Advanced Nickel-Catalyzed Enamine Synthesis for Scalable Pharmaceutical Manufacturing
Advanced Nickel-Catalyzed Enamine Synthesis for Scalable Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is documented in patent CN110041207B, which details a novel method for catalytically synthesizing enamine compounds using a nickel imine complex containing a pyridine ligand. This technology addresses critical bottlenecks in traditional enamine production, offering a robust pathway for generating high-value nitrogen-containing heterocyclic derivatives. For R&D directors and procurement specialists alike, this innovation represents a pivotal shift towards sustainable chemistry that does not compromise on yield or purity. By leveraging a divalent nickel imine complex, the process achieves exceptional atom economy and operational simplicity, making it an ideal candidate for integration into modern supply chains focused on reliability and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enamine compounds has been fraught with significant technical and economic challenges that hinder large-scale industrial adoption. Traditional routes often rely on condensation reactions, heterocyclic ring cracking, or complex rearrangement processes like the Curtius rearrangement, which impose severe constraints on reaction conditions. These legacy methods frequently require extreme temperatures, high pressures, or cryogenic environments, leading to excessive energy consumption and safety hazards. Furthermore, conventional catalysts often exhibit poor tolerance to functional groups, limiting the diversity of substrates that can be utilized. The stringent requirement for absolute absence of water and oxygen necessitates specialized equipment and rigorous inert atmosphere protocols, drastically increasing capital expenditure and operational complexity. Additionally, the limited variety of compatible amino reagents and the generation of substantial waste residues further exacerbate the environmental footprint and cost burden associated with these outdated synthetic strategies.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN110041207B introduces a paradigm shift by utilizing a specialized nickel imine complex containing a pyridine ligand to catalyze the hydroamination of cyclohexylacetylene. This innovative approach operates under remarkably mild conditions, typically between 60°C and 100°C, eliminating the need for energy-intensive heating or cooling systems. The reaction proceeds with 100% atom economy, meaning every atom of the starting materials is incorporated into the final product, thereby generating zero by-products and avoiding the formation of hazardous waste residues. This green chemistry profile is complemented by the catalyst's ability to function effectively in standard aromatic hydrocarbon solvents like toluene, without the need for exotic or expensive reaction media. The versatility of this system allows for the use of various secondary amines, including methyl, ethyl, isopropyl, and phenyl substituted variants, broadening the scope of accessible enamine derivatives for pharmaceutical applications.
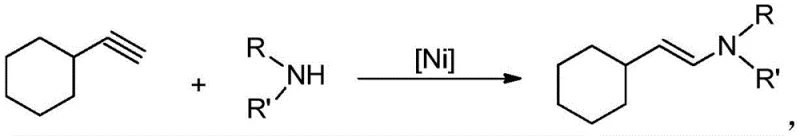
Mechanistic Insights into Nickel-Catalyzed Hydroamination
The core of this technological advancement lies in the unique structure and stability of the divalent nickel imine complex catalyst, represented by the molecular formula [R1R2C(C5H4N)2]Ni=NPh. Unlike many transition metal catalysts that are sensitive to air and moisture, this specific nickel complex exhibits remarkable physical and chemical stability, maintaining its catalytic activity even when exposed to ambient atmospheric conditions. This robustness is attributed to the strong coordination between the nickel center and the pyridine ligands, which stabilizes the metal in its active oxidation state throughout the reaction cycle. The catalyst facilitates the addition of the secondary amine across the triple bond of cyclohexylacetylene with high precision, ensuring exclusive formation of the anti-Markov addition product. This regioselectivity is crucial for pharmaceutical synthesis, where the position of the double bond and the nitrogen substituent can dramatically influence the biological activity and downstream reactivity of the intermediate.
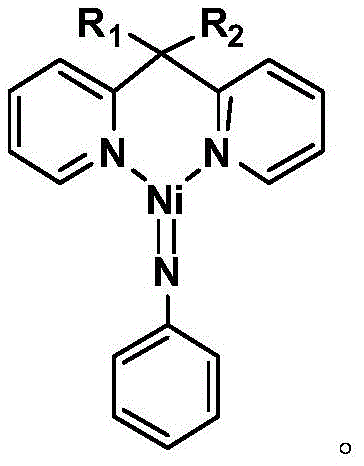
From an impurity control perspective, the mechanism of this hydroamination reaction offers distinct advantages over traditional acid-catalyzed condensations. Because the reaction is additive in nature and does not involve the elimination of small molecules like water or alcohol, there is no equilibrium to drive, and no side reactions associated with dehydration occur. The high selectivity of the nickel catalyst minimizes the formation of regioisomers or oligomeric by-products that often complicate purification in conventional processes. Consequently, the crude reaction mixture is significantly cleaner, reducing the load on downstream purification steps such as column chromatography or recrystallization. This inherent purity profile not only enhances the overall yield, which consistently ranges from 89% to 97% across different substrates, but also ensures that the final enamine product meets the stringent quality specifications required for active pharmaceutical ingredient (API) manufacturing.
How to Synthesize Enamine Efficiently
The practical implementation of this catalytic system is designed for ease of operation, making it highly attractive for process chemists aiming to scale up production. The general procedure involves simply mixing cyclohexylacetylene and the chosen secondary amine in a solvent like toluene, followed by the addition of the nickel catalyst. The reaction mixture is then heated to the specified temperature, typically between 60°C and 100°C, and maintained for a duration of 6 to 12 hours depending on the specific amine substrate. Post-reaction workup is straightforward, involving standard extraction with ethyl acetate and water, drying over anhydrous sodium sulfate, and concentration. The resulting crude product can be purified via silica gel column chromatography or recrystallization to afford the high-purity enamine. For detailed standardized operating procedures and specific parameter optimization, please refer to the guide below.
- Combine cyclohexylacetylene and the selected secondary amine in an aromatic hydrocarbon solvent such as toluene.
- Add the pyridine ligand-containing nickel imine complex catalyst to the reaction mixture under standard atmospheric conditions.
- Heat the reaction mixture to a temperature between 60°C and 100°C for 6 to 12 hours to complete the hydroamination.
- Perform post-treatment involving extraction with ethyl acetate and water, drying, concentration, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed hydroamination technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of harsh reaction conditions and the removal of the need for inert atmosphere handling significantly lower the barrier to entry for manufacturing these intermediates. This simplification of the process infrastructure reduces both capital investment in specialized reactors and the ongoing operational costs associated with maintaining strict environmental controls. Furthermore, the use of readily available and inexpensive starting materials, such as cyclohexylacetylene and common secondary amines, ensures a stable and resilient supply chain that is less susceptible to market volatility. The high atom economy and lack of by-products mean that raw material utilization is maximized, directly contributing to a reduction in the cost of goods sold (COGS) and minimizing waste disposal fees.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the drastic simplification of the synthetic route and the high efficiency of the catalyst. By achieving near-quantitative yields and 100% atom economy, the process minimizes the loss of valuable raw materials, which is a major cost driver in fine chemical synthesis. The catalyst itself, while sophisticated, is used in relatively low loading and can potentially be recovered or optimized for reuse, further driving down unit costs. Additionally, the simplified workup procedure reduces the consumption of solvents and purification media, leading to substantial savings in utility and material expenses. These factors combined create a highly competitive cost structure that allows for significant margin improvement or more aggressive pricing strategies in the marketplace.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex synthesis routes that rely on scarce reagents or fragile process conditions. This new method mitigates those risks by utilizing robust catalysts that are stable in air and common solvents that are widely available globally. The tolerance of the reaction to various functional groups means that supply chain disruptions regarding specific, highly specialized amines can be managed by switching to alternative substrates without redesigning the entire process. Moreover, the mild reaction conditions reduce the risk of batch failures due to thermal runaway or equipment malfunction, ensuring consistent delivery schedules. This reliability is critical for long-term contracts with pharmaceutical clients who demand uninterrupted supply of key intermediates for their own production lines.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden complexities, but this hydroamination method is inherently scalable due to its simplicity and safety profile. The absence of high-pressure requirements and the use of non-hazardous solvents facilitate a smoother transition to larger reactor volumes without extensive re-engineering. From an environmental standpoint, the green nature of the process aligns perfectly with increasingly stringent global regulations on chemical manufacturing emissions and waste. The generation of zero by-products and the avoidance of toxic heavy metal waste streams simplify regulatory compliance and reduce the environmental liability of the manufacturing site. This sustainability advantage is becoming a key differentiator in vendor selection processes for major multinational corporations committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed enamine synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows or for assessing the quality of intermediates sourced from suppliers utilizing this advanced chemistry.
Q: What are the stability characteristics of the nickel imine complex catalyst?
A: The catalyst exhibits high physical and chemical stability, remaining effective even in the presence of air and at elevated temperatures, which eliminates the need for strict anhydrous or oxygen-free environments.
Q: Does this method produce significant by-products or waste?
A: No, the reaction boasts 100% atom economy with no by-products generated, significantly reducing waste residue and simplifying the purification process compared to traditional methods.
Q: What is the regioselectivity of the hydroamination reaction?
A: The reaction demonstrates excellent regioselectivity, exclusively yielding anti-Markov addition products with high yields ranging from 89% to 97% across various substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the nickel imine complex system described in CN110041207B. As a leading CDMO and supplier in the fine chemical sector, we possess the technical expertise and infrastructure to rapidly adopt and optimize such cutting-edge synthetic routes for our global clientele. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of enamine intermediate meets the highest standards required for pharmaceutical applications, providing our partners with the confidence they need to advance their drug development pipelines.
We invite you to explore how our capabilities can enhance your supply chain and reduce your manufacturing costs. By leveraging our expertise in nickel-catalyzed transformations, we can offer a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and discover how we can serve as your trusted partner for high-quality, cost-effective enamine intermediates. Let us help you navigate the complexities of modern chemical synthesis with solutions that are both scientifically superior and commercially viable.
