Scalable Silver-Catalyzed Synthesis of 3-Sulfonyl-1,2-Dihydronaphthalenes for Advanced Pharmaceutical Applications
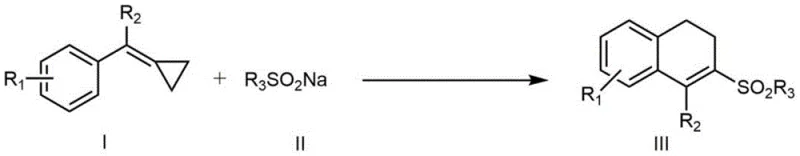
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex sulfur-containing scaffolds, which serve as critical building blocks for bioactive molecules. Patent CN109651210B introduces a groundbreaking preparation method for 3-sulfonyl-1,2-dihydronaphthalene compounds, addressing significant synthetic challenges in accessing these privileged structures. This technology leverages a silver-catalyzed cascade reaction involving methylenecyclopropane derivatives and organic sodium sulfinates, enabling the efficient construction of the dihydronaphthalene core through C-C sigma bond cleavage and intramolecular cyclization. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more accessible and versatile synthetic routes for generating sulfone-functionalized intermediates used in treatments for Alzheimer's disease, HIV, and cancer.
Historically, the synthesis of sulfone-containing polycyclic systems often relied on multi-step sequences involving harsh reagents, unstable intermediates, or expensive transition metal catalysts that were difficult to remove from the final API. Conventional approaches frequently suffered from limited substrate scope, poor atom economy, and the generation of hazardous waste streams, creating bottlenecks in both process development and commercial manufacturing. These legacy methods often required cryogenic conditions or stoichiometric amounts of toxic heavy metals, complicating regulatory compliance and increasing the overall cost of goods sold (COGS) for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for accessing 3-sulfonyl-1,2-dihydronaphthalenes typically involved Friedel-Crafts type sulfonylations or cross-coupling reactions that demanded rigorous exclusion of moisture and oxygen, alongside the use of pyrophoric reagents. These processes often exhibited low functional group tolerance, necessitating extensive protecting group manipulations that increased step counts and reduced overall yield. Furthermore, the activation of inert C-C bonds in cyclic precursors usually required extreme thermal conditions or highly specialized catalysts that were not economically viable for large-scale production, leading to inconsistent batch quality and supply chain vulnerabilities for downstream drug manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN109651210B utilizes a mild, silver-promoted oxidative system that operates effectively at moderate temperatures around 80°C. By employing readily available sodium sulfinates and methylenecyclopropanes, this route bypasses the need for pre-functionalized halides or organometallic reagents, significantly simplifying the starting material supply chain. The reaction proceeds through a unique mechanism where the silver catalyst facilitates the activation of the strained cyclopropane ring, triggering a cascade of sulfonylation and cyclization events that construct the naphthalene skeleton in a single operational step with high atom efficiency.
Mechanistic Insights into Silver-Catalyzed C-C Bond Activation
The core innovation of this process lies in the ability of the silver catalyst, specifically AgNO3, to coordinate with the alkene moiety of the methylenecyclopropane, thereby lowering the activation energy for C-C sigma bond cleavage. In the presence of a persulfate oxidant like K2S2O8, the reaction likely proceeds via a radical pathway where the sulfinate species is oxidized to a sulfonyl radical, which then adds to the activated cyclopropane system. This addition triggers the ring-opening of the three-membered carbocycle, generating a distal radical intermediate that subsequently undergoes intramolecular electrophilic aromatic substitution to close the six-membered ring, restoring aromaticity and forming the stable dihydronaphthalene product.
From an impurity control perspective, the mild reaction conditions and the specific selectivity of the silver catalyst minimize the formation of polymeric byproducts or over-oxidized species that often plague radical reactions. The use of toluene as a solvent provides an optimal balance of solubility for both organic substrates and inorganic salts, ensuring homogeneous reaction kinetics that lead to consistent product profiles. This mechanistic clarity allows process chemists to confidently scale the reaction, knowing that the primary side reactions are suppressed by the controlled generation of radical species and the rapid cyclization step that traps the reactive intermediate.
How to Synthesize 3-Sulfonyl-1,2-Dihydronaphthalene Efficiently
To implement this synthesis effectively, operators must adhere to strict protocols regarding atmosphere control and reagent stoichiometry to maximize yield and purity. The process begins with the precise weighing of the methylenecyclopropane substrate and the sodium sulfinate salt, followed by the addition of the silver catalyst and oxidant in anhydrous toluene. Detailed standardized operating procedures for mixing, heating, and workup are essential to ensure reproducibility across different batch sizes, from laboratory grams to pilot plant kilograms.
- Charge a Schlenk sealed tube reactor with methylenecyclopropane compound (Formula I), sodium sulfinate (Formula II), AgNO3 catalyst (10 mol%), and K2S2O8 oxidant (2 equiv) in toluene solvent.
- Heat the mixture to 80°C under an argon atmosphere with stirring for 36 to 48 hours, monitoring progress via TLC or GC-MS until completion.
- Upon completion, wash the mixture with saturated brine, extract the aqueous phase with ethyl acetate, dry the combined organic layers over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver-catalyzed methodology offers substantial strategic benefits by streamlining the sourcing of critical raw materials and reducing operational complexity. The reliance on commercially available sodium sulfinates and simple methylenecyclopropanes eliminates the need for custom-synthesized, high-cost precursors that often have long lead times and single-source dependencies. This shift to commodity-grade starting materials enhances supply chain resilience, ensuring that production schedules are not disrupted by the scarcity of exotic reagents.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions and the use of a relatively inexpensive silver salt catalyst in catalytic amounts rather than stoichiometric quantities significantly lowers the direct material costs associated with the synthesis. Furthermore, the simplified workup procedure, which avoids complex extraction or distillation steps, reduces energy consumption and labor hours, contributing to a leaner manufacturing process that improves overall margin potential without compromising product quality.
- Enhanced Supply Chain Reliability: By utilizing robust reaction conditions that tolerate air and moisture to a reasonable extent (though inert atmosphere is preferred for optimization), the process reduces the risk of batch failures due to minor environmental fluctuations. The broad substrate scope means that a single manufacturing line can be adapted to produce a diverse library of sulfone derivatives, allowing for flexible inventory management and rapid response to changing market demands for specific pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste compared to traditional methods that might utilize chlorinated solvents or heavy metal stoichiometric oxidants. The use of toluene, a common industrial solvent, facilitates easier recycling and disposal compliance, while the high yields reported in the patent reduce the volume of waste per kilogram of product, aligning with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability, safety, and optimization of this novel synthetic route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this chemistry into their existing production workflows.
Q: What are the optimal reaction conditions for this silver-catalyzed cyclization?
A: The patent specifies that the optimal conditions involve using AgNO3 (10 mol%) as the catalyst and K2S2O8 (2 equiv) as the oxidant in toluene at 80°C for 36-48 hours under an inert argon atmosphere, yielding up to 88% isolated product.
Q: Does this method support a wide range of substrate substituents?
A: Yes, the methodology demonstrates excellent substrate tolerance, accommodating various electron-donating and electron-withdrawing groups on both the methylenecyclopropane and the sodium sulfinate components, including halogens, alkoxy groups, and heterocycles.
Q: How is the final product purified to meet pharmaceutical standards?
A: The crude reaction mixture is subjected to a standard workup involving brine washing and ethyl acetate extraction, followed by purification using silica gel column chromatography with a hexane/ethyl acetate gradient to ensure high purity suitable for downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Sulfonyl-1,2-Dihydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the silver-catalyzed synthesis described in CN109651210B for advancing the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-sulfonyl-1,2-dihydronaphthalene meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and reliability for your supply chain.
