Advanced Chiral Biphenyl Phase Transfer Catalysts for Scalable Pharmaceutical Manufacturing
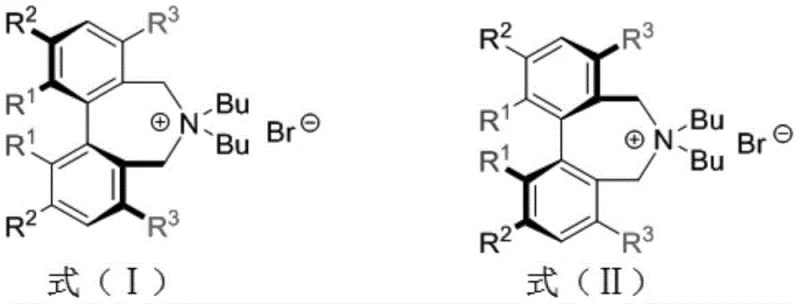
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical sector. Patent CN111574450A introduces a significant breakthrough in this domain by disclosing a novel series of chiral biphenyl quaternary ammonium salt phase transfer catalysts. These catalysts, structurally defined by Formula (I) and Formula (II), represent a refined evolution of the classic Maruoka catalyst family. The core innovation lies not just in their catalytic performance, which achieves yields as high as 97% and enantiomeric excess up to 95%, but critically in the methodology used to construct their chiral backbone. By shifting away from prohibitively expensive resolving agents to more accessible alternatives, this technology addresses a fundamental bottleneck in the commercial manufacturing of chiral fine chemicals. For R&D directors and procurement specialists alike, this patent signals a pathway toward more economically viable and scalable production of complex amino acid derivatives and peptide mimics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial application of modified Maruoka phase transfer catalysts has been hindered by the high cost and limited availability of the chiral resolving agents required to establish axial chirality in the biphenyl skeleton. Conventional protocols frequently utilize (R,R)-diphenylethylenediamine for the chemical resolution of racemic biphenol precursors. While effective in achieving high optical purity, this diamine is a high-value specialty chemical, making the overall cost of goods sold (COGS) for the resulting catalyst prohibitive for large-scale commodity chemical production. Furthermore, the reliance on such specific, expensive reagents introduces supply chain fragility; any disruption in the availability of the resolving agent can halt the entire catalyst production line. Additionally, traditional synthesis routes often involve harsh conditions or toxic transition metals that complicate waste management and regulatory compliance, creating significant hurdles for green chemistry initiatives in modern manufacturing facilities.
The Novel Approach
The methodology outlined in CN111574450A fundamentally restructures the economic model of catalyst production by employing (R)-methoxyphenylacetic acid as the resolving agent. This compound is not only significantly cheaper than traditional diamines but is also widely available in the bulk chemical market, ensuring a stable and reliable supply chain. The process involves a streamlined sequence where the biphenyl backbone is constructed, esterified with the chiral acid, and then separated via crystallization to achieve an enantiomeric excess greater than 99%. Following resolution, the chiral auxiliary is removed, leaving behind the optically pure biphenyl scaffold ready for functionalization. This approach eliminates the dependency on scarce chiral pool resources and simplifies the purification workflow. Consequently, manufacturers can achieve substantial cost reduction in pharmaceutical intermediate manufacturing while maintaining the rigorous stereochemical control necessary for drug synthesis, effectively bridging the gap between academic elegance and industrial practicality.
Mechanistic Insights into Asymmetric Phase Transfer Alkylation
The efficacy of these chiral biphenyl quaternary ammonium salts stems from their ability to create a highly organized chiral environment at the interface of immiscible phases. In the asymmetric alkylation of N-(diphenylmethylene)glycine tert-butyl ester, the catalyst functions by extracting the glycine enolate into the organic phase as a tight ion pair. The rigid biphenyl backbone, locked in a specific axial configuration, positions the quaternary ammonium cation and the bulky aryl substituents to shield one face of the planar enolate intermediate. As shown in the structural variations in Figure 2, substituents at the 3, 3', 5, and 5' positions play a pivotal role in defining the geometry of this chiral pocket. For instance, the presence of 3,5-bis(trifluoromethyl)phenyl groups at the 5, 5' positions enhances selectivity through both steric bulk and electronic modulation, creating a deep chiral cavity that forces the electrophile to approach from a single trajectory. This precise spatial arrangement minimizes the formation of unwanted enantiomers, ensuring that the resulting amino acid derivatives possess the high optical purity required for downstream API synthesis.
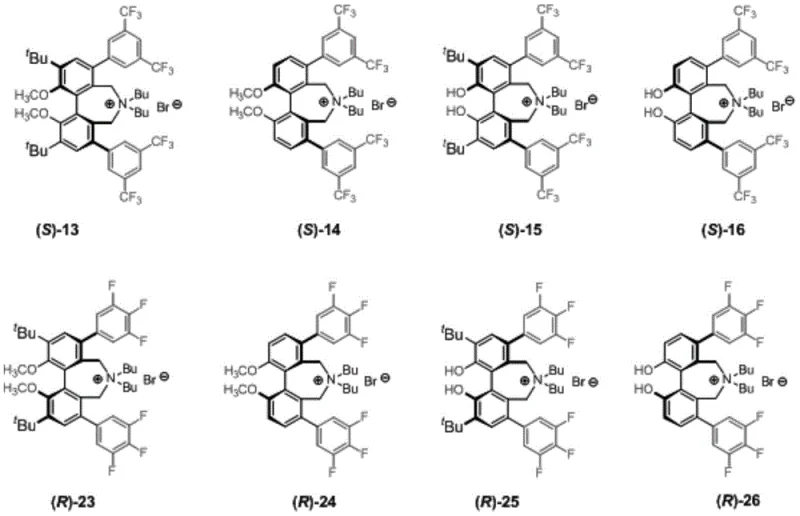
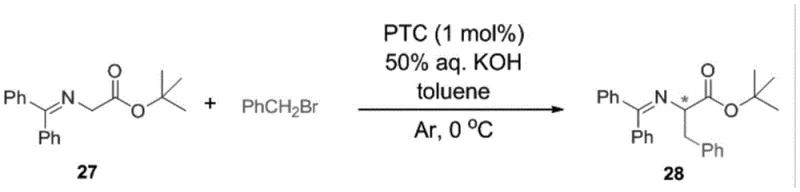
The reaction mechanism proceeds under mild biphasic conditions, typically utilizing 50% aqueous KOH and toluene at 0°C, as illustrated in the reaction scheme below. The phase transfer catalyst shuttles hydroxide ions into the organic phase to deprotonate the glycine Schiff base, generating the reactive nucleophile. The chiral cation then associates with this anion, and the subsequent attack on the alkyl halide (e.g., benzyl bromide) occurs within the protective chiral sphere of the catalyst. The data indicates that catalysts with methoxy groups at the 2, 2' positions generally exhibit slightly improved enantioselectivity compared to their hydroxy counterparts, likely due to increased conformational rigidity. Furthermore, the absence of transition metals in the catalytic cycle eliminates the risk of heavy metal contamination in the final product, a critical quality attribute for pharmaceutical intermediates. This metal-free nature simplifies the purification process, removing the need for expensive scavenging resins or complex chromatography steps often required to meet strict residual metal specifications.
How to Synthesize Chiral Biphenyl Quaternary Ammonium Catalysts Efficiently
The synthesis of these high-performance catalysts follows a logical, step-wise progression designed to maximize yield and optical purity at every stage. The process begins with the construction of the biphenyl core via oxidative coupling of brominated phenols, followed by the crucial resolution step using the cost-effective chiral acid. Subsequent functionalization involves methylation, bromomethylation of the methyl groups, and finally, quaternization with dibutylamine to install the active nitrogen center. Each step has been optimized to ensure robustness, with detailed protocols available for scaling from gram to kilogram quantities. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for process chemists aiming to implement this technology.
- Initiate the synthesis by brominating 5-methyl-2-tert-butylphenol followed by oxidative coupling to form the biphenyl backbone.
- Perform chiral resolution using cost-effective (R)-methoxyphenylacetic acid to establish axial chirality with high enantiomeric excess.
- Complete the catalyst assembly through methylation, bromomethylation, and final quaternization with dibutylamine to yield the active species.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst technology offers distinct strategic advantages that extend beyond mere technical performance. The primary value proposition is the drastic simplification of the raw material portfolio. By replacing expensive, niche resolving agents with commodity-grade chiral acids, the volatility of input costs is significantly dampened. This stability allows for more accurate long-term budgeting and reduces the financial risk associated with raw material price fluctuations. Moreover, the synthetic route avoids the use of toxic transition metal catalysts in the final application, which streamlines the environmental compliance process. Facilities can operate with reduced waste treatment burdens, as there is no need for specialized heavy metal disposal protocols, leading to a leaner and more sustainable operational footprint.
- Cost Reduction in Manufacturing: The economic impact of switching to (R)-methoxyphenylacetic acid as a resolving agent cannot be overstated. In traditional catalyst synthesis, the resolving agent often accounts for a disproportionate share of the total material cost. By utilizing a widely available and inexpensive alternative, the overall production cost of the catalyst is substantially lowered. This reduction cascades down to the cost of the final pharmaceutical intermediate, improving the margin profile for the entire value chain. Additionally, the high yields reported (up to 97%) mean less raw material is wasted per unit of product, further enhancing the cost-efficiency of the process without requiring capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of common chemical feedstocks. Unlike specialized chiral diamines that may have single-source suppliers or long lead times, the key reagents for this synthesis are produced by multiple global manufacturers. This diversification mitigates the risk of supply disruptions caused by geopolitical issues or production outages at a single vendor. Furthermore, the robustness of the reaction conditions—operating at ambient or near-ambient temperatures with standard solvents like toluene and acetonitrile—means that production can be easily transferred between different manufacturing sites without extensive re-validation, ensuring continuous supply even in the face of logistical challenges.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on heterogeneous phase transfer catalysis, a technology well-understood in large-scale chemical engineering. The use of aqueous bases and the absence of sensitive organometallic reagents make the reaction safer to run in large reactors, reducing the risk of thermal runaways. From an environmental perspective, the elimination of transition metals aligns with increasingly stringent global regulations regarding impurity profiles in drug substances. This 'green' attribute facilitates faster regulatory approval and reduces the complexity of the quality control workflow, allowing for quicker time-to-market for new drug candidates relying on these chiral building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral biphenyl catalyst technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this catalytic system into existing production lines.
Q: What is the primary advantage of this new catalyst synthesis method over traditional Maruoka catalysts?
A: The primary advantage lies in the resolution strategy. Traditional methods often rely on expensive resolving agents like (R,R)-diphenylethylenediamine. This patent utilizes (R)-methoxyphenylacetic acid, which is significantly cheaper and more readily available, drastically reducing the raw material cost burden for large-scale production without compromising optical purity.
Q: How does the catalyst structure influence enantioselectivity in asymmetric alkylation?
A: The steric environment created by substituents at the 3, 3', 5, and 5' positions is critical. The patent data indicates that introducing bulky groups like tert-butyl or electron-withdrawing groups like trifluoromethyl phenyl rings constructs a rigid chiral cavity. This cavity effectively differentiates between prochiral faces of the substrate, leading to high enantiomeric excess values up to 95%.
Q: Is this catalytic system suitable for industrial scale-up?
A: Yes, the process is designed for scalability. It employs mild reaction conditions, such as using 50% aqueous KOH and operating at 0°C, which are safer and easier to manage in large reactors compared to cryogenic organometallic methods. Furthermore, the use of robust phase transfer catalysis simplifies workup procedures, enhancing overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Biphenyl Quaternary Ammonium Salt Supplier
The technological advancements detailed in CN111574450A represent a significant opportunity for optimizing the synthesis of chiral pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high optical purity and yield demonstrated in the patent are maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of catalyst or intermediate meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this cost-effective and high-performance catalytic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our supply chain capabilities can support your long-term growth objectives in the competitive pharmaceutical market.
