Advanced Synthesis of 2-Amino-6-Piperidine Pyridine Compounds for Migraine Therapeutics
Advanced Synthesis of 2-Amino-6-Piperidine Pyridine Compounds for Migraine Therapeutics
The pharmaceutical landscape for migraine treatment has shifted significantly with the advent of selective 5-HT1F receptor agonists, most notably Lasmiditan. As the demand for these novel therapeutics grows, the efficiency of synthesizing their key precursors becomes a critical bottleneck for global supply chains. Patent CN110981854B discloses a robust and innovative methodology for producing 2-amino-6-(1-alkylpiperidine-4-carbonyl)pyridine compounds, which serve as essential building blocks for this class of drugs. This technical insight explores the mechanistic advantages of this new route, contrasting it with legacy methods to highlight its potential for cost-effective and sustainable manufacturing. By leveraging specific organometallic exchange reactions, this process addresses long-standing issues regarding yield and purity that have plagued earlier synthetic attempts.
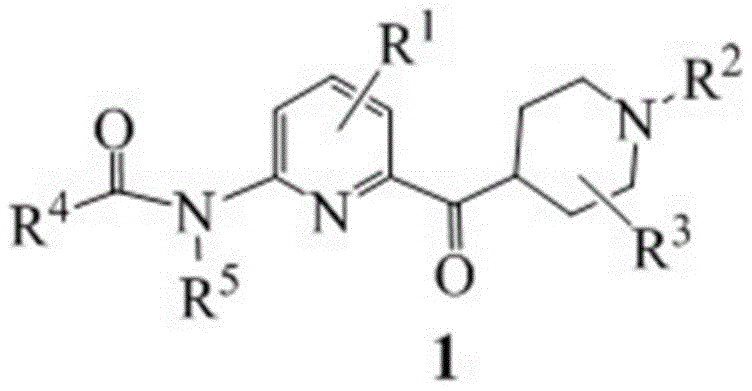
The core innovation lies in the strategic manipulation of the pyridine ring substitution pattern. Traditional approaches often struggle with regioselectivity and the harsh conditions required to install the carbonyl-piperidine moiety at the 6-position while maintaining an amino group at the 2-position. The disclosed method utilizes a protected N-(6-bromopyridin-2-yl)amide as a stable starting point, allowing for precise metal-halogen exchange. This precision is vital for R&D directors focused on impurity profiles, as it minimizes the formation of regioisomers that are notoriously difficult to separate. The versatility of the R groups defined in the patent suggests this platform technology can be adapted for various analogues, providing a flexible foundation for medicinal chemistry campaigns aimed at optimizing pharmacokinetic properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as those described in WO2003/084949, present significant hurdles for industrial scale-up. Route A, for instance, relies on the lithiation of 2-chloropyridine followed by reaction with a piperidine formamide. However, this pathway is marred by extremely low yields, often hovering around merely 10 percent, which is economically unviable for commercial production. Furthermore, the subsequent conversion of the chloro-group to an amino group necessitates the use of expensive palladium catalysts (Pd2dba3) and bulky phosphine ligands like BINAP. These noble metals not only drive up raw material costs but also introduce stringent requirements for residual metal control in the final API, complicating the purification workflow and increasing the burden on quality control laboratories.
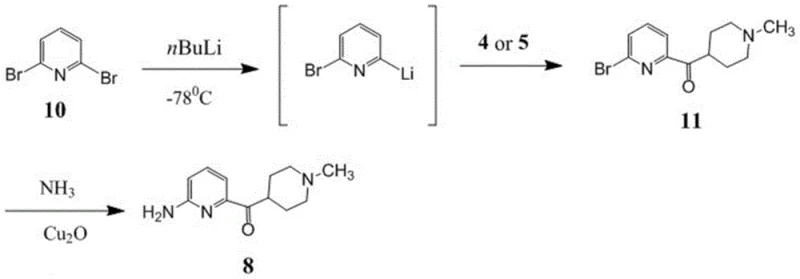
Route B attempts to mitigate some issues by using 2,6-dibromopyridine as a starting material, yet it introduces new inefficiencies. To achieve acceptable conversion, this method requires a substantial excess of the dibromopyridine starting material, typically 1.5 equivalents or more. This results in poor atom economy, as the molecular weight of the unused bromine leaving group constitutes a significant portion of the input mass that ends up as waste. Additionally, the ammoniation step in Route B often involves high-pressure conditions with excess ammonia, generating large volumes of waste gas and wastewater. The separation of the desired product from unreacted starting materials and oily by-products is complex, often requiring column chromatography which is impractical for multi-kilogram or ton-scale manufacturing.
The Novel Approach
The methodology presented in CN110981854B offers a transformative solution by reordering the synthetic logic. Instead of struggling with late-stage amination or inefficient lithiation of unprotected pyridines, the new route begins with a pre-functionalized N-(6-bromopyridin-2-yl)amide. This protecting group strategy stabilizes the nitrogen atom, preventing unwanted side reactions during the subsequent organometallic step. The process employs a metal-halogen exchange using either Grignard reagents (like isopropyl magnesium chloride) or organolithium reagents (like n-butyllithium) under strictly controlled thermal conditions. This exchange generates a highly reactive pyridine-metal species in situ, which then undergoes a nucleophilic addition to a Weinreb amide or ester derivative of the piperidine ring.
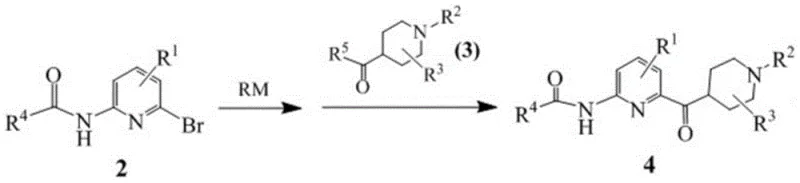
This approach dramatically improves the overall yield and operational simplicity. By avoiding the need for excess dibromopyridine and eliminating the high-pressure ammoniation step, the process reduces both material consumption and environmental impact. The reaction temperatures are optimized to balance reactivity and selectivity; for example, Grignard exchanges are conducted at mild temperatures between 0°C and 5°C, whereas lithiation requires cryogenic conditions of -78°C to -70°C. The resulting protected intermediate can be isolated with high purity, often through simple crystallization rather than chromatography. Finally, the removal of the amide protecting group via hydrolysis is straightforward, utilizing common acids or bases to reveal the free amine, thus delivering the target 2-amino-6-(1-alkylpiperidine-4-carbonyl)pyridine compound in high yield and purity suitable for downstream coupling.
Mechanistic Insights into Organometallic Exchange and Addition
The success of this synthesis hinges on the precise control of the metal-halogen exchange equilibrium. When using isopropyl magnesium chloride lithium chloride complexes (Turbo Grignard), the exchange occurs rapidly even at near-ambient temperatures. The presence of LiCl enhances the solubility and reactivity of the organomagnesium species, facilitating the formation of the 2-pyridyl magnesium intermediate. This intermediate is then immediately trapped by the electrophilic carbonyl of the piperidine Weinreb amide. The mechanism proceeds through a tetrahedral intermediate which, due to the chelating nature of the methoxy-methyl-amino group, remains stable until acidic workup. This stability is crucial as it prevents over-addition of the Grignard reagent, which would otherwise lead to tertiary alcohol impurities that are difficult to remove.
Impurity control is further enhanced by the choice of the pivaloyl protecting group on the pyridine nitrogen. This bulky group sterically hinders nucleophilic attack at the amide carbonyl of the protecting group itself, ensuring that the organometallic reagent attacks only the intended piperidine electrophile. In contrast, less hindered protecting groups might succumb to nucleophilic degradation, leading to complex mixtures. The hydrolysis step is equally critical; using dilute hydrochloric acid or sodium hydroxide cleaves the pivaloyl group efficiently. The patent data indicates that molar ratios of acid or base are carefully tuned (4 to 8 equivalents for acid, 3 to 5 for base) to ensure complete deprotection without degrading the sensitive ketone linkage between the pyridine and piperidine rings. This mechanistic understanding allows process chemists to fine-tune reaction parameters for maximum robustness.
How to Synthesize 2-Amino-6-(1-alkylpiperidine-4-carbonyl)pyridine Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for laboratory and pilot-scale production. The process begins with the preparation of the bromo-pyridine precursor, followed by the critical metal exchange and addition sequence. Operators must maintain strict temperature control during the addition of the organometallic reagent to manage exotherms and prevent decomposition. The subsequent hydrolysis step requires careful pH adjustment to isolate the free base or salt form of the product. For detailed operational parameters, stoichiometry, and safety considerations regarding the handling of pyrophoric reagents, please refer to the standardized guide below.
- Perform a metal-halogen exchange on N-(6-bromopyridin-2-yl)amide using an organometallic reagent (RM) such as isopropyl magnesium chloride or n-butyllithium at controlled low temperatures.
- React the resulting pyridine metal species with a 1-alkylpiperidine-4-formamide derivative to form the protected ketone intermediate.
- Execute a hydrolysis step using acid or base to remove the amide protecting group, yielding the final 2-amino-6-(1-alkylpiperidine-4-carbonyl)pyridine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for exotic palladium catalysts and specialized phosphine ligands, the bill of materials becomes significantly more stable and less susceptible to geopolitical supply shocks affecting precious metals. Furthermore, the avoidance of high-pressure ammoniation equipment reduces the capital expenditure required for reactor infrastructure, allowing production to be outsourced to a wider range of CDMO partners who may not possess specialized high-pressure hydrogenation or ammoniation suites. This flexibility enhances supply chain resilience and reduces the risk of production bottlenecks.
- Cost Reduction in Manufacturing: The new route achieves substantial cost savings by improving atom economy and reducing raw material waste. Unlike previous methods that required a 50 percent excess of expensive dibromopyridine, this method utilizes stoichiometric or near-stoichiometric amounts of reagents. The elimination of noble metal catalysts removes a major cost driver and the associated expense of metal scavenging resins required to meet regulatory limits. Additionally, the ability to isolate intermediates via crystallization rather than silica gel chromatography significantly lowers solvent consumption and waste disposal costs, leading to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commodity chemicals such as isopropyl magnesium chloride and n-butyllithium, which are available from multiple global suppliers in bulk quantities. The process does not rely on custom-synthesized building blocks that might have long lead times. The robustness of the reaction conditions, particularly the tolerance for slightly varied temperatures in the Grignard variant, means that batch-to-batch variability is minimized. This consistency ensures that delivery schedules can be met with greater certainty, reducing the need for safety stock and allowing for just-in-time inventory strategies for downstream API synthesis.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is inherently greener. The removal of the high-pressure ammonia step eliminates a significant source of hazardous waste gas, simplifying permitting and compliance with local environmental regulations. The reduction in solvent usage, driven by the switch from chromatographic purification to crystallization, lowers the facility's carbon footprint and waste treatment burden. These factors make the technology highly scalable, capable of being transferred from kilogram-scale R&D batches to multi-ton commercial production with minimal re-engineering, ensuring a smooth ramp-up to meet market demand for migraine therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific improvements noted in the patent documentation compared to prior art. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific supply chain needs. The answers reflect the practical realities of scaling organometallic chemistry in a GMP environment.
Q: What are the primary limitations of conventional Lasmiditan intermediate synthesis routes?
A: Conventional routes often suffer from low yields (around 10% in early lithiation steps), require expensive noble metal catalysts like palladium, and utilize excessive amounts of starting materials like 2,6-dibromopyridine, leading to poor atom economy and difficult purification processes.
Q: How does the new organometallic exchange method improve process safety and efficiency?
A: The new method utilizes controlled temperature metal-halogen exchange (0-5°C for Grignard or -78°C for Lithium) which minimizes side reactions. It eliminates the need for high-pressure ammoniation steps that generate significant waste gas, thereby simplifying the downstream separation and reducing environmental hazards.
Q: Is this synthesis route scalable for commercial API production?
A: Yes, the route is designed for scalability by avoiding complex chromatographic purifications. The intermediates can often be isolated via crystallization, and the use of standard organometallic reagents allows for adaptation to large-scale reactors with established safety protocols for exothermic management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-6-(1-alkylpiperidine-4-carbonyl)pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation migraine treatments depends on a secure and high-quality supply of key intermediates. Our technical team has extensively analyzed the route disclosed in CN110981854B and possesses the expertise to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to full-scale market launch. Our facilities are equipped with state-of-the-art cryogenic reactors for lithiation and robust containment systems for Grignard reagents, guaranteeing adherence to stringent purity specifications and rigorous QC labs that monitor every critical parameter.
We invite you to collaborate with us to optimize your supply chain for Lasmiditan and related 5-HT1F agonists. Our engineering team can provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this new synthetic route can lower your overall cost of goods. We encourage you to contact our technical procurement team to request specific COA data from our pilot batches and to discuss route feasibility assessments for your specific derivative targets. Let us be your partner in delivering high-purity pharmaceutical intermediates that power the future of neurological medicine.
