Advanced Synthesis of 3-Benzyl Coumarin-Imidazole Salts for Commercial Anticancer Applications
Introduction to Patent CN108299401B Technology
The pharmaceutical landscape is continuously evolving through the strategic design of hybrid molecules that combine distinct pharmacophores to enhance therapeutic efficacy. Patent CN108299401B introduces a novel series of 3-benzyl substituted coumarin-imidazole salt compounds, representing a significant advancement in anticancer agent development. This technology leverages the molecular hybridization strategy, merging the biologically active 3-benzyl coumarin skeleton with imidazole or benzimidazole units to create compounds with potent in vitro antitumor cytotoxic activity. The structural versatility of these compounds allows for extensive SAR (Structure-Activity Relationship) studies, targeting various cancer cell lines including leukemia, liver cancer, and breast cancer. As a reliable pharmaceutical intermediates supplier, understanding the underlying chemistry of such patents is crucial for developing robust supply chains for next-generation oncology drugs.
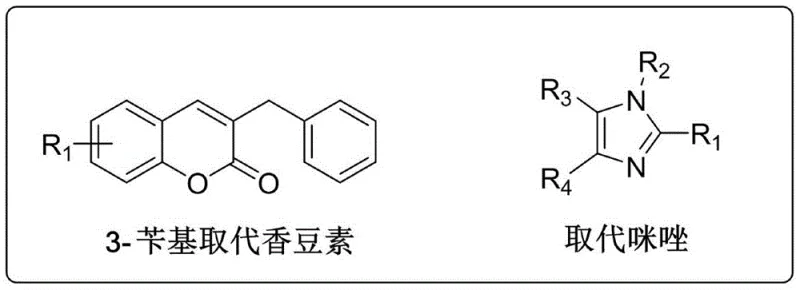
The core innovation lies in the efficient preparation method which ensures high purity and yield, critical factors for clinical translation. The patent details a systematic approach starting from readily available raw materials like salicylaldehyde and ethyl benzoylacetate. By optimizing reaction conditions such as solvent choice and catalyst loading, the synthesis achieves remarkable efficiency, with the initial condensation step reporting yields as high as 95%. This level of efficiency reduces waste and improves the overall economic viability of the process, addressing key concerns for procurement managers focused on cost reduction in pharmaceutical intermediates manufacturing. The resulting compounds, characterized by their specific substitution patterns on the imidazole ring, offer a diverse library for drug discovery programs aiming to overcome resistance mechanisms in tumor cells.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for coumarin derivatives often suffer from harsh reaction conditions, poor regioselectivity, and the use of toxic heavy metal catalysts that complicate downstream purification. Conventional alkylation methods to attach nitrogenous heterocycles frequently require strong bases or high temperatures, which can degrade the sensitive lactone ring of the coumarin structure, leading to significant impurity profiles. Furthermore, achieving selective reduction of ketone functionalities in the presence of conjugated double bonds typically poses a challenge, often resulting in mixtures of 1,2 and 1,4 reduction products that are difficult to separate. These inefficiencies translate to higher production costs, longer lead times, and increased environmental burden due to excessive solvent use and waste generation. For supply chain heads, these variables introduce unpredictability in delivery schedules and quality consistency, potentially jeopardizing clinical trial timelines.
The Novel Approach
The methodology described in CN108299401B overcomes these hurdles through a mild, step-wise protocol that prioritizes chemoselectivity and operational simplicity. By employing piperidine as an organocatalyst for the Knoevenagel condensation, the process avoids the need for corrosive acids or bases, proceeding smoothly at room temperature in acetonitrile. The subsequent use of Luche reduction conditions (CeCl3·7H2O/NaBH4) ensures exclusive 1,2-reduction of the carbonyl group, preserving the unsaturated lactone system essential for biological activity. This precision eliminates the need for complex chromatographic separations of reduction byproducts, streamlining the workflow. The final quaternization steps are conducted under reflux in common solvents like acetone or toluene, facilitating easy isolation of the product as a solid precipitate. This novel approach not only enhances the purity of the high-purity pharmaceutical intermediates but also significantly simplifies the manufacturing process, offering substantial cost savings and improved scalability.
Mechanistic Insights into Knoevenagel Condensation and Luche Reduction
The synthetic pathway begins with a base-catalyzed Knoevenagel condensation, where the active methylene group of ethyl benzoylacetate attacks the carbonyl carbon of salicylaldehyde. Piperidine acts as a nucleophilic catalyst, forming an iminium intermediate that enhances the electrophilicity of the aldehyde, facilitating the condensation followed by cyclization and ester exchange to form the 3-benzoyl coumarin core. This mechanism is highly efficient, driven by the thermodynamic stability of the aromatic coumarin system formed. Following this, the critical Luche reduction step utilizes the lanthanide salt cerium trichloride to coordinate with the ketone oxygen, increasing its electrophilicity towards the hydride source, sodium borohydride. This coordination effectively suppresses the 1,4-conjugate addition, directing the hydride attack exclusively to the carbonyl carbon to yield the allylic alcohol. This mechanistic control is vital for maintaining the structural integrity required for the subsequent substitution reactions.
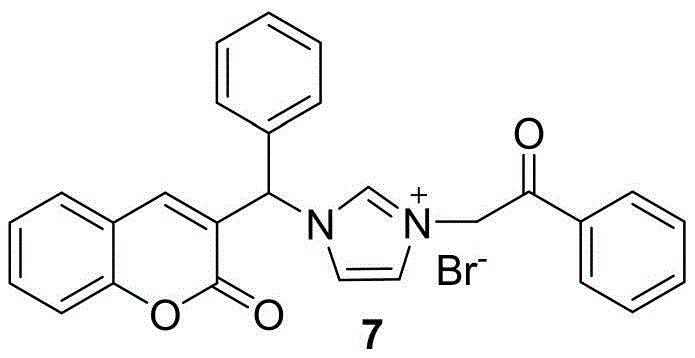
Following the reduction, the hydroxyl group is activated via mesylation using methanesulfonyl chloride and triethylamine. This converts the poor leaving group (-OH) into an excellent leaving group (-OMs), enabling a smooth nucleophilic substitution with imidazole or benzimidazole derivatives. The final quaternization involves the reaction of the tertiary nitrogen of the imidazole ring with various phenacyl bromides. This step forms the stable imidazolium salt, which often exhibits improved water solubility and bioavailability compared to the neutral precursors. The entire sequence is designed to minimize side reactions; for instance, the use of dichloromethane in the mesylation step prevents hydrolysis of the sensitive ester or lactone moieties. Understanding these mechanistic nuances allows R&D directors to optimize process parameters further, ensuring consistent quality and minimizing the formation of genotoxic impurities often associated with alkylating agents.
How to Synthesize 3-Benzyl Coumarin-Imidazole Salts Efficiently
The synthesis of these potent anticancer intermediates follows a logical four-step sequence that balances yield and purity. The process initiates with the condensation of salicylaldehyde and ethyl benzoylacetate, followed by the selective reduction of the resulting ketone. The third step involves activating the alcohol for nucleophilic attack, and the final step constructs the quaternary ammonium salt. Each stage has been optimized in the patent to use stoichiometric ratios that maximize conversion while minimizing excess reagent waste. For detailed operational parameters, safety data, and specific workup procedures required for GMP compliance, please refer to the standardized synthesis guide below.
- Perform Knoevenagel condensation of salicylaldehyde and ethyl benzoylacetate using piperidine in acetonitrile to yield 3-benzoyl coumarin.
- Execute selective Luche reduction of the ketone using CeCl3·7H2O and NaBH4 in methanol at 0°C to obtain 3-benzyl alcohol coumarin.
- Convert the alcohol to a mesylate intermediate using methanesulfonyl chloride and triethylamine in dichloromethane.
- React the mesylate with imidazole or benzimidazole derivatives followed by quaternization with haloalkanes to finalize the salt structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthesis route outlined in this patent offers distinct advantages that align with the goals of cost reduction and supply chain reliability. The reliance on commodity chemicals such as salicylaldehyde, ethyl benzoylacetate, and common solvents like methanol and acetonitrile ensures that raw material sourcing is stable and not subject to the volatility associated with exotic reagents. The high yields reported in the initial steps, particularly the 95% yield in the condensation phase, directly contribute to lower material costs per kilogram of final product. Additionally, the avoidance of transition metal catalysts like palladium or platinum eliminates the need for expensive metal scavenging processes and rigorous residual metal testing, which are significant cost drivers in API manufacturing. This streamlined process translates to a more competitive pricing structure for bulk purchasers.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts and harsh reaction conditions, significantly lowering the operational expenditure associated with energy consumption and specialized equipment maintenance. By achieving high conversion rates in the early stages, the amount of raw material required to produce a fixed quantity of the final intermediate is minimized, leading to substantial cost savings in material procurement. Furthermore, the simplified purification steps, primarily relying on precipitation and standard column chromatography rather than complex distillation or crystallization sequences, reduce labor hours and solvent consumption. These efficiencies collectively drive down the cost of goods sold (COGS), making the final anticancer agents more economically viable for development.
- Enhanced Supply Chain Reliability: The use of widely available starting materials mitigates the risk of supply disruptions that often plague specialized chemical supply chains. Since the synthesis does not depend on single-source reagents or geopolitically sensitive materials, procurement managers can establish multi-vendor sourcing strategies to ensure continuity. The robustness of the reaction conditions, which tolerate minor fluctuations in temperature and stoichiometry without significant yield loss, adds another layer of reliability to the manufacturing process. This stability ensures that production schedules can be met consistently, reducing lead time for high-purity pharmaceutical intermediates and preventing delays in downstream drug formulation and clinical trials.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, utilizing unit operations such as stirring, reflux, and filtration that are easily transferred from laboratory glassware to industrial reactors. The solvents employed are standard in the fine chemical industry and can be efficiently recovered and recycled, aligning with green chemistry principles and reducing the environmental footprint. The absence of heavy metal waste simplifies effluent treatment and regulatory compliance, avoiding the stringent disposal costs associated with toxic catalysts. This environmental compatibility facilitates smoother regulatory approvals and supports sustainable manufacturing practices, which are increasingly important for corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-benzyl coumarin-imidazole salts. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of this technology for potential partners and stakeholders.
Q: What is the key advantage of the Luche reduction step in this synthesis?
A: The Luche reduction utilizing cerium trichloride and sodium borohydride ensures high chemoselectivity for the 1,2-reduction of the ketone carbonyl group without affecting the conjugated double bond of the coumarin ring, preserving the core pharmacophore integrity.
Q: How does this molecular hybridization strategy improve biological activity?
A: By combining the 3-benzyl substituted coumarin scaffold with imidazole or benzimidazole moieties, the resulting hybrids exhibit synergistic effects, demonstrating superior in vitro cytotoxicity against multiple cancer cell lines compared to single-structure analogs.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes common organic solvents like acetonitrile, methanol, and dichloromethane, and avoids extreme temperatures or pressures, making it highly amenable to scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzyl Coumarin-Imidazole Salts Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the 3-benzyl coumarin-imidazole scaffold in modern oncology research. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to market supply. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for clinical grade intermediates. We understand the critical nature of impurity control in anticancer drug development and employ advanced analytical techniques to guarantee the quality and consistency of every batch produced.
We invite you to collaborate with us to leverage this patented technology for your drug discovery pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex organic synthesis can accelerate your path to commercialization while optimizing your overall project budget.
