Advanced Catalytic Synthesis of 3-Sulfonylated Indanones for Scalable Pharmaceutical Manufacturing
Advanced Catalytic Synthesis of 3-Sulfonylated Indanones for Scalable Pharmaceutical Manufacturing
The development of efficient synthetic routes for complex heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry and fine chemical manufacturing. A significant breakthrough in this domain is detailed in patent CN108689892B, which introduces a novel methodology for the construction of 3-sulfonylated indanone derivatives. These compounds are highly valued for their unique physical and biological activities, finding applications in drug discovery and agrochemical development. The disclosed technology leverages a copper-catalyzed radical cascade reaction that merges cyclization and sulfonylation into a single operational step. This approach not only streamlines the synthetic pathway but also addresses critical challenges related to regioselectivity and functional group tolerance. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols.
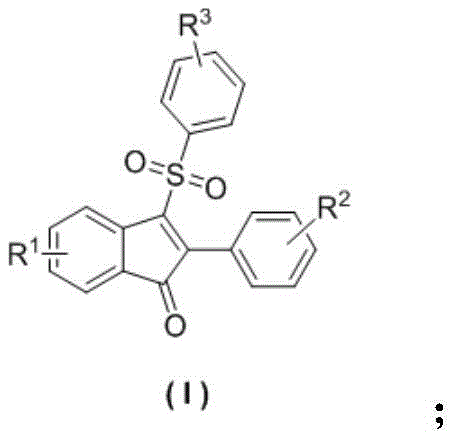
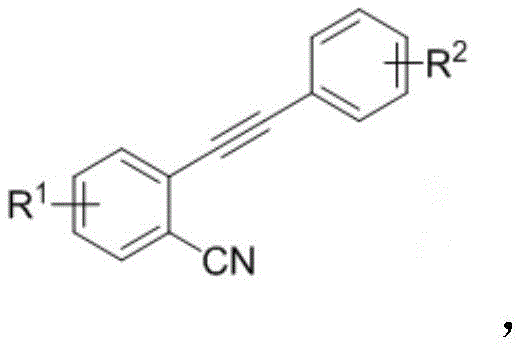
The core innovation lies in the ability to construct the indanone core while simultaneously installing a sulfonyl group at the 3-position. Traditionally, accessing such densely functionalized scaffolds would require multi-step sequences involving protection-deprotection strategies or the use of expensive transition metal catalysts. The method described in CN108689892B circumvents these limitations by utilizing inexpensive cuprous iodide (CuI) and tert-butyl hydroperoxide (TBHP) as the oxidant. This combination facilitates a radical mechanism that activates the alkyne and nitrile moieties of the starting material, leading to the formation of the five-membered ring with high efficiency. The versatility of this reaction is underscored by its compatibility with a wide range of substituents, including halogens, alkyl groups, and alkoxy groups, making it an ideal platform for generating diverse libraries of bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 3-sulfonylated indanone compounds was virtually unreported, highlighting a significant gap in the available chemical toolbox for medicinal chemists. Conventional approaches to indanone synthesis often rely on Friedel-Crafts acylation or aldol condensation reactions, which can suffer from poor regioselectivity and the generation of hazardous waste streams. Furthermore, introducing a sulfonyl group typically requires separate sulfonation steps using corrosive reagents like chlorosulfonic acid, posing severe safety risks and equipment corrosion issues in a plant setting. The lack of a direct, one-pot method meant that process chemists had to endure lengthy synthetic timelines, resulting in lower overall yields and increased production costs. These inefficiencies create bottlenecks in the supply chain, particularly when scaling up for clinical trials or commercial launch, where consistency and purity are paramount.
The Novel Approach
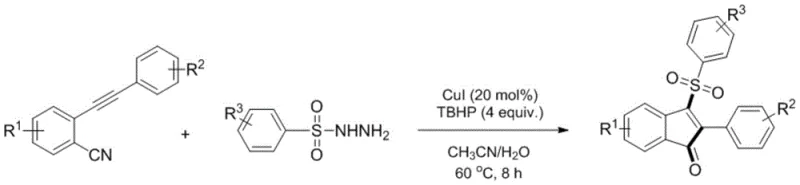
The novel approach presented in the patent data revolutionizes this landscape by employing a tandem radical cyclization strategy. By reacting o-aralkynyl benzonitriles with aryl sulfonyl hydrazides, the process achieves the simultaneous formation of the carbon-carbon and carbon-sulfur bonds necessary for the target structure. This telescoped operation eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption and processing time. The reaction conditions are remarkably mild, operating effectively between 45°C and 80°C, which significantly lowers energy consumption compared to high-temperature thermal cyclizations. Moreover, the use of a mixed solvent system comprising acetonitrile and water enhances the solubility of polar intermediates while maintaining an environmentally friendlier profile than pure organic solvents. This methodology offers a robust solution for cost reduction in pharmaceutical intermediate manufacturing by simplifying the workflow and improving the atom economy of the transformation.
Mechanistic Insights into CuI-Catalyzed Radical Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing process parameters and ensuring reproducibility at scale. The reaction is initiated by the interaction between the copper catalyst and tert-butyl hydroperoxide, generating tert-butoxyl radicals. These reactive species abstract a hydrogen atom from the sulfonyl hydrazide, producing a sulfonyl radical. This sulfonyl radical then adds selectively to the triple bond of the o-aralkynyl benzonitrile substrate. The resulting vinyl radical undergoes an intramolecular cyclization onto the nitrile group, forming an iminyl radical intermediate. Subsequent hydrolysis and oxidation steps lead to the final 3-sulfonylated indanone product. This radical cascade is highly efficient because each step is driven by the formation of stable bonds, minimizing side reactions such as polymerization or over-oxidation. The copper catalyst plays a dual role, facilitating the initial radical generation and potentially assisting in the final oxidation state adjustment, ensuring high turnover numbers.
From an impurity control perspective, the mechanism offers distinct advantages. The high regioselectivity of the radical addition ensures that the sulfonyl group is installed exclusively at the 3-position, preventing the formation of regioisomers that are difficult to separate. Additionally, the mild oxidative conditions prevent the degradation of sensitive functional groups often present in drug candidates, such as methoxy or halo substituents. The patent data indicates that the reaction tolerates electron-donating and electron-withdrawing groups equally well, suggesting a broad substrate scope. This predictability is vital for quality assurance teams, as it reduces the complexity of the impurity profile. By controlling the stoichiometry of the oxidant and the catalyst loading, manufacturers can further suppress the formation of over-oxidized byproducts, ensuring that the final API intermediate meets stringent purity specifications required by regulatory bodies.
How to Synthesize 3-Sulfonylated Indanone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction monitoring. The process begins with the preparation of the o-aralkynyl benzonitrile precursor, which can be efficiently synthesized via Sonogashira coupling, as illustrated in the patent examples. Once the starting materials are secured, the reaction is conducted in a standard vessel equipped with a heating mantle and stirrer. The protocol involves dissolving the substrates in the aqueous-organic solvent mixture, followed by the sequential addition of the catalyst and oxidant. Maintaining the temperature within the specified range is critical to balance reaction rate and selectivity. Upon completion, the workup procedure is straightforward, involving standard extraction and purification techniques that are easily adaptable to large-scale operations. Detailed standardized synthesis steps are provided in the guide below to ensure consistent results.
- Dissolve o-aralkynyl benzonitrile and aryl sulfonyl hydrazide in a mixed solvent of acetonitrile and water (3: 1 ratio).
- Add cuprous iodide (20 mol%) and tert-butyl hydroperoxide (4 equivalents) to the reaction mixture.
- Heat the mixture at 45-80°C for 6-12 hours, then perform extraction, drying, and column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the significant simplification of the supply chain for complex heterocyclic intermediates. By consolidating multiple synthetic steps into a single pot, the process reduces the number of unit operations, which directly correlates to lower capital expenditure and reduced operational overhead. The reliance on earth-abundant copper catalysts instead of precious metals like palladium or rhodium mitigates the risk associated with volatile metal prices and supply shortages. Furthermore, the use of readily available commercial feedstocks ensures that production schedules are not disrupted by raw material scarcity. This stability is essential for maintaining continuous manufacturing lines and meeting tight delivery deadlines for downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the elimination of expensive noble metal catalysts and the reduction in solvent usage. Traditional cross-coupling reactions often require costly ligands and rigorous purification to remove trace metals, adding substantial cost to the final product. In contrast, the CuI/TBHP system uses inexpensive reagents that are easy to handle and dispose of. The high yields reported in the patent examples, ranging consistently above 65%, mean that less raw material is wasted, improving the overall material throughput. Additionally, the simplified workup procedure reduces the labor hours and utility costs associated with distillation and chromatography, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain resilience. Because the reaction tolerates a wide variety of functional groups and operates under mild conditions, it is less prone to batch-to-batch variability caused by minor fluctuations in temperature or reagent quality. This reliability allows for more accurate forecasting and inventory management. The starting materials, o-aralkynyl benzonitriles and sulfonyl hydrazides, are commodity chemicals with established global supply networks, reducing the dependency on single-source vendors. This diversification of the supply base minimizes the risk of disruption, ensuring that the production of high-purity pharmaceutical intermediates can proceed uninterrupted even during market volatility.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is facilitated by the homogeneous nature of the reaction and the absence of hazardous gaseous reagents. The use of water as a co-solvent aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. This is increasingly important for meeting strict environmental regulations and corporate sustainability goals. The waste stream generated is primarily aqueous and organic solvent, which can be treated using standard effluent treatment protocols, avoiding the need for specialized hazardous waste disposal. The ease of scale-up ensures that the transition from R&D to commercial production is smooth, allowing companies to respond quickly to market demand without extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios. The answers cover aspects ranging from catalyst recovery to substrate scope, ensuring a comprehensive overview of the technology's practical application in an industrial setting.
Q: What are the key advantages of the CuI/TBHP catalytic system for indanone synthesis?
A: The system utilizes earth-abundant copper instead of precious metals, operates under mild temperatures (45-80°C), and achieves high regioselectivity without requiring harsh acidic or basic conditions.
Q: Are the raw materials for this synthesis readily available for industrial scale-up?
A: Yes, the process uses commercially available o-aralkynyl benzonitriles and aryl sulfonyl hydrazides, which are stable and easy to source, ensuring supply chain continuity for large-scale production.
Q: What is the typical yield range for this novel cyclization method?
A: According to the patent data, the method consistently delivers yields ranging from 65% to 78% across various substituted derivatives, demonstrating robust performance for diverse substrate scopes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Sulfonylated Indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN108689892B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our facilities are equipped to handle complex catalytic processes with precision, ensuring that every batch meets the rigorous quality standards demanded by the global pharmaceutical industry. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our deep knowledge of process optimization to maximize yield and purity. Our stringent purity specifications and rigorous QC labs guarantee that the 3-sulfonylated indanone derivatives we supply are free from critical impurities, ready for immediate use in drug substance manufacturing.
We invite you to collaborate with us to explore how this advanced chemistry can enhance your product pipeline. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to a secure supply chain and technical support that extends beyond simple transaction. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your development timelines and achieve your commercial goals with confidence and efficiency.
