Scalable Synthesis of Acalabrutinib Key Intermediate via Nickel-Catalyzed Oxidation for Commercial Production
Scalable Synthesis of Acalabrutinib Key Intermediate via Nickel-Catalyzed Oxidation for Commercial Production
The rapid advancement of targeted cancer therapies has placed immense pressure on the supply chain for high-quality pharmaceutical intermediates, particularly for Bruton's Tyrosine Kinase (BTK) inhibitors like Acalabrutinib. As the demand for this potent oral inhibitor grows, the industry requires robust, scalable, and cost-effective synthetic routes for its key building blocks. Patent CN109053780B introduces a groundbreaking preparation method for N-(pyridin-2-yl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzamide, a critical precursor in the Acalabrutinib value chain. This technology represents a significant departure from legacy methodologies by replacing hazardous cryogenic reagents and expensive noble metals with a benign, nickel-catalyzed oxidation system. For global procurement teams and R&D directors, this innovation offers a pathway to secure supply continuity while drastically reducing the environmental footprint and manufacturing costs associated with complex oncology drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key boronate intermediate has relied on pathways fraught with operational hazards and economic inefficiencies. One prominent prior art method, disclosed in patent WO2013116382A1, utilizes p-bromobenzoic acid which is converted to an acid chloride and subsequently coupled with 2-aminopyridine. The critical bottleneck in this route is the final lithiation step using n-butyllithium to install the boron moiety. This reagent is pyrophoric, requiring stringent safety protocols and cryogenic conditions (often below -70°C) to prevent thermal runaway and decomposition. Furthermore, the handling of large quantities of thionyl chloride generates significant corrosive waste streams, complicating regulatory compliance and waste disposal logistics.
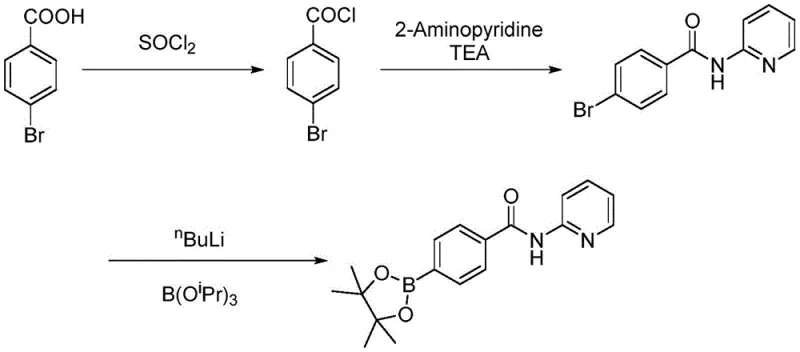
Another conventional approach described in US20140155406A1 attempts to bypass the lithiation step by starting directly with expensive 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoic acid. While this avoids n-butyllithium, it shifts the cost burden to the raw material stage, as the boronated starting material is significantly more expensive than simple halo-aromatics. Additionally, this route still necessitates the use of thionyl chloride for acyl chlorination, maintaining the high pollution profile. A third alternative involving palladium-catalyzed borylation (WO2013010868A1) introduces another layer of cost through the use of precious metal catalysts, which are not only expensive to purchase but also require complex removal steps to meet stringent residual metal limits in pharmaceutical products.
The Novel Approach
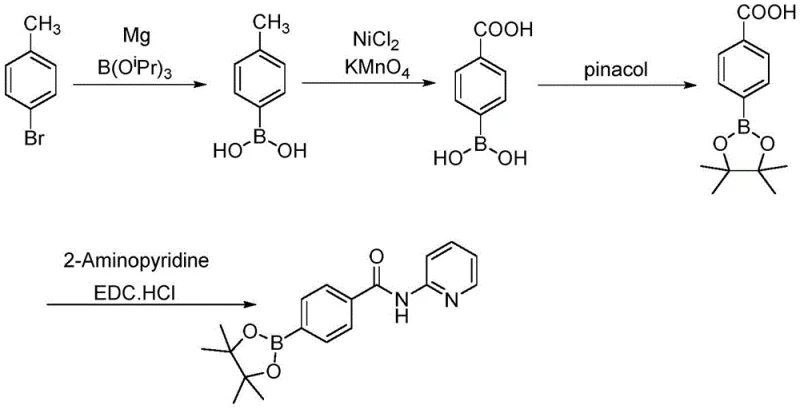
In stark contrast to these legacy methods, the novel process detailed in CN109053780B constructs the molecule from the ground up using p-bromotoluene, a commodity chemical with excellent availability and pricing stability. The core innovation lies in the strategic sequence: a Grignard reaction to install the boron functionality early, followed by a highly efficient nickel-catalyzed oxidation of the methyl group to a carboxylic acid. This sequence completely eliminates the need for cryogenic lithiation and precious metal catalysts. By utilizing potassium permanganate as the oxidant in the presence of a cheap nickel catalyst and a phase-transfer agent, the process achieves high conversion rates under mild conditions. This approach not only simplifies the operational workflow but also fundamentally alters the cost structure of the intermediate, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships focused on long-term cost reduction in API manufacturing.
Mechanistic Insights into Nickel-Catalyzed Benzylic Oxidation
The heart of this technological breakthrough is the nickel-catalyzed oxidation step, which transforms p-tolylboronic acid into p-carboxyphenylboronic acid. Mechanistically, this reaction leverages the ability of nickel species to facilitate electron transfer between the permanganate oxidant and the benzylic substrate. The addition of tetrabutylammonium bromide (TBAB) serves as a crucial phase-transfer catalyst, enhancing the solubility of the inorganic oxidant in the organic-aqueous interface where the reaction occurs. This synergistic catalytic system allows the oxidation to proceed efficiently at temperatures ranging from 20°C to 50°C, avoiding the thermal stress that often degrades sensitive boronic acid functionalities. The mildness of this oxidation is paramount, as harsher conditions could lead to protodeboronation or over-oxidation, generating difficult-to-remove impurities that would compromise the quality of the final API.
From an impurity control perspective, this mechanism offers superior selectivity compared to non-catalyzed permanganate oxidations. The nickel catalyst directs the oxidation specifically to the benzylic position, minimizing side reactions on the aromatic ring or the boronate ester group (once formed). Furthermore, the subsequent amidation step utilizes EDC hydrochloride and DMAP, a standard carbodiimide coupling protocol known for its high fidelity and low racemization risk (though not applicable here as the molecule is achiral, the principle of clean coupling holds). The combination of these selective steps ensures that the crude product profile is clean, often allowing the material to be used in downstream Suzuki couplings without rigorous chromatographic purification, thereby maximizing overall yield and throughput for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize N-(pyridin-2-yl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzamide Efficiently
The synthesis protocol outlined in the patent provides a clear, four-step roadmap that balances chemical efficiency with operational safety. The process begins with the formation of the Grignard reagent from p-bromotoluene, followed by quenching with triisopropyl borate to yield the boronic acid. This is followed by the pivotal nickel-catalyzed oxidation, esterification with pinacol, and final amide bond formation. Each step has been optimized to minimize solvent usage and maximize recovery, ensuring that the process is not just chemically sound but also industrially viable. The detailed standardized synthesis steps below provide the specific stoichiometric ratios and temperature controls necessary to replicate the high yields reported in the patent data.
- Perform Grignard reaction on p-bromotoluene with magnesium and triisopropyl borate to generate p-tolylboronic acid.
- Oxidize p-tolylboronic acid to p-carboxyphenylboronic acid using potassium permanganate catalyzed by nickel chloride and tetrabutylammonium bromide.
- React p-carboxyphenylboronic acid with pinacol in n-hexane to form the pinacol boronate ester intermediate.
- Couple the boronate ester acid with 2-aminopyridine using EDC hydrochloride and DMAP to yield the final target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible business value beyond mere chemical yield. The primary advantage is the drastic simplification of the raw material portfolio. By shifting from specialized, high-cost boronated starting materials to commodity p-bromotoluene, the supply chain becomes more resilient to market fluctuations. The elimination of n-butyllithium removes a major safety liability and the associated costs of specialized storage and handling equipment. Moreover, the replacement of palladium catalysts with nickel chloride represents a massive reduction in direct material costs, as nickel is orders of magnitude cheaper than palladium and does not require expensive recovery infrastructure to meet regulatory limits on heavy metals in drug substances.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive reagents with commodity chemicals. The use of p-bromotoluene instead of pre-functionalized boronic acids significantly lowers the entry cost of goods sold (COGS). Additionally, the nickel-catalyzed oxidation replaces costly palladium systems, and the avoidance of cryogenic conditions reduces energy consumption for cooling. The simplified workup procedures, which often allow for direct use of intermediates without purification, further reduce solvent consumption and labor hours, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of widely available starting materials. p-Bromotoluene, magnesium, and potassium permanganate are bulk chemicals produced globally, reducing the risk of single-source bottlenecks that often plague specialized fine chemical supply chains. The robustness of the reaction conditions—operating at ambient to mild temperatures rather than extreme cryogenic lows—means that production is less susceptible to equipment failures or utility interruptions. This stability ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, securing the production timeline for the final API.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally favorable. The process avoids the generation of sulfur dioxide gas (from thionyl chloride) and minimizes the use of hazardous pyrophoric reagents. The aqueous workup in the oxidation step is straightforward, and the overall E-factor (mass of waste per mass of product) is improved due to higher yields and fewer purification steps. This green chemistry profile facilitates easier regulatory approval and permits for large-scale manufacturing facilities, enabling seamless commercial scale-up from pilot plants to multi-ton annual production capacities without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing framework. Understanding these nuances is critical for R&D teams assessing process transfer feasibility and for procurement teams negotiating long-term supply agreements based on this superior methodology.
Q: Why is the nickel-catalyzed oxidation method superior to traditional palladium routes for this intermediate?
A: The nickel-catalyzed method utilizes significantly cheaper catalysts compared to palladium, eliminates the need for complex catalyst recycling processes, and avoids the use of hazardous reagents like n-butyllithium, resulting in a safer and more cost-effective industrial process.
Q: What are the purity advantages of this specific synthetic route?
A: This route avoids high-pollution solvents and harsh cryogenic conditions, leading to simpler post-treatment procedures. The high selectivity of the nickel-catalyzed oxidation ensures minimal byproduct formation, allowing the product to often proceed to the next reaction step without extensive purification.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It uses readily available raw materials like p-bromotoluene, operates at mild temperatures (20-70°C), and employs robust phase-transfer catalysis, making it highly adaptable for production scales ranging from hundreds of kilograms to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(pyridin-2-yl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this nickel-catalyzed process are fully realized at an industrial level. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including precise temperature control for the Grignard step and robust filtration systems for the oxidation workup. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for oncology drug development.
We invite you to collaborate with us to leverage this advanced technology for your Acalabrutinib supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your COGS. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
