Advanced Synthesis Of Chiral Biphenyl Diol Catalysts For Industrial Asymmetric Catalysis
Introduction To The Novel Catalytic Framework
The landscape of asymmetric catalysis is continually evolving, driven by the demand for more efficient and selective chiral ligands that can operate under mild industrial conditions. Patent CN115490732B introduces a significant advancement in this domain by disclosing a robust synthesis method for chiral 5,5',6'-tetramethyl-3,3'-di-tert-butyl-1,1'-biphenyl-2,2'-diol selenium and sulfide derivatives. These compounds represent a critical class of phosphoramidite ligands that have shown immense potential in facilitating high-enantioselective transformations, particularly in the synthesis of complex pharmaceutical intermediates and fine chemicals. The core innovation lies in the strategic combination of phosphorus trichloride, triethylamine, and various alkylamine reagents with the chiral biphenyl backbone, followed by a decisive chalcogenation step using selenium or sulfur powder. This approach not only streamlines the production of these high-value catalysts but also ensures that the resulting molecular architectures possess the necessary steric and electronic properties to induce chirality in downstream reactions with exceptional fidelity.
For research and development directors overseeing process chemistry, the implications of this patent are profound, as it offers a reliable pathway to access ligands that were previously difficult or costly to manufacture. The ability to tune the nitrogen substituents—ranging from ethyl to isobutyl groups—allows for precise optimization of the catalyst's performance across different reaction types. Furthermore, the methodology described avoids the use of exotic or hazardous reagents, relying instead on commodity chemicals that are easily sourced from the global supply chain. This accessibility is a key factor in reducing the barrier to entry for adopting these advanced catalytic systems in large-scale manufacturing environments, thereby supporting the broader goal of sustainable and cost-effective chemical production.
The Limitations Of Conventional Methods Vs. The Novel Approach
The Limitations Of Conventional Methods
Historically, the synthesis of chiral phosphoramidite ligands has been plagued by several inherent inefficiencies that hinder their widespread adoption in industrial settings. Traditional routes often necessitate the use of pre-functionalized phosphorus chlorides or require multiple synthetic steps involving sensitive protecting groups to preserve the integrity of the chiral backbone during functionalization. These multi-step sequences inevitably lead to cumulative yield losses, increased solvent consumption, and a significant generation of chemical waste, all of which drive up the overall cost of goods sold (COGS). Moreover, many conventional protocols demand stringent anhydrous conditions and cryogenic temperatures that are energy-intensive and difficult to maintain consistently on a commercial scale. The reliance on specialized reagents that are not readily available in bulk quantities further exacerbates supply chain vulnerabilities, creating bottlenecks that can delay project timelines and increase procurement risks for manufacturing teams.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN115490732B presents a streamlined one-pot strategy that dramatically simplifies the construction of the target selenium and sulfide catalysts. By utilizing phosphorus trichloride as the central phosphorus source and reacting it directly with the chiral diol and amine components in a single vessel, the process eliminates the need for intermediate isolation and purification steps. This telescoped approach not only enhances the overall atomic economy of the reaction but also significantly reduces the operational complexity associated with handling reactive intermediates. The reaction conditions are notably mild, proceeding efficiently from 0°C to room temperature in dichloromethane, which is a standard solvent in fine chemical synthesis. This operational simplicity translates directly into reduced labor costs and shorter cycle times, making the technology highly attractive for scale-up operations.
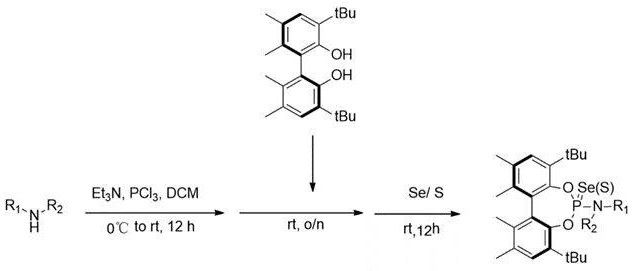
The versatility of this novel approach is further evidenced by its compatibility with a diverse range of alkylamine substrates, allowing for the rapid generation of a library of catalyst variants to screen for optimal activity. The final chalcogenation step, involving the addition of elemental selenium or sulfur, is straightforward and high-yielding, ensuring that the desired P-Se or P-S bonds are formed with high selectivity. This robustness makes the method an ideal candidate for reliable agrochemical intermediate supplier networks and pharmaceutical manufacturers seeking to diversify their catalytic toolkit without compromising on quality or consistency.
Mechanistic Insights Into Phosphoramidite Chalcogenation
The mechanistic pathway underpinning this synthesis involves a sequential nucleophilic substitution and oxidation process that is both elegant and efficient. Initially, phosphorus trichloride reacts with triethylamine and the selected alkylamine to generate a reactive chlorophosphine intermediate in situ. This species is highly electrophilic and readily undergoes nucleophilic attack by the hydroxyl groups of the chiral 5,5',6'-tetramethyl-3,3'-di-tert-butyl-1,1'-biphenyl-2,2'-diol. The steric bulk of the tert-butyl and methyl groups on the biphenyl backbone plays a crucial role here, directing the formation of the phosphite ester while preventing unwanted side reactions such as oligomerization. Once the phosphite framework is established, the introduction of selenium or sulfur powder serves as the oxidizing agent, converting the trivalent phosphorus center into the corresponding selenide or sulfide. This oxidation step is thermodynamically favorable and proceeds cleanly at room temperature, preserving the axial chirality of the biphenyl scaffold which is essential for the catalyst's enantioinduction capabilities.
From an impurity control perspective, the one-pot nature of the reaction minimizes the exposure of reactive intermediates to atmospheric moisture and oxygen, which are common sources of degradation in phosphorus chemistry. The use of excess selenium or sulfur ensures complete conversion of the phosphite precursor, driving the equilibrium towards the desired product and minimizing the presence of unreacted starting materials in the crude mixture. Subsequent purification via reduced pressure distillation and column chromatography effectively removes inorganic salts and excess reagents, yielding a high-purity catalyst suitable for sensitive asymmetric transformations. This level of purity is critical for R&D teams focused on developing robust processes for high-purity OLED material or API intermediate manufacturing, where trace metal or halide contaminants can poison downstream catalysts or affect product stability.
How To Synthesize Chiral Biphenyl Diol Derivatives Efficiently
The practical implementation of this synthesis protocol is designed to be accessible to process chemists equipped with standard laboratory infrastructure. The procedure begins with the dissolution of phosphorus trichloride in ultra-dry dichloromethane, followed by the controlled addition of triethylamine at low temperature to manage the exotherm. After warming to room temperature, the alkylamine is introduced, and the mixture is stirred to allow for the formation of the amino-chlorophosphine species. The chiral diol is then added, and the reaction is allowed to proceed overnight to ensure complete coupling. Finally, the chalcogen source is added, and the reaction is stirred for an additional period to effect oxidation. Detailed standardized synthesis steps see the guide below.
- Dissolve phosphorus trichloride in dichloromethane and cool to 0°C, then add triethylamine and warm to room temperature before introducing the alkylamine reagent.
- Add the chiral 5,5',6'-tetramethyl-3,3'-di-tert-butyl-1,1'-biphenyl-2,2'-diol to the reaction mixture at room temperature and stir for approximately 12 hours.
- Introduce selenium powder or sulfur powder to the system at room temperature, continue stirring for 12 hours, and purify the final product via distillation and column chromatography.
Commercial Advantages For Procurement And Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis methodology offers tangible benefits that extend beyond mere technical performance. The reliance on commodity chemicals such as phosphorus trichloride, triethylamine, and common alkylamines means that the raw material supply base is broad and competitive, reducing the risk of single-source dependency. This diversity in sourcing options allows purchasing teams to negotiate better pricing and secure long-term supply agreements, thereby stabilizing the cost structure for catalyst production. Furthermore, the elimination of complex multi-step sequences reduces the overall inventory holding costs and working capital requirements, as fewer intermediates need to be stored and managed throughout the production lifecycle.
- Cost Reduction In Manufacturing: The one-pot design of this process inherently lowers manufacturing costs by consolidating multiple reaction steps into a single unit operation. This consolidation reduces the consumption of solvents, energy, and labor hours, leading to substantial cost savings in the overall production budget. Additionally, the avoidance of expensive protecting group chemistry and specialized reagents further drives down the direct material costs, making the final catalyst more economically viable for large-scale applications. The high yields reported for various substrates also contribute to cost efficiency by maximizing the output per batch and minimizing waste disposal fees associated with low-yielding processes.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials, the supply chain for these catalysts becomes significantly more resilient to market fluctuations and geopolitical disruptions. The simplicity of the synthesis also means that production can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without the need for specialized equipment or extensive retraining of personnel. This flexibility ensures continuity of supply, which is paramount for maintaining uninterrupted production schedules in the pharmaceutical and agrochemical sectors. The robustness of the reaction conditions further guarantees consistent product quality across different batches, reducing the need for extensive quality control interventions.
- Scalability And Environmental Compliance: The mild reaction conditions and the use of standard solvents like dichloromethane facilitate easy scale-up from kilogram to tonne levels without encountering significant engineering challenges. The process generates minimal hazardous waste compared to traditional methods, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The ability to recycle solvents and recover excess reagents adds another layer of environmental compliance and cost recovery. This green chemistry profile enhances the marketability of the final products to eco-conscious customers and helps companies meet their carbon footprint reduction targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral catalysts, based on the detailed disclosures within the patent documentation. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this technology into their existing workflows. The answers provided reflect the specific experimental data and scope defined in the intellectual property, ensuring accuracy and relevance for decision-making processes.
Q: What are the primary advantages of this one-pot synthesis method for chiral ligands?
A: The method utilizes economical and readily available raw materials such as phosphorus trichloride and common alkylamines, avoiding complex multi-step protection and deprotection sequences typically required in traditional ligand synthesis.
Q: Can this synthesis protocol be adapted for different alkyl substituents?
A: Yes, the patent demonstrates wide substrate applicability, successfully synthesizing derivatives with ethyl, isopropyl, and isobutyl groups on the nitrogen atom with yields ranging from 44% to 89%.
Q: What specific catalytic applications are demonstrated for these new selenium/sulfide derivatives?
A: The synthesized catalysts are effectively applied in the asymmetric sulfenylation of biphenyl diols, achieving high yields and demonstrating enantioselectivity when combined with chiral phosphoric acids.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable Chiral Biphenyl Diol Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands in driving the next generation of asymmetric synthesis. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that meet the most rigorous specifications, supported by our state-of-the-art analytical capabilities and rigorous QC labs. Our infrastructure is designed to handle complex chemistries with precision, guaranteeing the consistency and reliability that global pharmaceutical and fine chemical partners demand.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and process constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your project timelines while optimizing your overall manufacturing economics. Together, we can unlock the full potential of chiral catalysis to create value and drive innovation in the chemical industry.
