Advancing Pharmaceutical Intermediates: A Catalyst-Free Route to Complex Heterocycles
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more efficient synthetic routes that can support the rigorous demands of modern drug discovery. Patent CN115043837A introduces a groundbreaking methodology for the synthesis of 4-imidazopyridylthioisoquinoline heterocyclic compounds, a structural motif that merges two pharmacologically privileged scaffolds: isoquinoline and imidazopyridine. These nitrogen-containing heterocycles are renowned for their diverse biological activities, ranging from vasodilation and antibacterial properties to potent antitumor effects. The significance of this patent lies not only in the novel molecular architectures it enables but also in its departure from traditional, resource-intensive synthetic strategies. By leveraging a direct double C-H bond sulfuration strategy, this technology offers a streamlined pathway to complex biologically active molecules, positioning it as a critical asset for R&D teams seeking to optimize their pipeline of anticancer agents.
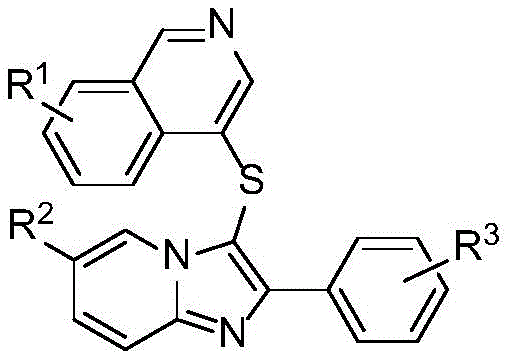
Traditionally, the construction of thioether linkages in heterocyclic systems has been fraught with challenges that hinder both economic efficiency and environmental compliance. Conventional methods often necessitate the use of malodorous and toxic thiophenols or disulfides, which pose significant safety hazards and require specialized handling infrastructure. Furthermore, many established protocols rely on pre-activated substrates, such as expensive halogenated isoquinolines, which increase the raw material cost burden substantially. Perhaps most critically for large-scale manufacturing, legacy processes frequently employ transition metal catalysts like palladium or copper. While effective, these metals introduce a major downstream bottleneck: the stringent requirement for heavy metal removal to meet pharmaceutical purity standards. This additional purification step not only extends production timelines but also generates substantial hazardous waste, complicating the supply chain for reliable pharmaceutical intermediate suppliers who must adhere to strict regulatory guidelines regarding residual metals.
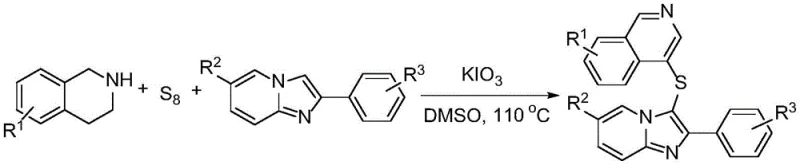
In stark contrast, the novel approach detailed in CN115043837A circumvents these historical limitations through an elegant, catalyst-free design. The core innovation involves the direct coupling of imidazo[1,2-a]pyridine compounds with 1,2,3,4-tetrahydroisoquinoline derivatives using elemental sulfur powder as the sulfur source. This reaction is mediated by potassium iodate (KIO3) acting as a mild yet effective oxidant in dimethyl sulfoxide (DMSO) solvent. By eliminating the need for transition metal catalysts entirely, the process inherently avoids the contamination issues associated with heavy metals, thereby simplifying the purification workflow. Moreover, the use of elemental sulfur—a cheap, abundant, and non-toxic commodity—replaces costly and hazardous thiol reagents. This shift represents a paradigm change in cost reduction in API manufacturing, as it removes multiple cost centers related to reagent procurement, safety management, and waste disposal, while simultaneously enhancing the atom economy of the transformation.
Mechanistic Insights into Oxidative Double C-H Sulfuration
The mechanistic elegance of this synthesis lies in its ability to activate inert C-H bonds directly without the aid of precious metal catalysts. The reaction proceeds through an oxidative sulfuration pathway where potassium iodate serves as the terminal oxidant to facilitate the formation of the C-S-C bond. In this system, DMSO acts not merely as a solvent but potentially plays a role in stabilizing reactive intermediates during the oxidation cycle. The process effectively bridges the gap between the C-4 position of the isoquinoline ring and the C-3 position of the imidazopyridine moiety via a sulfur atom. This direct functionalization avoids the thermodynamic and kinetic barriers typically associated with unactivated C-H bonds. For R&D directors focused on impurity profiles, this mechanism is particularly advantageous because it minimizes the formation of metal-complexed byproducts that are notoriously difficult to separate. The absence of a metal catalyst means the impurity spectrum is dominated by organic side products which are generally easier to remove via standard chromatographic techniques, ensuring a cleaner crude product and higher overall yield.
Furthermore, the tolerance of this reaction system towards various functional groups is a testament to its robustness. The conditions are mild enough to preserve sensitive substituents such as halogens (bromo, chloro) and electron-donating groups (methoxy) on the aromatic rings, which are often crucial for the biological activity of the final drug candidate. This chemoselectivity ensures that the synthetic route is versatile enough to generate a diverse library of analogs for structure-activity relationship (SAR) studies without requiring extensive protection-deprotection strategies. The ability to construct these complex heterocyclic frameworks in a single pot from readily available starting materials underscores the practical utility of this method for generating high-purity pharmaceutical intermediates efficiently.
How to Synthesize 4-Imidazopyridylthioisoquinoline Efficiently
The practical implementation of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale optimization and industrial production. The protocol begins with the preparation of the imidazo[1,2-a]pyridine precursor, followed by the key sulfuration step. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and thermal conditions required to maximize yield and purity. This streamlined workflow is specifically engineered to reduce lead time for high-purity intermediates by consolidating what would traditionally be a multi-step sequence into a more direct process.
- Prepare imidazo[1,2-a]pyridine compounds by reacting 2-aminopyridine derivatives with acetophenone compounds using cuprous iodide and boron trifluoride ether in DMF at 60°C.
- Combine the prepared imidazopyridine, 1,2,3,4-tetrahydroisoquinoline, elemental sulfur powder, and potassium iodate in dimethyl sulfoxide (DMSO).
- Heat the reaction mixture to 110°C for 10 hours under air atmosphere to achieve direct C-S-C bond construction, followed by standard extraction and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The elimination of expensive transition metal catalysts and hazardous thiol reagents fundamentally alters the cost structure of producing these intermediates. By relying on commodity chemicals like sulfur powder and potassium iodate, the volatility associated with sourcing specialized reagents is significantly mitigated. This stability in raw material sourcing ensures a more predictable cost base, allowing for better long-term budget planning and cost reduction in pharmaceutical intermediate manufacturing. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and solvents, further driving down the operational expenditure per kilogram of product.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the removal of transition metal catalysts. In traditional cross-coupling reactions, the cost of palladium or copper salts, combined with the expensive ligands often required, constitutes a significant portion of the COGS (Cost of Goods Sold). More importantly, the downstream processing to remove trace metals to ppm levels often requires specialized resins or repeated recrystallizations, which lowers overall yield. By adopting this catalyst-free method, manufacturers can bypass these expensive purification stages entirely. The use of elemental sulfur, which is vastly cheaper than aryl thiols or disulfides, further compresses the raw material costs. This dual saving mechanism—lower input costs and reduced processing complexity—creates a substantial margin improvement for commercial scale-up of complex heterocycles.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. Thiophenols and halogenated heterocycles are often subject to supply fluctuations due to their specialized nature and regulatory restrictions on transport and storage. In contrast, elemental sulfur and potassium iodate are bulk industrial chemicals with robust global supply chains. By shifting the dependency to these stable commodities, the risk of production delays caused by raw material shortages is drastically reduced. Furthermore, the reaction conditions (110°C in DMSO) are compatible with standard stainless steel reactors, meaning there is no need for exotic metallurgy or specialized equipment that could become a bottleneck. This compatibility ensures that the commercial scale-up of complex pharmaceutical intermediates can proceed smoothly without requiring significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to run "green" processes is becoming a license to operate rather than just a nice-to-have. This synthesis method aligns perfectly with green chemistry principles by avoiding toxic reagents and minimizing waste generation. The absence of heavy metals means the wastewater treatment load is significantly lighter, reducing the cost and complexity of effluent management. For supply chain heads, this translates to fewer regulatory hurdles and a lower risk of shutdowns due to environmental non-compliance. The high atom economy and the use of air as a co-oxidant (in some variations) or simple inorganic oxidants make this process inherently scalable. It allows for the seamless transition from gram-scale R&D batches to multi-ton commercial production, ensuring a continuous and reliable supply of critical drug intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of this route for potential partners and stakeholders.
Q: What are the primary advantages of this synthesis method over conventional thioarylation?
A: Unlike conventional methods that rely on odorous thiophenols, expensive halogenated substrates, or toxic transition metal catalysts, this patent describes a catalyst-free approach using elemental sulfur and potassium iodate. This eliminates the need for costly heavy metal removal steps and significantly improves the environmental profile of the manufacturing process.
Q: How does this method impact the scalability of pharmaceutical intermediate production?
A: The method utilizes common chemical commodities like elemental sulfur and DMSO, which are readily available and inexpensive. By avoiding sensitive catalysts and multi-step pre-activation of substrates, the process simplifies the operational workflow, making it highly suitable for commercial scale-up and reducing lead time for high-purity intermediates.
Q: What biological activities do these heterocyclic compounds exhibit?
A: Evaluation data indicates that specific derivatives within this class, particularly those with bromo or chloro substitutions, demonstrate significant inhibitory activity against human gastric cancer (BGC) and esophageal cancer (9706) cell lines, highlighting their potential as valuable scaffolds for anticancer drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Imidazopyridylthioisoquinoline Supplier
The technological breakthroughs encapsulated in CN115043837A represent a significant opportunity for pharmaceutical companies looking to innovate their anticancer pipelines with novel heterocyclic scaffolds. However, translating a patent into a commercially viable product requires a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands ready to bridge this gap, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting the most stringent purity specifications required for clinical-grade intermediates. We understand that consistency and quality are paramount, and our processes are designed to deliver batch-to-batch reproducibility that meets international regulatory standards.
We invite R&D and procurement leaders to engage with us to explore how this catalyst-free synthesis can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Together, we can accelerate the development of next-generation therapeutics while optimizing cost and sustainability.
