Advanced Chiral Binaphthyl-Aza Ligands: Scalable Synthesis for High-Performance Asymmetric Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for higher enantiomeric purity in pharmaceutical intermediates. Patent CN112142638A introduces a groundbreaking class of chiral binaphthyl-aza polycyclic ligands that uniquely combine axial chirality from the binaphthyl backbone with central chirality from nitrogen-containing heterocycles. This dual-chirality architecture addresses a critical gap in current catalytic technologies, offering superior stereocontrol compared to traditional mono-chiral ligands. The invention details a robust preparation method starting from readily available 1,1'-binaphthyl-2,2'-diphenol, utilizing a sequence of protection, nickel-catalyzed functionalization, and nucleophilic coupling. For R&D directors seeking reliable chiral ligands supplier partnerships, this technology represents a significant leap forward in designing catalysts that deliver both high reactivity and exceptional enantioselectivity in complex organic transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing chiral ligands often suffer from structural rigidity or limited tunability, which restricts their application scope in diverse asymmetric reactions. Many existing binaphthyl-based ligands rely solely on axial chirality, which may not provide sufficient steric hindrance or electronic modulation for challenging substrates. Furthermore, conventional synthetic routes frequently involve harsh conditions, expensive transition metal catalysts that are difficult to remove, or multi-step sequences with poor overall yields. These inefficiencies lead to increased production costs and supply chain vulnerabilities, making cost reduction in pharmaceutical intermediates manufacturing a persistent challenge. The inability to easily modify the electronic properties of the ligand backbone often results in suboptimal catalytic performance, necessitating extensive trial-and-error screening that delays project timelines.
The Novel Approach
The novel approach described in the patent overcomes these hurdles by introducing a modular synthetic strategy that allows for precise tuning of both steric and electronic environments. By incorporating an aza-polycyclic moiety, such as a pyrrolidine or piperidine ring derived from amino acids, the ligand gains a second chiral center that works synergistically with the binaphthyl axis. This design enhances the coordination ability with transition metals through multiple donor atoms (nitrogen and oxygen), leading to more stable catalytic complexes. The synthetic route is streamlined, avoiding exotic reagents in favor of standard organic transformations like methylation, triflation, and Grignard additions. This simplicity not only improves the overall yield but also facilitates the commercial scale-up of complex chiral ligands, ensuring a consistent supply of high-purity materials for industrial applications.
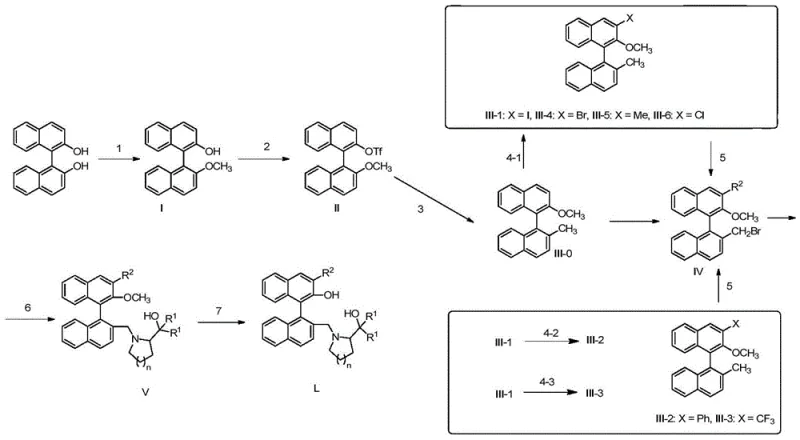
Mechanistic Insights into Nickel-Catalyzed Functionalization and Coupling
The core of this synthesis lies in the strategic functionalization of the binaphthyl skeleton, particularly the nickel-catalyzed methylation and subsequent lithiation steps. The process begins with the selective protection of one phenolic hydroxyl group, followed by triflation of the other to create a leaving group for cross-coupling. The use of NiCl2(Dppp) as a catalyst for the methylation with methylmagnesium bromide is a critical innovation, allowing for the introduction of a methyl group under mild conditions with high regioselectivity. This step is pivotal for establishing the steric bulk necessary for effective chirality transfer. Subsequent lithiation at low temperatures (-78°C) enables the introduction of various substituents (iodine, phenyl, trifluoromethyl) at the 3-position, providing a versatile platform for library generation. This level of control ensures that the resulting ligands can be tailored for specific reaction requirements, enhancing their utility in reducing lead time for high-purity chiral intermediates.
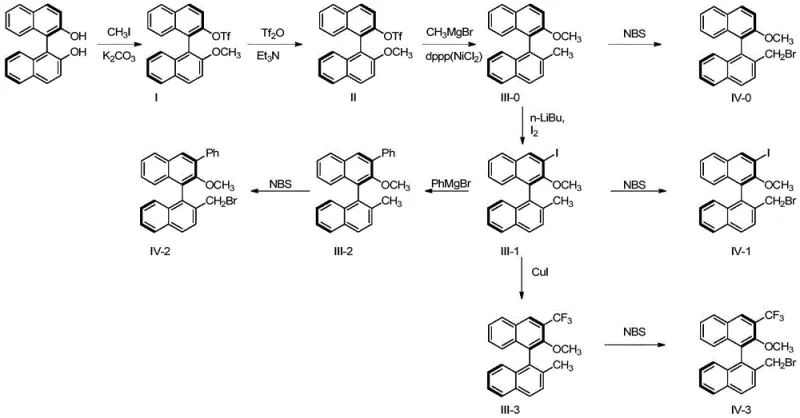
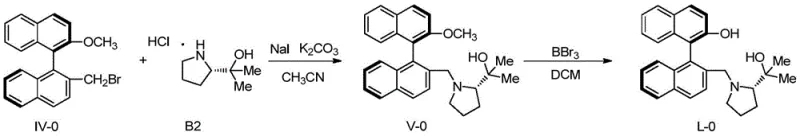
Following the backbone modification, the installation of the chiral amine component is achieved through a nucleophilic substitution reaction. The bromomethyl intermediate, generated via radical bromination with NBS, reacts with chiral amino alcohols derived from proline or pipecolic acid. This coupling step is facilitated by base and phase transfer catalysts, ensuring efficient bond formation without racemization of the sensitive chiral centers. The final demethylation using strong Lewis acids like BBr3 reveals the free phenolic hydroxyl group, completing the ligand structure. The patent data demonstrates that these ligands exhibit remarkable performance, with ligand L-1 achieving 96% ee in the addition of diethylzinc to benzaldehyde. This high level of stereocontrol validates the mechanistic design, proving that the combination of axial and central chirality effectively directs the approach of substrates to the metal center.
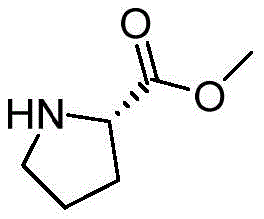
How to Synthesize Chiral Binaphthyl-Aza Ligands Efficiently
The synthesis of these advanced ligands follows a logical, seven-step protocol that balances chemical efficiency with operational safety. The process leverages widely available starting materials and standard laboratory equipment, making it accessible for both pilot-scale and large-scale production. Key to the success of this route is the careful control of reaction temperatures during the lithiation and Grignard steps to prevent side reactions. The purification methods, primarily column chromatography and recrystallization, are well-established techniques that ensure the final product meets stringent purity specifications required for catalytic applications. Detailed standardized synthesis steps are outlined below to guide process development teams in replicating these high-performance materials.
- Protect the phenolic hydroxyl group of binaphthol using methyl iodide and potassium carbonate.
- Perform nickel-catalyzed methylation followed by lithiation and halogenation to functionalize the aromatic ring.
- Couple the bromomethyl intermediate with chiral amino alcohols under basic conditions, followed by demethylation to yield the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits beyond mere technical performance. The reliance on commodity chemicals like BINOL, methyl iodide, and common amino acids significantly mitigates the risk of raw material shortages. Unlike processes dependent on scarce precious metals or proprietary reagents, this method utilizes nickel and magnesium, which are abundant and cost-effective. This shift in reagent profile translates directly into substantial cost savings in fine chemical manufacturing, as the input costs are lower and more predictable. Furthermore, the robustness of the reaction conditions reduces the likelihood of batch failures, enhancing supply chain reliability and ensuring consistent delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts in favor of nickel-based systems drastically lowers the catalyst cost per kilogram of product. Additionally, the high yields reported in the patent examples minimize waste generation and reduce the burden on purification processes. By streamlining the synthesis into fewer distinct operations with high atom economy, the overall production cost is significantly optimized, allowing for more competitive pricing in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of stable intermediates that can be stored or transported safely adds a layer of resilience to the supply chain. The synthetic pathway avoids highly unstable or hazardous intermediates that require specialized handling, simplifying logistics and storage requirements. This stability ensures that production can be maintained continuously, even in fluctuating market conditions, providing partners with a dependable source of critical chiral building blocks for their own API synthesis workflows.
- Scalability and Environmental Compliance: The solvent systems employed, such as THF, DCM, and acetonitrile, are standard in the industry and have well-defined recovery and recycling protocols. This facilitates adherence to environmental regulations regarding volatile organic compound (VOC) emissions. The scalability of the process is evidenced by the straightforward workup procedures, which do not require complex separation technologies. This makes the transition from gram-scale laboratory synthesis to ton-scale commercial production seamless, supporting long-term growth strategies for manufacturers of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of these chiral ligands. Understanding these aspects is crucial for integrating this technology into existing production pipelines. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this opportunity.
Q: What is the enantioselectivity performance of these new ligands?
A: According to patent CN112142638A, these ligands demonstrate exceptional stereocontrol, achieving up to 96% ee in the asymmetric addition of diethylzinc to benzaldehyde.
Q: Are the raw materials for this synthesis readily available?
A: Yes, the synthesis starts from commercially available 1,1'-binaphthyl-2,2'-diphenol (BINOL) and common amino acids like proline, ensuring a stable supply chain.
Q: Can this process be scaled for industrial production?
A: The patent describes robust reaction conditions using standard reagents and solvents like THF and DCM, indicating high feasibility for commercial scale-up without exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Binaphthyl Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral binaphthyl-aza ligands described in CN112142638A for the future of asymmetric synthesis. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate these patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of ligand delivered meets the highest industry standards, enabling your catalytic processes to perform at their peak efficiency.
We invite you to collaborate with us to explore how these advanced ligands can enhance your drug discovery and development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline to market while optimizing your overall production budget.
