Advanced Synthesis of Borazaphenanthrene Derivatives for High-Performance OLED Applications
Advanced Synthesis of Borazaphenanthrene Derivatives for High-Performance OLED Applications
The rapid evolution of the organic electronics sector demands continuous innovation in molecular design, particularly for materials that can enhance the efficiency and longevity of optoelectronic devices. Patent CN109232623B introduces a groundbreaking synthetic methodology for borazaphenanthrene and its derivatives, addressing critical challenges in the production of BN-doped polycyclic aromatic hydrocarbons. This technology leverages a strategic combination of Suzuki coupling, metal reduction, and Lewis base-guided electrophilic boron cyclization to construct complex heterocyclic frameworks with exceptional precision. The resulting compounds are not merely academic curiosities but represent a new class of functional materials with tunable photoelectric properties, specifically tailored for next-generation organic light-emitting diodes and photovoltaic applications. By utilizing commercially available disubstituted naphthalene as a starting point, this process significantly lowers the barrier to entry for manufacturing these high-value intermediates.
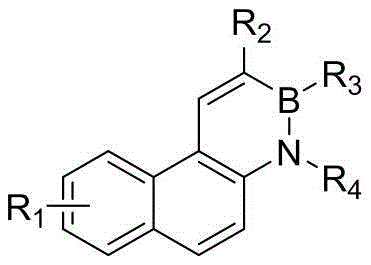
The core innovation lies in the versatility of the molecular scaffold, where substituents R1 through R4 can be independently modified to include alkyl, aryl, or heteroaryl groups, allowing for fine-tuning of electronic characteristics. This structural flexibility is paramount for R&D teams aiming to optimize charge transport and emission wavelengths in device architectures. Furthermore, the ability to introduce halogen atoms at specific positions opens avenues for further functionalization via cross-coupling reactions, thereby expanding the chemical space available for material scientists. As a reliable electronic chemical supplier, understanding the depth of such synthetic capabilities is crucial for securing a competitive edge in the supply of advanced OLED materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of boron-nitrogen doped polycyclic aromatic hydrocarbons has been plagued by severe limitations that hindered their widespread commercial adoption. Early methodologies, such as those pioneered by the Dewar group in the mid-20th century, often relied on harsh reaction conditions and multi-step sequences that resulted in poor overall yields and significant impurity profiles. These traditional routes frequently required extreme temperatures or highly reactive reagents that were difficult to handle on a large scale, posing substantial safety risks and operational costs for manufacturing facilities. Moreover, the lack of regioselectivity in older cyclization strategies often led to complex mixtures of isomers, necessitating expensive and time-consuming purification processes that eroded profit margins. For procurement managers, these inefficiencies translated into volatile pricing and unreliable supply chains for critical electronic chemical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data offers a streamlined and robust pathway that overcomes these historical bottlenecks through modern catalytic techniques. The process initiates with the activation of a naphthol derivative via triflation, followed by a palladium-catalyzed Suzuki coupling that efficiently installs the necessary vinyl functionality under mild conditions. This is subsequently followed by a chemoselective reduction of the nitro group using iron powder, a cost-effective and scalable reagent that avoids the need for high-pressure hydrogenation equipment. The final cyclization step utilizes boron trichloride and lithium aluminum hydride to close the ring and establish the B-N bond with high fidelity. This sequence not only simplifies the operational workflow but also ensures a cleaner reaction profile, directly contributing to cost reduction in organic semiconductor manufacturing by minimizing waste and maximizing throughput.
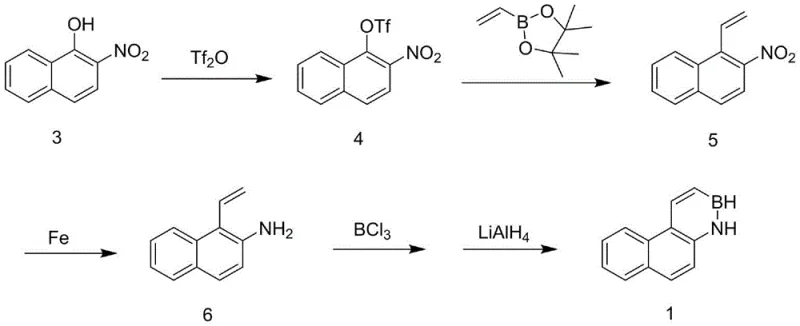
Mechanistic Insights into Electrophilic Boron Cyclization
The mechanistic elegance of this synthesis is rooted in the precise control of electrophilic aromatic substitution and transition metal catalysis. The initial triflation step converts the hydroxyl group into an excellent leaving group (triflate), which is essential for the subsequent oxidative addition of the palladium catalyst during the Suzuki coupling. This step is critical for establishing the carbon-carbon bond that extends the conjugation of the naphthalene system. Following the coupling, the reduction of the nitro group to an amine is achieved through a single-electron transfer mechanism facilitated by iron, which is particularly advantageous as it tolerates the sensitive vinyl group without causing polymerization or side reactions. The final cyclization involves the coordination of the amine nitrogen to the boron center of boron trichloride, followed by an intramolecular electrophilic attack on the vinyl double bond. This Lewis base-guided process ensures that the boron atom is incorporated exactly where needed to form the stable azaborine ring.
From an impurity control perspective, the choice of reagents plays a pivotal role in ensuring the high purity required for electronic applications. The use of iron powder for reduction, rather than catalytic hydrogenation, eliminates the risk of over-reduction of the aromatic system or the vinyl double bond, which are common pitfalls in alternative methods. Furthermore, the final reduction with lithium aluminum hydride serves to convert the intermediate chloroborane species into the desired B-H or B-substituted final product, effectively quenching any remaining reactive boron species that could act as degradation sites in the final material. This rigorous control over the reaction pathway minimizes the formation of regioisomers and oligomeric byproducts, ensuring that the final borazaphenanthrene product meets the stringent purity specifications demanded by the display industry. Such mechanistic understanding is vital for scaling up complex heterocyclic intermediates without compromising quality.
How to Synthesize Borazaphenanthrene Efficiently
The synthesis of these high-value compounds requires strict adherence to the optimized reaction conditions outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the triflate intermediate at low temperatures to prevent decomposition, followed by the Suzuki coupling which requires careful control of the aqueous-organic interface for optimal catalyst turnover. The reduction and cyclization steps demand anhydrous conditions and inert atmosphere protection to prevent the hydrolysis of sensitive boron intermediates. While the general workflow is robust, the specific stoichiometry and temperature profiles are critical for achieving the reported high yields. For detailed operational parameters and safety protocols, please refer to the standardized synthesis guide below.
- Triflation of 2-nitro-1-naphthol using trifluoromethanesulfonic anhydride to activate the hydroxyl group for subsequent cross-coupling.
- Palladium-catalyzed Suzuki coupling with vinyl pinacol borate to introduce the vinyl moiety onto the naphthalene core.
- Reduction of the nitro group to an amine using iron powder and ammonium chloride, followed by electrophilic boron cyclization with boron trichloride and lithium aluminum hydride.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers transformative benefits that extend far beyond simple chemical transformation. The reliance on commercially available starting materials, such as substituted naphthols and vinyl boronates, drastically reduces the dependency on exotic or custom-synthesized precursors that often suffer from long lead times and supply volatility. This accessibility ensures a more resilient supply chain, capable of withstanding market fluctuations and geopolitical disruptions that frequently impact the availability of specialized fine chemicals. Furthermore, the elimination of harsh conditions and the use of standard laboratory equipment for the majority of the steps lower the capital expenditure required for setting up production lines, making it an attractive option for contract development and manufacturing organizations looking to diversify their portfolio.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of inexpensive reagents like iron powder and ammonium chloride for the reduction step, replacing costly noble metal catalysts or high-pressure hydrogenation setups. Additionally, the high selectivity of the reactions minimizes the formation of byproducts, which reduces the burden on downstream purification processes such as column chromatography or recrystallization. This efficiency translates directly into lower operational expenditures and a reduced environmental footprint, aligning with modern green chemistry principles while delivering substantial cost savings in the production of high-purity OLED materials.
- Enhanced Supply Chain Reliability: By utilizing a linear and convergent synthetic strategy, the risk of bottlenecking at any single stage of production is markedly decreased. The robustness of the Suzuki coupling and the subsequent cyclization steps allows for flexible batch sizing, enabling manufacturers to respond rapidly to changes in demand without the need for extensive process re-validation. This agility is crucial for maintaining continuity of supply for downstream clients in the competitive display and lighting markets, ensuring that production schedules are met consistently without unexpected delays caused by complex or finicky chemistry.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that can be safely translated from gram-scale laboratory experiments to multi-kilogram pilot plant operations. The avoidance of extremely hazardous reagents and the generation of manageable waste streams simplify the regulatory compliance landscape, reducing the administrative burden associated with environmental health and safety reporting. This ease of scale-up facilitates the commercial production of complex polymer additives and electronic chemicals, allowing suppliers to meet the growing global demand for advanced optoelectronic materials with confidence and reliability.
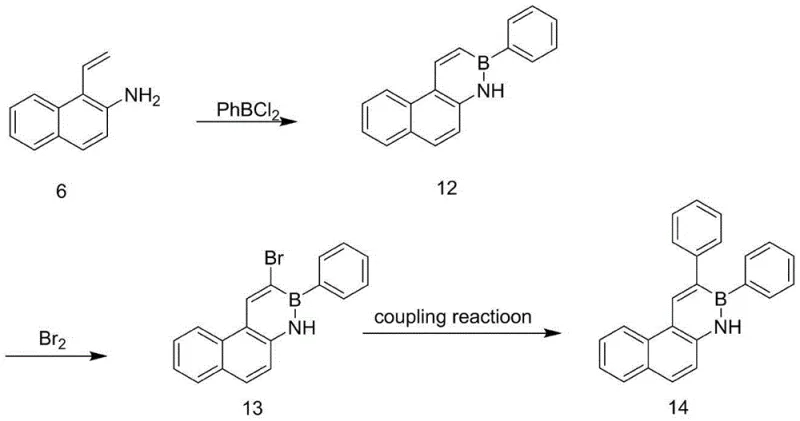
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of borazaphenanthrene derivatives. These insights are derived directly from the technical disclosures and experimental data provided in the patent documentation, offering clarity on the feasibility and advantages of this technology. Understanding these nuances is essential for stakeholders evaluating the integration of these materials into their existing product lines or research pipelines.
Q: What are the key advantages of this borazaphenanthrene synthesis method?
A: The method utilizes commercially available disubstituted naphthalene as a raw material and features simple operation, mild reaction conditions, and high yields, making it suitable for industrial scale-up.
Q: What are the potential applications of these boron-nitrogen doped compounds?
A: These compounds exhibit unique photoelectric physical properties due to the BN-doped conjugated system, making them highly valuable for organic field effect transistors, organic light emitting diodes (OLEDs), and organic photovoltaic cells.
Q: How does the BN-doping affect the electronic properties compared to all-carbon analogs?
A: Replacing a carbon-carbon double bond with a boron-nitrogen bond improves local polarity within the molecule while maintaining the aromatic configuration, effectively adjusting photoelectric properties for better device performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Borazaphenanthrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of boron-nitrogen doped polycyclic aromatic hydrocarbons in the realm of organic electronics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity borazaphenanthrene intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch delivered to our partners adheres to the highest standards required for sensitive optoelectronic applications.
We invite forward-thinking companies to collaborate with us to leverage this advanced synthetic technology for their next-generation products. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can accelerate your development timelines and optimize your supply chain for the future of organic electronics.
