Advanced Palladium-Catalyzed Synthesis of 2-Phenylquinazolinones for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of 2-Phenylquinazolinones for Scalable Pharmaceutical Manufacturing
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting broad biological activity. Patent CN108033919B introduces a transformative methodology for the synthesis of 2-phenylquinazolinone compounds, leveraging a palladium-catalyzed oxidative cleavage strategy that fundamentally alters the economic and operational landscape of producing these valuable intermediates. This technology addresses the critical industry demand for sustainable, cost-effective processes that utilize abundant feedstocks rather than expensive, pre-functionalized building blocks. By employing styrene derivatives and 2-aminobenzamide as primary starting materials, the process bypasses traditional multi-step sequences, offering a direct pathway to complex heterocyclic cores essential for antimicrobial and anticancer drug development. The strategic integration of molecular oxygen as a green oxidant further underscores the environmental compatibility of this approach, aligning with global initiatives to reduce chemical waste in pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone skeleton has relied heavily on the condensation of 2-aminobenzoic acid derivatives with imido esters or nitriles, methodologies that often suffer from significant practical drawbacks in an industrial setting. These traditional routes frequently necessitate the use of harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to the degradation of sensitive functional groups and complicate downstream purification efforts. Moreover, the requirement for specialized precursors like imido esters introduces substantial supply chain vulnerabilities, as these reagents are often costly, moisture-sensitive, and not available in the bulk quantities required for commercial production. The atom economy of such classical approaches is frequently suboptimal, generating stoichiometric amounts of salt byproducts that require extensive aqueous workups, thereby increasing the volume of wastewater and the overall environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the innovative protocol detailed in the patent utilizes a palladium-catalyzed system to effect the oxidative cleavage of the carbon-carbon double bond in styrene derivatives, generating the requisite aldehyde intermediate in situ for subsequent cyclization. This tandem transformation allows for the direct use of styrenes, which are ubiquitous petrochemical commodities characterized by their low cost and exceptional availability across global markets. The reaction proceeds under remarkably mild conditions, typically at temperatures between 80°C and 110°C under an oxygen atmosphere, eliminating the need for high-pressure equipment or hazardous oxidants like chromates or permanganates. 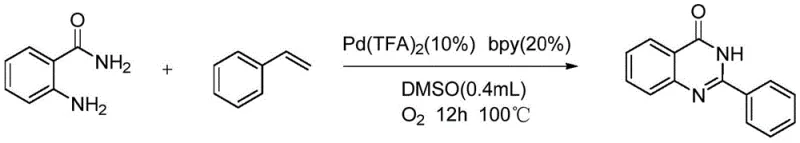 This operational simplicity translates directly into reduced capital expenditure for reactor infrastructure and lower energy consumption per kilogram of product. Furthermore, the use of dimethyl sulfoxide (DMSO) as a polar aprotic solvent facilitates the solubility of both organic substrates and the palladium catalyst, ensuring homogeneous reaction kinetics and consistent product quality across different batches.
This operational simplicity translates directly into reduced capital expenditure for reactor infrastructure and lower energy consumption per kilogram of product. Furthermore, the use of dimethyl sulfoxide (DMSO) as a polar aprotic solvent facilitates the solubility of both organic substrates and the palladium catalyst, ensuring homogeneous reaction kinetics and consistent product quality across different batches.
Mechanistic Insights into Pd-Catalyzed Oxidative Cleavage and Cyclization
The core of this technological breakthrough lies in the sophisticated catalytic cycle mediated by palladium trifluoroacetate in conjunction with a bipyridine ligand, which orchestrates the selective activation of the styrene olefinic bond. The mechanism initiates with the coordination of the palladium species to the alkene moiety of the styrene, followed by nucleophilic attack and subsequent beta-carbon elimination to effect the C-C bond cleavage. This oxidative fragmentation generates a benzaldehyde species transiently within the reaction matrix, which immediately undergoes condensation with the adjacent amine group of the 2-aminobenzamide. The resulting imine intermediate then participates in an intramolecular nucleophilic attack by the amide nitrogen onto the carbonyl carbon, followed by dehydration to aromatize the pyrimidine ring and furnish the final 2-phenylquinazolinone structure.  The presence of molecular oxygen is critical not only for regenerating the active palladium(II) species from the reduced palladium(0) state but also for driving the thermodynamic equilibrium towards the desired oxidation products, ensuring high turnover numbers for the catalyst.
The presence of molecular oxygen is critical not only for regenerating the active palladium(II) species from the reduced palladium(0) state but also for driving the thermodynamic equilibrium towards the desired oxidation products, ensuring high turnover numbers for the catalyst.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of side products commonly associated with stepwise syntheses. The in situ generation of the aldehyde prevents its accumulation and potential polymerization or over-oxidation to benzoic acid, which are common pitfalls when isolating reactive intermediates. The specificity of the palladium catalyst for the terminal alkene ensures that other potentially sensitive functional groups on the aromatic ring, such as halogens or methoxy groups, remain intact, thereby preserving the structural integrity required for downstream biological testing. The rigorous optimization of ligand-to-metal ratios, specifically utilizing 2,2'-bipyridine, stabilizes the palladium center against aggregation into inactive black palladium metal, maintaining catalytic activity throughout the extended reaction period and resulting in a cleaner crude profile that simplifies final purification.
How to Synthesize 2-Phenylquinazolinone Efficiently
The execution of this synthesis requires precise adherence to the optimized parameters regarding catalyst loading, solvent choice, and atmospheric control to achieve the reported high yields. The process begins with the careful charging of 2-aminobenzamide, the palladium trifluoroacetate catalyst, and the bipyridine ligand into a dry reaction vessel, followed by a critical degassing procedure involving three consecutive cycles of vacuum and oxygen filling to establish a strictly aerobic environment essential for catalyst turnover. Subsequent addition of the styrene substrate and DMSO solvent initiates the reaction upon heating to 100°C, where the mixture is stirred for approximately 12 hours to ensure complete conversion. For a comprehensive understanding of the precise operational parameters and safety considerations, the detailed standardized synthesis steps are outlined in the guide below.
- Charge 2-aminobenzamide, palladium trifluoroacetate catalyst, and bipyridine ligand into a reaction vessel, followed by three cycles of vacuum-oxygen exchange to ensure an aerobic environment.
- Add dimethyl sulfoxide (DMSO) as the solvent and the styrene substrate, then heat the mixture to 100°C with stirring for approximately 12 hours to facilitate oxidative C-C bond cleavage and cyclization.
- Upon completion, remove the solvent via evaporation and purify the crude residue using silica gel column chromatography with a dichloromethane and ethyl acetate eluent system to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this styrene-based route represents a strategic opportunity to decouple production costs from the volatility of specialized fine chemical markets. By shifting the raw material base to styrenes, which are produced on a megaton scale for the polymer industry, manufacturers can secure a stable and inexpensive supply of starting materials that is far less susceptible to the price fluctuations typical of niche pharmaceutical intermediates. The elimination of expensive coupling reagents and the reduction in synthetic steps significantly lower the direct material costs, while the use of oxygen from air as the oxidant removes the recurring expense of purchasing stoichiometric chemical oxidants. This fundamental shift in the bill of materials creates a robust buffer against inflationary pressures in the chemical sector, ensuring long-term margin stability for high-volume production campaigns.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the synthetic sequence and the utilization of commodity-grade feedstocks. By avoiding the isolation of unstable aldehyde intermediates and combining the oxidation and cyclization into a single pot, the process reduces labor hours, solvent consumption, and waste disposal costs associated with multiple workup stages. The high atom efficiency of the oxidative cleavage means that a larger proportion of the input mass is converted into the final product, minimizing the cost per kilogram of active pharmaceutical ingredient precursor and enhancing overall process profitability.
- Enhanced Supply Chain Reliability: Relying on styrene derivatives as key building blocks significantly mitigates supply chain risks, as these materials are manufactured by numerous global suppliers with established logistics networks capable of delivering bulk quantities on short notice. This diversification of the supplier base prevents single-source bottlenecks that often plague the procurement of complex heterocyclic precursors, ensuring continuous manufacturing operations even during periods of regional disruption. Furthermore, the stability of the starting materials allows for extended storage without significant degradation, facilitating strategic stockpiling and just-in-time inventory management strategies.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of high-pressure hydrogen or toxic heavy metal oxidants make this process inherently safer and easier to scale from pilot plant to commercial tonnage production. The use of DMSO, a solvent with a high boiling point and favorable safety profile, simplifies containment and recovery systems, while the generation of water as the primary byproduct of the oxidation aligns with increasingly stringent environmental regulations regarding effluent discharge. This green chemistry profile not only reduces the regulatory burden but also enhances the corporate sustainability metrics of the manufacturing entity, appealing to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route, providing clarity on its operational feasibility and strategic value. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, ensuring that the information reflects the actual capabilities and limitations of the technology. Understanding these nuances is essential for R&D teams evaluating process transfer and for commercial teams assessing the viability of this route for long-term supply agreements.
Q: What are the key advantages of using styrene compounds over traditional precursors for quinazolinone synthesis?
A: Utilizing styrene compounds offers significant economic and logistical benefits as they are commodity chemicals that are cheap and readily available compared to specialized imido esters or pre-functionalized aldehydes. Furthermore, this route avoids the need for harsh pre-activation steps, streamlining the supply chain and reducing overall raw material costs.
Q: How does the palladium catalyst loading impact the reaction efficiency and cost?
A: The process utilizes a relatively low catalyst loading of 10 mol% palladium trifluoroacetate, which balances high conversion rates with cost efficiency. While lower loadings might reduce metal costs, the patented optimization ensures that 10% provides the necessary turnover frequency to maintain short reaction times and high yields without requiring excessive reaction durations.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability, operating at atmospheric pressure with oxygen as the terminal oxidant, which eliminates the safety risks associated with high-pressure hydrogenation or hazardous oxidants. The simple workup procedure involving solvent removal and standard chromatography further supports its viability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylquinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic methodologies in securing the supply of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch of 2-phenylquinazolinone derivatives we produce. Our facility is equipped to handle the specific requirements of palladium-catalyzed reactions, including effective metal scavenging protocols to ensure residual metal levels comply with the most demanding international regulatory standards.
We invite you to collaborate with us to optimize your supply chain and leverage the cost efficiencies offered by this advanced synthetic technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to obtain specific COA data for our catalog items and to discuss route feasibility assessments for custom analogues, ensuring that your project timelines are met with precision and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
