Advanced VAPOL-Based Selenium-Sulfur Catalysts for High-Precision Asymmetric Synthesis
Introduction to Next-Generation Chiral Catalysis
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for higher enantioselectivity and more robust catalytic systems in the production of complex organic molecules. A significant breakthrough in this domain is documented in patent CN115155658B, which discloses a sophisticated synthetic method for generating selenium-sulfur compounds based on the VAPOL (3,3'-diphenyl-[2,2'-binaphthalene]-1,1'-diol) framework. This innovation addresses the limitations of existing ligand libraries by introducing heavy chalcogen atoms (selenium and sulfur) into the axial chiral VAPOL structure, thereby creating a new class of catalysts with unique electronic properties. The technology leverages the well-established steric bulk of the VAPOL backbone while enhancing Lewis basicity and coordination capabilities through the incorporation of selenium or sulfur, offering a powerful tool for R&D teams focused on developing novel pharmaceutical intermediates and fine chemicals.
For procurement and supply chain leaders, the significance of this patent extends beyond mere chemical novelty; it represents a shift towards more accessible and economically viable catalytic solutions. The described method utilizes cheap and easily prepared raw materials, avoiding the complex multi-step routes often associated with specialized ligand synthesis. By enabling the preparation of diverse catalyst variants through simple alkylamine substitution, this technology provides a flexible platform for optimizing reaction conditions without incurring prohibitive costs. As a reliable chiral catalyst supplier, understanding these underlying technological shifts is crucial for securing a competitive edge in the global market for high-value asymmetric synthesis reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of axially chiral catalysts has relied heavily on the BINOL (1,1'-bi-2-naphthol) framework, which, while effective, presents certain limitations in terms of structural diversity and specific catalytic activities for sulfur-involved transformations. Conventional methods for functionalizing these backbones often require harsh reaction conditions, expensive transition metal catalysts, or tedious protection-deprotection sequences that drive up manufacturing costs and extend lead times. Furthermore, the introduction of heavier chalcogen atoms like selenium into these rigid frameworks has historically been challenging, limiting the exploration of selenium-based organocatalysts which possess distinct redox properties and nucleophilicity compared to their oxygen or nitrogen counterparts. These bottlenecks often result in lower overall yields and complicate the purification process, creating significant hurdles for the commercial scale-up of complex organocatalysts in an industrial setting.
The Novel Approach
The methodology outlined in patent CN115155658B offers a transformative solution by employing a streamlined, one-pot synthetic strategy that bypasses these traditional complexities. The core innovation lies in the direct reaction of chiral VAPOL with phosphorus trichloride, triethylamine, and various alkylamines, followed by the introduction of elemental selenium or sulfur powder. This approach not only simplifies the operational workflow but also allows for the rapid generation of a diverse library of catalysts by merely varying the alkylamine component. The reaction proceeds under mild conditions, typically initiating at low temperatures (around 0°C) and warming to room temperature, which minimizes energy consumption and reduces the risk of side reactions. 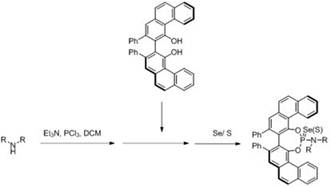 This efficient pathway ensures high-purity chiral catalysts can be obtained with excellent repeatability, making it an ideal candidate for cost reduction in fine chemical manufacturing where process efficiency is paramount.
This efficient pathway ensures high-purity chiral catalysts can be obtained with excellent repeatability, making it an ideal candidate for cost reduction in fine chemical manufacturing where process efficiency is paramount.
Mechanistic Insights into VAPOL-Derived Selenium-Sulfur Catalysis
The mechanistic superiority of these VAPOL-derived selenium-sulfur catalysts stems from the synergistic combination of axial chirality and the unique electronic characteristics of the phosphorus-selenium/sulfur moiety. The VAPOL backbone provides a rigid, sterically demanding environment that effectively differentiates between enantiotopic faces of the substrate, a critical factor in achieving high enantioselectivity. Upon incorporation of selenium or sulfur, the catalyst gains enhanced nucleophilic character and the ability to engage in specific non-covalent interactions, such as chalcogen bonding, which can activate electrophiles more effectively than traditional oxygen-based analogues. This is particularly evident in reactions involving aryl sulfides, where the soft nature of the selenium or sulfur atom in the catalyst complements the substrate, facilitating smoother transition states and faster reaction kinetics.
Furthermore, the versatility of this catalytic system is demonstrated by its successful application in the synthesis of six-membered ring chiral lactones and axial chiral sulfur-containing diphenyl derivatives. In the lactone synthesis, the catalyst likely activates the thioaryl species through a Lewis acid-base interaction, promoting the cyclization with high stereocontrol. 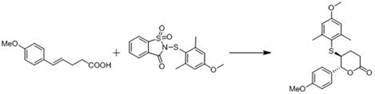 The ability to tune the steric environment around the active site by selecting different alkylamines (e.g., diisopropylamine, diethylamine) allows chemists to fine-tune the catalyst's performance for specific substrates. This level of mechanistic control is essential for R&D directors aiming to optimize impurity profiles and maximize yield in the production of sensitive pharmaceutical intermediates, ensuring that the final products meet stringent regulatory standards for chirality and purity.
The ability to tune the steric environment around the active site by selecting different alkylamines (e.g., diisopropylamine, diethylamine) allows chemists to fine-tune the catalyst's performance for specific substrates. This level of mechanistic control is essential for R&D directors aiming to optimize impurity profiles and maximize yield in the production of sensitive pharmaceutical intermediates, ensuring that the final products meet stringent regulatory standards for chirality and purity.
How to Synthesize VAPOL Selenium-Sulfur Catalysts Efficiently
The synthesis protocol described in the patent is designed for robustness and ease of execution, making it highly suitable for both laboratory-scale optimization and larger production runs. The process begins with the activation of phosphorus trichloride in an anhydrous solvent like dichloromethane, followed by the controlled addition of base and amine components to form the reactive phosphorus intermediate. Subsequent addition of the VAPOL substrate and elemental selenium or sulfur completes the catalyst assembly. This modular approach allows for the rapid screening of different catalyst variants to identify the optimal performer for a given transformation. For detailed operational parameters, stoichiometry, and purification techniques, please refer to the standardized guide below.
- Dissolve phosphorus trichloride in dichloromethane and cool to 0°C, then add triethylamine and warm to room temperature.
- Add the selected alkylamine and stir, followed by the addition of chiral VAPOL substrate at room temperature.
- Introduce selenium or sulfur powder to the mixture, stir for reaction completion, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this VAPOL-based selenium-sulfur catalyst technology offers substantial strategic benefits for procurement and supply chain management. The reliance on economically available starting materials, such as commercial VAPOL and common alkylamines, drastically reduces the raw material cost burden compared to proprietary or rare-metal catalysts. Additionally, the one-pot nature of the synthesis eliminates the need for intermediate isolation and purification steps, which translates to significant savings in solvent usage, labor hours, and waste disposal costs. This streamlined process enhances the overall economic viability of producing high-value chiral intermediates, allowing manufacturers to offer more competitive pricing without compromising on quality or performance.
- Cost Reduction in Manufacturing: The elimination of complex multi-step synthetic routes and the use of inexpensive reagents like sulfur powder and phosphorus trichloride directly lower the cost of goods sold (COGS). By avoiding the use of precious transition metals, the process also removes the need for expensive metal scavenging and removal steps, further reducing downstream processing costs and simplifying the regulatory filing for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Since the raw materials required for this synthesis are commodity chemicals with stable global supply chains, the risk of supply disruption is minimized. The robustness of the reaction conditions, which tolerate room temperature operations after initiation, ensures consistent batch-to-batch reproducibility. This reliability is critical for maintaining continuous production schedules and meeting the tight delivery windows demanded by downstream clients in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metals align well with green chemistry principles, facilitating easier environmental compliance and waste management. The process is inherently scalable, as the exothermic nature of the initial steps can be managed effectively in larger reactors, supporting the transition from gram-scale R&D to ton-scale commercial production without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and sourcing of these advanced catalytic systems. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing workflows. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industrial applications.
Q: What are the primary advantages of VAPOL-derived selenium-sulfur catalysts over traditional BINOL ligands?
A: VAPOL frameworks offer distinct steric and electronic properties compared to BINOL, and the introduction of selenium or sulfur atoms enhances catalytic activity for specific transformations like six-membered ring lactone synthesis, providing new options for enantioselective reactions.
Q: Is the synthesis process for these catalysts scalable for industrial production?
A: Yes, the patent describes a one-pot synthesis method using commercially available raw materials like VAPOL and alkylamines under mild conditions (0°C to room temperature), which significantly simplifies the process and facilitates commercial scale-up.
Q: What types of chemical reactions can these catalysts effectively promote?
A: These catalysts demonstrate excellent activity in the synthetic reaction of six-membered ring chiral lactones participated by aryl sulfides and the synthesis of axial chiral sulfur-containing diphenyl derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable VAPOL Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced catalytic technologies play in driving innovation within the fine chemical and pharmaceutical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Whether you require custom synthesis of specific VAPOL derivatives or large-scale supply of established catalysts, our infrastructure is designed to support your growth and technical requirements.
We invite you to collaborate with us to explore how these novel selenium-sulfur catalysts can enhance your synthetic routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact us to request specific COA data, route feasibility assessments, and volume pricing, and let us partner with you to achieve superior efficiency and quality in your chemical manufacturing processes.
