Advanced Synthesis of Chiral Tetrahydroquinoline: High Purity and Scalable Manufacturing Solutions
Introduction to Breakthrough Chiral Synthesis Technology
The pharmaceutical industry continuously demands more efficient and stereoselective pathways for constructing complex heterocyclic scaffolds, particularly tetrahydroquinolines, which serve as critical cores in numerous bioactive molecules. Patent CN110066244B introduces a transformative method for synthesizing chiral tetrahydroquinoline using saturated aldehydes, addressing long-standing challenges in stereocontrol and yield optimization. This technology leverages a sophisticated dual-catalytic system involving diaryl prolinol silicon ether and a transition metal catalyst under an oxygen atmosphere, achieving unprecedented enantiomeric excess (ee) values greater than 99% and yields surpassing 80%. For R&D directors and procurement strategists, this represents a pivotal shift towards more sustainable and high-value manufacturing processes that eliminate the need for hazardous reagents while delivering superior product quality.
The significance of this innovation extends beyond mere academic interest; it provides a robust solution for the commercial production of high-purity pharmaceutical intermediates. By effectively controlling the stereoselectivity of the cyclization process through the precise matching of organocatalysts and metal catalysts, the method ensures consistent batch-to-batch reproducibility. This level of control is essential for meeting the stringent regulatory requirements of global health authorities, thereby reducing the risk of costly delays in drug approval pipelines. Furthermore, the use of readily available saturated aldehydes as starting materials simplifies the supply chain, offering a distinct competitive advantage in terms of raw material sourcing and cost management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydroquinoline derivatives has relied on methods that suffer from significant drawbacks regarding safety, environmental impact, and stereochemical control. For instance, prior art such as CN102993094A utilizes Lewis acids like SnCl2 combined with oxidants, which, while effective for non-chiral derivatives, generates corrosive byproducts like SnCl4 and fails to induce chirality, rendering it unsuitable for modern chiral drug synthesis. Other approaches, such as those disclosed in CN101429160A, involve the use of azide reagents which pose severe safety hazards due to their explosive potential, complicating industrial scale-up and increasing insurance and handling costs. Additionally, hydrogenation methods described in patents like CN106432072A often require high-pressure equipment and specialized catalysts, yet still struggle to produce chiral outcomes, limiting their utility in the synthesis of enantiomerically pure active pharmaceutical ingredients (APIs).
The Novel Approach
In stark contrast, the method disclosed in CN110066244B employs a mild, oxidative cyclization strategy that elegantly bypasses these limitations. By reacting a compound of Formula I in the presence of diaryl prolinol silicon ether and a catalyst like triphenylphosphine rhodium chloride under an oxygen atmosphere, the process achieves a highly efficient ring closure. This approach not only avoids the use of toxic tin reagents and dangerous azides but also operates at moderate temperatures ranging from 20°C to 50°C, significantly reducing energy consumption. The synergy between the organocatalyst and the metal catalyst allows for precise spatial arrangement during the bond-forming steps, resulting in products with exceptional optical purity.
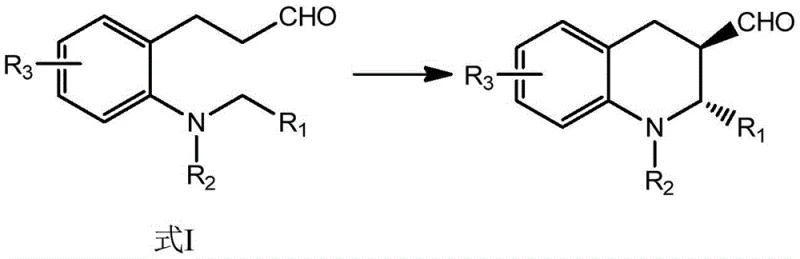
As illustrated in the reaction scheme above, the transformation proceeds smoothly to yield the chiral tetrahydroquinoline core with defined stereochemistry at the 2-position. The versatility of this method is further demonstrated by its tolerance to various substituents (R1, R2, R3), allowing for the synthesis of a diverse library of derivatives without compromising yield or selectivity. This flexibility is crucial for medicinal chemists exploring structure-activity relationships (SAR) during the drug discovery phase, as it enables rapid access to analogues with high fidelity.
Mechanistic Insights into Rhodium-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the intricate interplay between the diaryl prolinol silicon ether and the rhodium catalyst, which together orchestrate a highly stereoselective cyclization. The diaryl prolinol silicon ether acts as a chiral auxiliary, forming a transient enamine intermediate with the aldehyde moiety of the substrate. This enamine species is then activated by the rhodium catalyst, likely through coordination to the nitrogen or the pi-system, facilitating the intramolecular attack on the aromatic ring or the subsequent oxidative step. The bulky silyl ether group on the prolinol backbone creates a steric environment that shields one face of the reactive intermediate, thereby directing the incoming nucleophile or electrophile to the opposite face and ensuring the formation of a single enantiomer with >99% ee.
Furthermore, the use of oxygen as the terminal oxidant is a key feature that enhances the green chemistry profile of this process. Unlike stoichiometric oxidants that generate substantial waste, molecular oxygen is atom-economical and produces water as the only byproduct. This mechanistic pathway also contributes to a cleaner impurity profile, as the mild oxidative conditions prevent the over-oxidation or degradation of sensitive functional groups often seen in harsher chemical environments. For quality control teams, this means simpler downstream processing and higher overall recovery rates, directly impacting the cost of goods sold (COGS). The ability to fine-tune the catalyst loading and solvent system, such as using a DMSO and 1,2-dichloroethane mixture, further optimizes the reaction kinetics, ensuring that the process remains robust even when scaled to multi-kilogram batches.
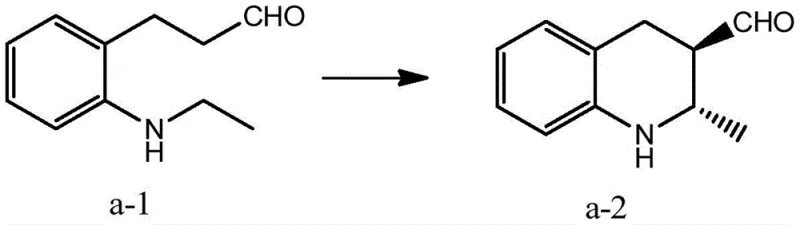
How to Synthesize Chiral Tetrahydroquinoline Efficiently
Implementing this synthesis route requires careful attention to solvent selection and molar ratios to replicate the high yields reported in the patent examples. The protocol involves dissolving the starting aldehyde, the chiral organocatalyst, the rhodium catalyst, and the base in a specific solvent mixture, followed by stirring under an oxygen balloon or flow. Detailed operational parameters, including exact temperatures and workup procedures, are critical for maintaining the integrity of the chiral center. For process chemists looking to adopt this technology, the following standardized steps outline the general procedure derived from the patent's most successful embodiments.
- Prepare the reaction mixture by combining the Formula I compound, diaryl prolinol silicon ether, triphenylphosphine rhodium chloride catalyst, and tri-n-butylamine in a mixed solvent of dimethyl sulfoxide and 1,2-dichloroethane.
- Maintain the reaction temperature between 20°C and 50°C under an oxygen atmosphere for a duration of 50 to 100 hours to ensure complete cyclization and stereoselectivity.
- Upon completion, filter the mixture, wash with water and saturated sodium bicarbonate, extract with dichloromethane, and purify via silica gel column chromatography to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound benefits for procurement managers and supply chain leaders focused on cost reduction and reliability. The elimination of hazardous reagents like azides and corrosive Lewis acids translates directly into lower safety compliance costs and reduced waste disposal fees. Moreover, the high yield and exceptional stereoselectivity minimize the need for expensive chiral resolution steps, which are traditionally a major bottleneck and cost driver in the production of chiral intermediates. By achieving >99% ee directly from the reaction, manufacturers can bypass complex recrystallization or chromatographic separation processes, leading to substantial savings in both time and materials.
- Cost Reduction in Manufacturing: The process utilizes relatively inexpensive and commercially available saturated aldehydes as starting materials, avoiding the need for specialized, high-cost precursors. Additionally, the catalyst system, while utilizing rhodium, operates efficiently at low loadings, and the recovery of the product is streamlined due to the clean reaction profile. This combination of factors results in a significantly lower cost per kilogram of the final API intermediate compared to legacy methods that require multiple protection-deprotection steps or harsh conditions.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as dimethyl sulfoxide, 1,2-dichloroethane, and simple amines ensures a stable and resilient supply chain. Unlike methods dependent on scarce natural products or complex custom-synthesized reagents, the inputs for this process are widely sourced from global chemical suppliers. This reduces the risk of supply disruptions and allows for better inventory planning, ensuring continuous production schedules even in volatile market conditions.
- Scalability and Environmental Compliance: The mild reaction conditions (20°C to 50°C) and the use of oxygen gas make this process inherently safer and easier to scale than high-pressure hydrogenation or cryogenic reactions. The reduced generation of heavy metal waste and toxic byproducts aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes for new manufacturing facilities. This scalability ensures that the technology can seamlessly transition from pilot plant trials to full commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is crucial for stakeholders evaluating the potential integration of this method into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over traditional Lewis acid approaches?
A: Unlike traditional methods using Lewis acids like SnCl2 which fail to produce chiral structures, this novel approach utilizes diaryl prolinol silicon ether to achieve extremely high stereoselectivity with ee values exceeding 99%, making it suitable for chiral drug development.
Q: What are the optimal reaction conditions for maximizing yield and optical purity?
A: The patent specifies that using a mixed solvent of dimethyl sulfoxide and 1,2-dichloroethane (volume ratio 1:1.2) at temperatures between 20°C and 50°C under an oxygen atmosphere yields the best results, with yields reaching over 83% and ee values up to 99%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (20-50°C) and atmospheric pressure (oxygen atmosphere), avoiding the high-pressure hydrogenation or hazardous azide reagents found in other methods, which significantly enhances its feasibility for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tetrahydroquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN110066244B can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch meets the exacting standards required by global pharmaceutical clients.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you accelerate your drug development timeline with reliable, high-purity chiral tetrahydroquinoline supplies.
