Advanced Synthesis of Palbociclib Parent Nucleus: A Cost-Effective Route for Commercial Scale-up
The pharmaceutical landscape for oncology treatments continues to evolve, with CDK4/6 inhibitors like Palbociclib playing a pivotal role in managing advanced breast cancer. Patent CN111362939B introduces a groundbreaking preparation method for the Palbociclib parent nucleus structure compound, addressing critical bottlenecks in existing manufacturing technologies. This innovation shifts the synthetic paradigm from expensive, hard-to-source trisubstituted pyrimidines to readily available Cytosine, offering a robust pathway for high-purity pharmaceutical intermediates. By re-engineering the synthetic route, this technology not only enhances chemical selectivity but also aligns with modern green chemistry principles, ensuring a sustainable supply chain for global drug manufacturers.
![Chemical structure of Palbociclib showing the core pyrido[2,3-d]pyrimidin-7-one scaffold](/insights/img/palbociclib-parent-nucleus-synthesis-pharma-supplier-20260303005258-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Palbociclib core has relied heavily on routes described in patents such as WO2014128588, which utilize 5-bromo-2,4-dichloropyrimidine as the primary starting material. This conventional approach presents significant challenges for commercial scale-up of complex pharmaceutical intermediates. The presence of multiple halogen atoms with similar reactivity on the pyrimidine ring leads to poor regioselectivity during nucleophilic substitution. Consequently, this results in a myriad of side reactions and isomeric impurities that are notoriously difficult to separate. The cumulative effect is a compromised overall yield, often hovering around 50%, and a rigorous purification burden that escalates production costs and extends lead times.
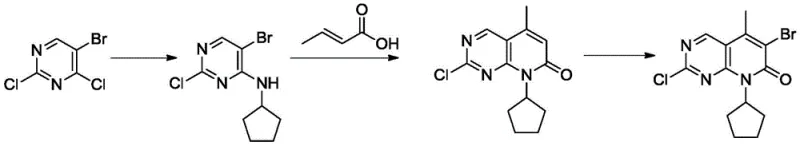
The Novel Approach
In stark contrast, the methodology disclosed in CN111362939B leverages Cytosine as a foundational building block, fundamentally altering the economic and technical feasibility of the process. This novel approach circumvents the selectivity issues inherent in trisubstituted pyrimidines by constructing the substitution pattern stepwise. By initiating the synthesis with Cytosine, manufacturers can achieve precise control over halogenation and alkylation positions. This strategic shift not only simplifies the reaction profile but also drastically reduces the formation of by-products. The result is a streamlined process that delivers the target parent nucleus with exceptional purity (>99.3%) and a significantly improved total yield exceeding 65%, representing a major leap forward in cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Stepwise Functionalization and Cyclization
The core of this technological advancement lies in its meticulously orchestrated five-step sequence, which transforms simple precursors into the complex pyrido[2,3-d]pyrimidin-7-one scaffold. The process begins with the chlorination of Cytosine to form 2-chloro-4-aminopyrimidine, followed by a highly selective bromination to yield 5-bromo-2-chloro-4-aminopyrimidine. Crucially, the subsequent N-alkylation with halogenated cyclopentane proceeds with high efficiency due to the activated nature of the intermediate. The mechanistic highlight is the palladium-catalyzed Heck reaction coupled with intramolecular amidation cyclization using crotonic acid. This tandem transformation efficiently constructs the fused ring system, establishing the critical carbon-carbon and carbon-nitrogen bonds required for the biological activity of the final API.
Impurity control is inherently built into this mechanism through the choice of reagents and reaction conditions. For instance, the use of specific brominating agents like liquid bromine in the presence of KBr catalyst ensures that bromination occurs exclusively at the desired position, avoiding poly-brominated side products. Furthermore, the final bromination step utilizes optimized catalyst systems (such as glacial acetic acid with KBr or oxalic acid) to ensure complete conversion without degrading the sensitive fused ring structure. This precision minimizes the generation of structurally related impurities, thereby reducing the complexity of downstream purification and ensuring that the final intermediate meets the stringent quality standards required for oncology drug production.
How to Synthesize Palbociclib Parent Nucleus Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize the benefits of the novel route. The process is designed to be operationally simple, utilizing common solvents like toluene, glacial acetic acid, and THF, which facilitates easy solvent recovery and waste management. The detailed standardized synthetic steps involve precise temperature controls, such as maintaining reflux conditions for chlorination and specific low-temperature crystallization protocols to isolate high-purity intermediates. For a comprehensive guide on executing this synthesis with optimal yield and safety, please refer to the technical protocol below.
- Chlorinate Cytosine using phosphorus pentachloride in toluene to obtain 2-chloro-4-aminopyrimidine (Intermediate 1).
- Brominate Intermediate 1 with bromine and KBr catalyst in glacial acetic acid to yield 5-bromo-2-chloro-4-aminopyrimidine (Intermediate 2).
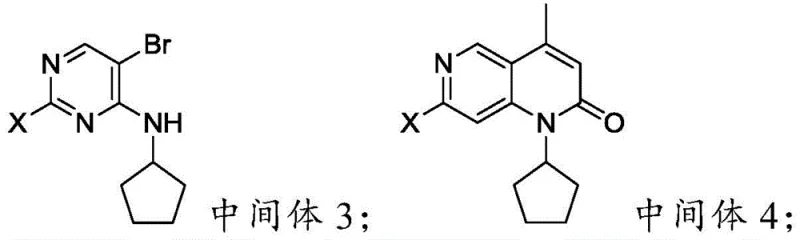
- Perform N-alkylation of Intermediate 2 with halogenated cyclopentane using triethylamine to form Intermediate 3.
- Execute a palladium-catalyzed Heck reaction and intramolecular amidation cyclization with crotonic acid to generate Intermediate 4.
- Conduct final bromination of Intermediate 4 using a brominating agent in a catalyst/solvent system to obtain the target Formula (I) compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this Cytosine-based route offers tangible strategic benefits beyond mere technical superiority. The primary advantage stems from the raw material profile; Cytosine is a commodity chemical with a stable global supply, unlike the specialized and often supply-constrained 5-bromo-2,4-dichloropyrimidine. This shift mitigates supply chain risks and provides a buffer against market volatility in raw material pricing. Additionally, the simplified purification process reduces the consumption of chromatography media and recrystallization solvents, directly lowering the variable costs associated with production.
- Cost Reduction in Manufacturing: The elimination of expensive, low-selectivity starting materials drives substantial cost savings. By avoiding the need for complex separation techniques to remove isomeric impurities, the process reduces both material loss and energy consumption. The higher overall yield means less raw material is required per kilogram of final product, optimizing the cost of goods sold (COGS) and improving margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Relying on widely available feedstocks like Cytosine and crotonic acid ensures a more resilient supply chain. This reduces the dependency on niche suppliers who may have limited capacity or long lead times. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by batch failures or quality deviations, ensuring consistent delivery of reliable API intermediate supplier commitments.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations such as reflux, filtration, and crystallization that are easily adapted from pilot to commercial scale. Furthermore, the reduction in side reactions leads to a cleaner waste stream, simplifying effluent treatment and helping facilities meet increasingly strict environmental regulations. This alignment with green chemistry principles supports long-term sustainability goals and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational feasibility for industrial partners considering this technology.
Q: What are the advantages of using Cytosine over 5-bromo-2,4-dichloropyrimidine?
A: Using Cytosine as a starting material significantly reduces raw material costs and improves regioselectivity. Unlike 5-bromo-2,4-dichloropyrimidine, which suffers from poor selectivity due to similar halogen activities leading to side reactions, the Cytosine route allows for controlled stepwise functionalization, resulting in higher overall yields (>65%) and purity (>99.3%).
Q: How does this process improve impurity control?
A: The novel route minimizes side reactions by avoiding the use of trisubstituted pyrimidine raw materials where halogen displacement is difficult to control. Each step, particularly the bromination and cyclization, is optimized with specific catalysts (e.g., KBr, PdCl2) and solvents to ensure high conversion rates, simplifying downstream purification and reducing the burden on QC labs.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the process is designed for green industrial production. It utilizes widely available and low-cost starting materials like Cytosine and crotonic acid. The reaction conditions are moderate (e.g., reflux in toluene or glacial acetic acid), and the workup procedures involve standard crystallization and filtration, making it highly adaptable for multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Palbociclib Parent Nucleus Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical methods to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can enhance your production efficiency and secure your supply of this vital pharmaceutical intermediate.
