Advanced Electrochemical Synthesis of Trans-Olefins: A Green Alternative for Pharmaceutical Intermediates Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking sustainable alternatives to traditional transition-metal catalysis, particularly for the synthesis of valuable olefinic intermediates. Patent CN114645288A introduces a groundbreaking electrochemical protocol that fundamentally shifts the paradigm of trans-olefin construction. By leveraging electricity as a traceless reagent, this method successfully circumvents the longstanding reliance on expensive gold catalysts and complex ligand systems. The technology enables the direct coupling of halogenated alkanes with 1,3-butadiene under remarkably mild conditions, specifically at room temperature and in an inert atmosphere. This innovation not only streamlines the synthetic route but also addresses critical environmental concerns associated with heavy metal waste. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates.
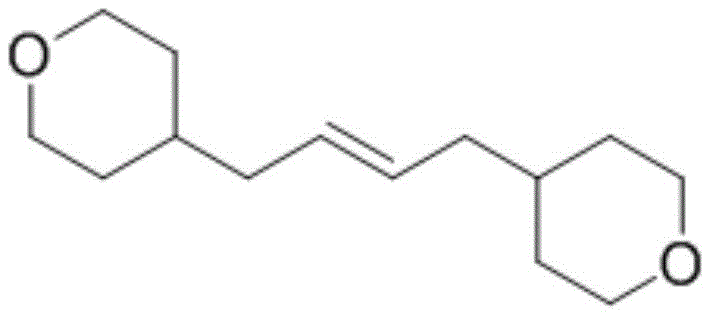
The resulting trans-olefin compounds, such as the bis(tetrahydropyran) derivative shown above, serve as versatile building blocks for complex organic synthesis. The ability to construct these carbon-carbon bonds with high stereoselectivity and purity is crucial for downstream applications in drug discovery and material science. Furthermore, the method demonstrates exceptional substrate tolerance, accommodating various halogenated precursors including iodides, bromides, and even chlorides. This flexibility ensures that manufacturers can adapt the process to available raw materials without compromising on the quality of the final product. As the industry moves towards greener manufacturing practices, such electrochemical solutions are becoming increasingly vital for maintaining competitiveness and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trans-olefins has relied heavily on classical cross-coupling reactions such as the Heck, Suzuki, or Wittig reactions. While these methods are well-established, they suffer from significant drawbacks that hinder efficient large-scale production. Primarily, they necessitate the use of precious metal catalysts like palladium or gold, which are not only costly but also pose severe challenges in terms of residual metal removal from the final active pharmaceutical ingredients (APIs). Additionally, these reactions often require stoichiometric amounts of phosphine ligands and strong bases, which generate substantial chemical waste and complicate the purification process. The need for rigorous purification to meet stringent pharmaceutical standards often leads to reduced overall yields and increased production timelines. Moreover, many traditional protocols require elevated temperatures or strictly anhydrous conditions, further escalating energy consumption and operational complexity. These cumulative factors create a bottleneck for cost reduction in fine chemical manufacturing, driving the urgent need for more sustainable alternatives.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent offers a streamlined and environmentally benign alternative that eliminates the need for external chemical oxidants or reductants. By utilizing a simple undivided cell equipped with inexpensive iron and nickel electrodes, the system generates reactive radical species directly at the electrode surface. This approach effectively replaces the function of expensive gold catalysts with electrical energy, thereby removing the burden of metal contamination entirely. The reaction proceeds smoothly at room temperature in common solvents like DMF, utilizing readily available electrolytes such as tetrabutylammonium iodide (nBu4NI). This simplicity translates to a drastic reduction in raw material costs and a significant simplification of the workup procedure. The absence of toxic ligands and heavy metals means that the downstream purification steps are far less intensive, allowing for faster turnaround times and higher throughput. This novel strategy exemplifies how modern electrosynthesis can overcome the inherent limitations of thermal catalysis.
Mechanistic Insights into Electrochemical Radical-Anion Coupling
The core of this innovation lies in its unique mechanistic pathway, which harnesses the power of cathodic reduction to drive the formation of carbon-carbon bonds. As illustrated in the mechanism diagram below, the process initiates at the cathode where the halogenated alkane undergoes a single-electron transfer to generate an alkyl radical species. This highly reactive intermediate immediately adds to the 1,3-butadiene molecule, forming a stabilized allylic radical. Subsequently, this allylic radical accepts a second electron at the cathode surface to transform into a nucleophilic allylic anion. This anion then attacks a second molecule of the halogenated alkane via an SN2-type mechanism, ultimately yielding the trans-olefin product. Simultaneously, the iron anode is oxidized to Fe3+, maintaining charge balance within the cell without introducing foreign catalytic species. This elegant cascade of radical and anionic intermediates ensures high selectivity for the trans-isomer, minimizing the formation of unwanted cis-byproducts or polymerization side reactions.
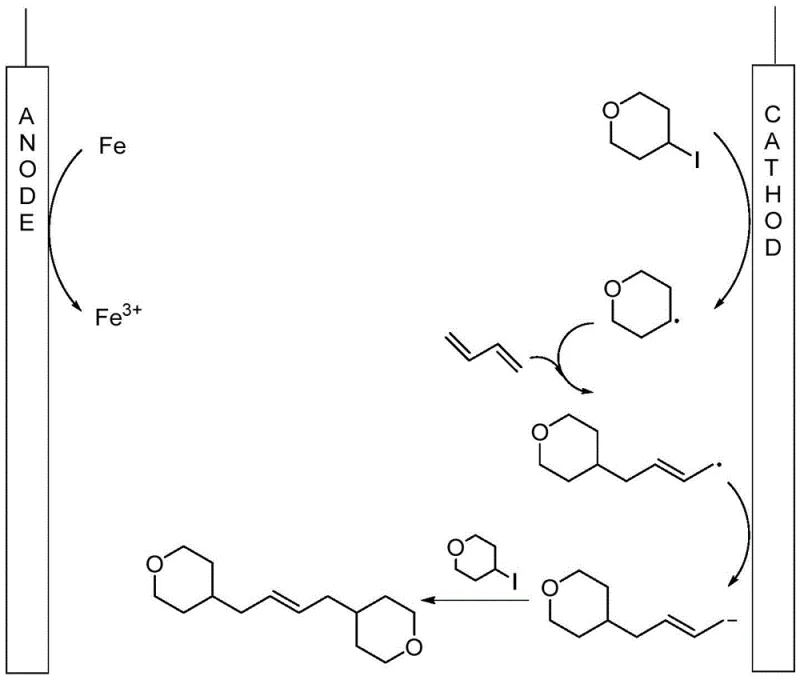
Understanding this mechanism is critical for optimizing reaction parameters such as current density and electrolyte concentration. The use of nBu4NI serves a dual purpose: it acts as the supporting electrolyte to ensure conductivity and provides iodide ions that may facilitate the initial electron transfer process. The choice of iron as the anode material is particularly strategic, as it is sacrificial and inexpensive, preventing the passivation issues often seen with inert anodes. For R&D teams, this mechanistic clarity allows for precise tuning of the reaction conditions to accommodate different substrate electronic properties. The ability to control the reaction purely through electrical parameters offers a level of precision that is difficult to achieve with thermal methods. This deep understanding of the electrocatalytic cycle underscores the robustness of the method and its potential for adaptation to a wide range of olefin synthesis challenges.
How to Synthesize Trans-Olefin Compounds Efficiently
Implementing this electrochemical protocol in a laboratory or pilot plant setting requires careful attention to cell configuration and reaction monitoring. The standard procedure involves dissolving the halogenated alkane substrate and the electrolyte in a polar aprotic solvent such as DMF, followed by the addition of a THF solution of 1,3-butadiene. The mixture is then subjected to constant current electrolysis using iron and nickel sheets as electrodes. Detailed standardized synthesis steps see the guide below.
- Mix halogenated alkane, 1,3-butadiene, and nBu4NI electrolyte in DMF solvent under inert atmosphere.
- Electrolyze the mixture using Iron (anode) and Nickel (cathode) electrodes at a constant current of 4mA for 8-14 hours.
- Wash the crude product with saline, extract with ethyl acetate, and purify via thin-layer chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology presents a compelling value proposition centered on cost efficiency and supply security. The elimination of precious metal catalysts removes a major source of price volatility and supply risk from the manufacturing equation. Furthermore, the simplified purification process reduces the consumption of solvents and silica gel, leading to substantial cost savings in waste disposal and material usage. The mild reaction conditions also imply lower energy requirements compared to high-temperature thermal processes, contributing to a smaller carbon footprint and reduced utility costs. These factors collectively enhance the economic viability of producing complex olefin intermediates on a commercial scale.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of gold catalysts and phosphine ligands, which are among the most expensive reagents in organic synthesis. By substituting these with electricity and cheap iron electrodes, the direct material cost of the reaction is drastically lowered. Additionally, the high purity of the crude product minimizes the need for extensive chromatographic purification, saving both time and expensive stationary phases. This leaner manufacturing process allows for tighter margins and more competitive pricing for the final intermediates without sacrificing quality standards.
- Enhanced Supply Chain Reliability: Relying on scarce precious metals often exposes manufacturers to geopolitical risks and supply disruptions. This electrochemical method utilizes commodity chemicals like halogenated alkanes and butadiene, which are produced in vast quantities globally, ensuring a stable and continuous supply of raw materials. The robustness of the reaction against moisture and oxygen variations further reduces the risk of batch failures due to environmental fluctuations. This reliability is crucial for maintaining consistent production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is increasingly feasible with modern flow cell technology, offering a clear path from gram-scale optimization to ton-scale production. The absence of heavy metal waste simplifies compliance with increasingly stringent environmental regulations regarding effluent discharge. This green profile not only reduces the cost of waste treatment but also enhances the corporate sustainability image of the manufacturer. The ability to operate at ambient pressure and temperature further lowers the capital expenditure required for specialized high-pressure reactors, making the technology accessible for rapid capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation. They are intended to provide clarity on the practical aspects of adopting this technology for industrial applications.
Q: Why is this electrochemical method superior to traditional gold-catalyzed synthesis?
A: Traditional methods rely on expensive gold catalysts and phosphine ligands which are difficult to remove and generate heavy metal waste. This electrochemical approach eliminates the need for external catalysts and ligands entirely, using electricity as the primary reagent, which drastically simplifies purification and reduces environmental impact.
Q: What yields and purity levels can be expected from this process?
A: According to patent CN114645288A, the method consistently achieves high purity levels of 99% across various substrates. Yields are robust, ranging from 44% to 77% depending on the specific halogenated alkane used, demonstrating the versatility and efficiency of the electrochemical protocol.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates at room temperature and atmospheric pressure using simple iron and nickel electrodes. These mild conditions, combined with the absence of hazardous catalysts, make the reaction inherently safer and easier to scale up compared to high-pressure or high-temperature traditional coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Olefin Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this can be seamlessly translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing our partners with a secure source of critical building blocks.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this catalyst-free approach can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments for your target trans-olefin compounds. Let us help you build a more sustainable and cost-effective supply chain for your next generation of therapeutic agents.
