Advanced Manufacturing of Heterocyclic Arylamines for Oncology Drug Development
Advanced Manufacturing of Heterocyclic Arylamines for Oncology Drug Development
The pharmaceutical industry is constantly seeking robust synthetic routes for novel heterocyclic scaffolds that exhibit potent biological activity, particularly in the field of oncology. Patent CN112250639B introduces a significant advancement in the preparation of heterocyclic substituted arylamine compounds, specifically those containing a triazine core structure. These compounds are not merely theoretical constructs but have demonstrated tangible efficacy against human osteosarcoma cells, positioning them as high-value candidates for next-generation anticancer therapeutics. The disclosed methodology offers a streamlined, catalytic approach that overcomes many of the thermodynamic and kinetic barriers associated with traditional triazine functionalization. By leveraging transition metal catalysis, this technology enables the construction of complex molecular architectures under relatively mild conditions, which is a critical factor for maintaining product integrity and minimizing degradation pathways during scale-up.
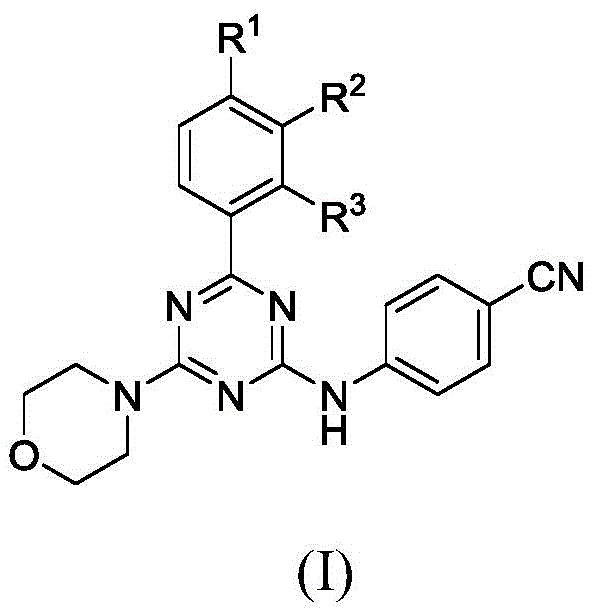
For R&D directors and process chemists, the structural versatility offered by this platform is paramount. The general formula (I) allows for extensive diversification at the R1, R2, and R3 positions, enabling the rapid generation of analog libraries for structure-activity relationship (SAR) studies. This flexibility is essential for optimizing pharmacokinetic properties and reducing off-target toxicity in early-stage drug discovery. Furthermore, the inclusion of a morpholine moiety and a cyano-substituted aniline group provides specific hydrogen-bonding acceptors and donors that are often crucial for binding affinity within kinase active sites or other protein targets. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this chemical space allows us to support clients in navigating the complex landscape of oncology drug development with precision and speed.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted triazine derivatives has been plagued by several significant challenges that hinder efficient commercial production. Traditional methods often rely on harsh reaction conditions, such as extremely high temperatures or the use of strong, corrosive acids, which can lead to the decomposition of sensitive functional groups and the formation of complex impurity profiles. Moreover, conventional aromatization strategies frequently require stoichiometric amounts of toxic oxidants, generating substantial quantities of hazardous waste that complicate downstream processing and environmental compliance. The lack of regioselectivity in older coupling protocols often results in difficult-to-separate isomers, drastically reducing overall yield and increasing the cost of goods sold. These inefficiencies create bottlenecks in the supply chain, making it difficult to secure consistent, high-purity material for clinical trials and eventual market launch.
The Novel Approach
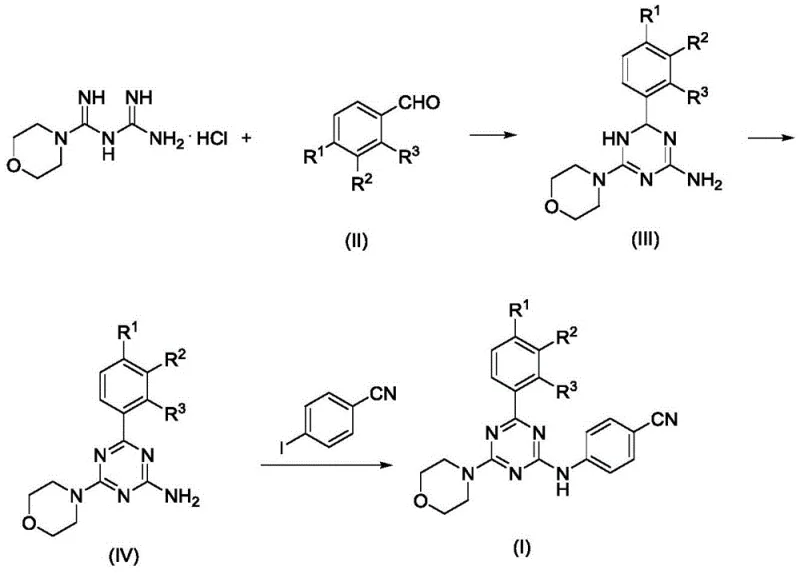
In stark contrast, the methodology described in the patent utilizes a sophisticated multi-step catalytic sequence that addresses these legacy issues with elegance and efficiency. The process begins with a condensation reaction to form a dihydrotriazine intermediate, followed by a ruthenium-catalyzed dehydrogenation to achieve aromatization without the need for stoichiometric oxidants. This is subsequently followed by a copper-catalyzed C-N coupling reaction to install the critical arylamine functionality. This stepwise approach allows for precise control over each transformation, ensuring high conversion rates and minimizing side reactions. The use of catalytic amounts of metals like ruthenium and copper significantly reduces the heavy metal load in the final product, simplifying purification and ensuring compliance with strict ICH guidelines for residual solvents and metals in pharmaceutical ingredients.
Mechanistic Insights into Ru/Cu-Catalyzed Triazine Functionalization
The core of this technological breakthrough lies in the mechanistic efficiency of the transition metal catalysis employed in the second and third steps. The dehydrogenation step, mediated by Ru(PPh3)3Cl2 in the presence of a base like potassium tert-butoxide, facilitates the removal of hydrogen from the dihydrotriazine ring to form the stable aromatic triazine system. This oxidative dehydrogenation is thermodynamically favorable and proceeds with high atom economy, as the only byproduct is typically hydrogen gas or a reduced species of the base, rather than heavy metal waste. Following aromatization, the introduction of the p-cyanoaniline group via Ullmann-type coupling is achieved using cuprous iodide and a diamine ligand. This copper-catalyzed cross-coupling is highly tolerant of various functional groups, allowing for the incorporation of electron-withdrawing groups like fluorine or trifluoromethyl without compromising reaction efficiency.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic alternatives. The specificity of the metal-ligand complexes ensures that the reaction occurs primarily at the desired halogenated position on the triazine ring, minimizing the formation of regioisomers that are notoriously difficult to remove via crystallization. Furthermore, the mild reaction temperatures—101°C for dehydrogenation and 82°C for coupling—prevent thermal degradation of the sensitive nitrile group and the morpholine ring. This thermal stability is crucial for maintaining the optical purity and structural integrity of the molecule, which directly correlates to its biological efficacy. By understanding these mechanistic details, manufacturers can optimize reaction parameters such as solvent choice (dioxane vs. toluene) and catalyst loading to maximize yield and minimize production costs.
How to Synthesize Heterocyclic Substituted Arylamine Efficiently
The synthesis of these high-value oncology intermediates requires a disciplined approach to process parameters to ensure reproducibility and safety on a commercial scale. The patented route outlines a clear three-step protocol that balances reaction kinetics with operational simplicity, making it highly suitable for transfer from the laboratory to pilot and production plants. The initial condensation sets the foundation for the molecular scaffold, while the subsequent catalytic steps refine the structure into the bioactive conformation. Detailed standard operating procedures regarding reagent addition rates, temperature ramping, and workup protocols are essential to mitigate risks associated with exothermic events and to ensure consistent product quality batch after batch.
- Condense morpholine biguanide hydrochloride with substituted benzaldehydes in acetic acid at 118°C to form dihydrotriazine intermediates.
- Perform catalytic dehydrogenation using Ru(PPh3)3Cl2 and potassium tert-butoxide in dioxane at 101°C to aromatize the triazine ring.
- Execute copper-catalyzed Ullmann-type coupling with p-cyanoiodobenzene in acetonitrile at 82°C to finalize the arylamine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere chemical yield. The reliance on commodity chemicals such as morpholine biguanide hydrochloride and various substituted benzaldehydes ensures that the raw material supply chain is robust and resistant to market volatility. Unlike processes that depend on exotic, single-source reagents, this method leverages widely available feedstocks, significantly reducing the risk of supply disruptions that can halt production lines. Furthermore, the elimination of stoichiometric oxidants and the use of recoverable catalysts contribute to a greener manufacturing profile, which aligns with the increasingly stringent environmental regulations faced by modern chemical enterprises.
- Cost Reduction in Manufacturing: The catalytic nature of the dehydrogenation and coupling steps dramatically lowers the consumption of expensive reagents compared to traditional stoichiometric methods. By utilizing low loadings of ruthenium and copper catalysts, the direct material costs are significantly optimized. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, leading to lower utility costs per kilogram of product. The simplified workup procedures, which avoid complex quenching of hazardous oxidants, further reduce labor and waste disposal expenses, resulting in a more competitive cost structure for the final API intermediate.
- Enhanced Supply Chain Reliability: The modular nature of this synthesis allows for flexible manufacturing strategies. Since the dihydrotriazine intermediate can be produced and stockpiled independently, production can be decoupled into manageable stages, enhancing overall throughput capacity. The use of common organic solvents like acetic acid, dioxane, and acetonitrile ensures that solvent supply is never a bottleneck. This reliability is critical for meeting tight delivery schedules for clinical trial materials and commercial launches, providing partners with a dependable source of high-quality intermediates that keeps their drug development timelines on track.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily managed in standard stainless steel reactors. The absence of highly toxic reagents simplifies the engineering controls required for operator safety and environmental protection. Waste streams are less hazardous and easier to treat, reducing the burden on effluent treatment plants and lowering the overall environmental footprint of the manufacturing site. This compliance readiness accelerates regulatory approvals and facilitates smoother audits, ensuring uninterrupted supply to global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triazine-based compounds. Understanding these details helps stakeholders make informed decisions about integrating this technology into their development pipelines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industrial applications.
Q: What represents the key innovation in this triazine synthesis pathway?
A: The patent utilizes a tandem catalytic approach featuring ruthenium-mediated dehydrogenation followed by copper-catalyzed amination, avoiding harsh stoichiometric oxidants and enabling milder reaction conditions compared to traditional methods.
Q: What biological activity do these heterocyclic compounds exhibit?
A: The synthesized compounds, particularly derivatives I-4 and I-6, demonstrate significant inhibitory activity against human osteosarcoma cells (U2OS), with IC50 values as low as 2.13 μM, indicating strong potential for oncology drug development.
Q: Are the raw materials for this process commercially scalable?
A: Yes, the process relies on readily available starting materials such as morpholine biguanide hydrochloride and substituted benzaldehydes, which are commodity chemicals, ensuring robust supply chain continuity for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazine Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving oncology therapies. Our team of expert process chemists has thoroughly analyzed the synthetic route described in CN112250639B and is fully prepared to execute this chemistry with the highest standards of quality and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demands whether you are in the early discovery phase or preparing for global market launch. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch we deliver.
We invite you to collaborate with us to leverage this innovative technology for your drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can accelerate your path to market while optimizing your overall project economics.
