closely to ensure alignment with their specific development timelines and quality objectives.
Q: What are the primary advantages of this copper-catalyzed method over traditional triazine synthesis?
A: Unlike conventional methods that often require harsh conditions and hydrazide precursors, this patent describes a mild, one-pot cyclization using readily available S,N-substituted olefins and diazonium salts, significantly simplifying the operational complexity and improving functional group tolerance.
Q: Can this synthesis protocol be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes inexpensive copper catalysts and operates at near-ambient temperatures (25-40°C), which minimizes energy consumption and safety risks associated with high-pressure or high-temperature reactors, making it highly suitable for commercial scale-up.
Q: What is the substrate scope for the aryl diazonium salt component?
A: The method demonstrates broad adaptability, accommodating aryl diazonium salts with various substituents including methyl, methoxy, halogens (fluoro, chloro, bromo), and electron-withdrawing groups like nitro or cyano, allowing for the diverse modification of the N2 position.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N2-Aryl-1,2,4-Triazine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN111018800B for advancing pharmaceutical research and development. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust, GMP-compliant manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering high-purity N2-aryl-1,2,4-triazine derivatives that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next drug discovery program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of heterocyclic synthesis and secure a competitive advantage in the global pharmaceutical market through efficient, scalable, and cost-effective manufacturing solutions.
