Scalable Synthesis of Stable Diradical Intermediates for Molecular Ferromagnets
Scalable Synthesis of Stable Diradical Intermediates for Molecular Ferromagnets
The landscape of advanced functional materials is rapidly evolving, driven by the demand for high-performance molecular magnets and spintronic components. A pivotal development in this sector is detailed in patent CN101786989A, which discloses a robust preparation method for 4,4'-bis(4,4,5,5-tetramethylimidazoline-3-oxide-1-oxyl) phenyl ether. This specific diradical compound represents a significant breakthrough because it combines structural stability with the unique magnetic properties required for coordinating with transition metals. Unlike transient radical species that degrade rapidly, this molecule maintains integrity under ambient conditions, making it an ideal candidate for industrial-scale applications in electronic chemical manufacturing. The patent outlines a streamlined synthetic pathway that avoids the pitfalls of low-yield, multi-step protections typically associated with radical chemistry.
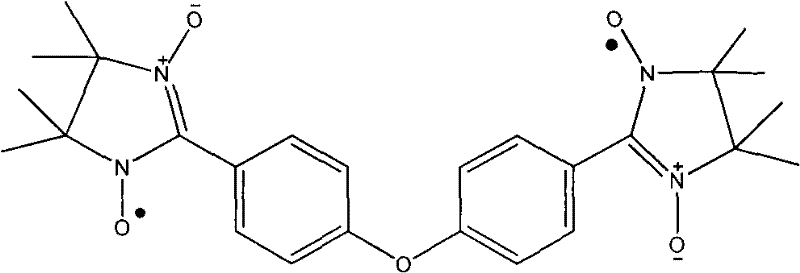
For research directors and procurement specialists, the implications of this technology are profound. The ability to produce such a complex diradical through a simplified process translates directly into cost reduction in advanced magnetic material intermediate manufacturing. By utilizing readily available starting materials like 2-nitropropane and 4,4'-diformyl diphenyl ether, the process minimizes supply chain bottlenecks. Furthermore, the operational simplicity—characterized by standard reflux and stirring conditions rather than exotic high-pressure or cryogenic setups—suggests that this methodology is highly amenable to commercial scale-up. As a reliable advanced magnetic material intermediate supplier, understanding these mechanistic advantages allows us to offer clients a product that balances high purity with economic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of molecular ferromagnets has been hindered by the instability and structural limitations of available radical ligands. Most conventional free radicals are single-radical species that, when coordinated with paramagnetic metal ions, often fail to establish the long-range magnetic order necessary for practical device functionality. Traditional synthetic routes for generating multi-radical systems are notoriously cumbersome, frequently requiring harsh reaction conditions, sensitive protecting groups, and extensive purification protocols that drive up costs and reduce overall yield. Additionally, many existing radicals decompose rapidly at room temperature or in polar solvents, severely restricting their utility in solution-phase processing and device fabrication. This instability creates a significant barrier for supply chain heads who require consistent, shelf-stable inventory for continuous production lines.
The Novel Approach
The methodology presented in the patent data introduces a paradigm shift by focusing on the Ullman-type nitroxide radical framework, which is inherently more stable. The novel approach leverages a convergent synthesis strategy where a symmetric dialdehyde is condensed with a dihydroxylamine derivative. This design not only ensures the formation of a symmetric diradical capable of bridging metal centers effectively but also drastically simplifies the operational workflow. The process eliminates the need for complex chromatographic separations at intermediate stages, as the crude imidazolidine product can often proceed directly to oxidation. This efficiency is a game-changer for commercial scale-up of complex polymer additives and electronic materials, as it reduces solvent consumption and waste generation while maximizing the throughput of the final crystalline product.
Mechanistic Insights into Nitroxide Radical Formation
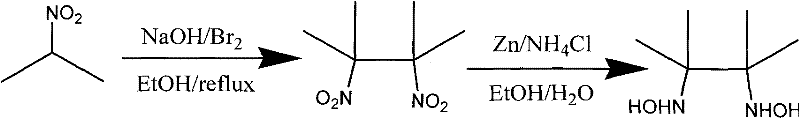
The core of this synthesis lies in the precise construction of the imidazolidine ring followed by a controlled oxidation to generate the nitroxide moiety. The process begins with the preparation of 2,3-dimethyl-2,3-dihydroxylaminobutane, achieved through the reduction of the corresponding dinitro compound using zinc powder in an ethanol-water system. This step is critical as it generates the nucleophilic nitrogen species required for the subsequent cyclization. The reaction conditions are mild, utilizing ice-water baths initially to control exothermicity during the addition of zinc, followed by stirring at room temperature to ensure complete reduction. The resulting dihydroxylamine is then isolated and purified, serving as the key building block for the radical scaffold.
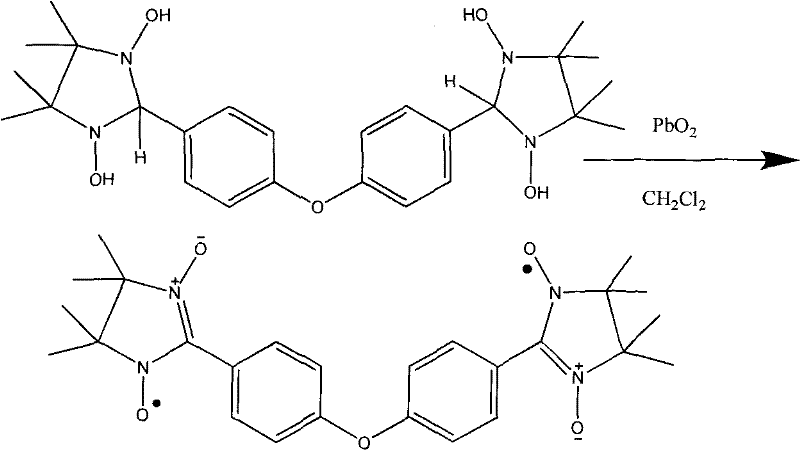
Following the preparation of the amine, the cyclization step involves reacting the dihydroxylamine with 4,4'-diformyl diphenyl ether in anhydrous methanol. This condensation reaction proceeds under reflux conditions, driving the equilibrium towards the formation of the five-membered imidazolidine rings. The symmetry of the diphenyl ether linker ensures that the two radical centers are spatially separated yet connected, a geometry that is essential for mediating magnetic exchange interactions in the final metal complexes. The final transformation involves the oxidation of the imidazolidine intermediate using lead dioxide (PbO2) in dichloromethane. This heterogeneous oxidation selectively converts the hydroxylamine groups into stable nitroxide radicals without degrading the organic backbone, yielding the target blue-black crystalline diradical.
How to Synthesize 4,4'-Bis(tetramethylimidazoline) Phenyl Ether Efficiently
The synthesis protocol described in the patent offers a reproducible pathway for generating high-purity diradicals suitable for research and industrial applications. The process is designed to minimize technical risk by using standard laboratory equipment and widely available reagents. Operators should pay close attention to the stoichiometry during the condensation phase, maintaining a 2:1 molar ratio of dihydroxylamine to dialdehyde to prevent oligomerization. The oxidation step requires vigorous stirring to ensure efficient contact between the solid oxidant and the dissolved substrate. For a detailed breakdown of the specific temperatures, reaction times, and workup procedures, please refer to the standardized guide below.
- Synthesize 2,3-dimethyl-2,3-dihydroxylaminobutane via zinc reduction of the corresponding dinitro compound in ethanol/water.
- Condense the dihydroxylamine with 4,4'-diformyl diphenyl ether in anhydrous methanol under reflux to form the imidazolidine intermediate.
- Oxidize the imidazolidine intermediate using lead dioxide (PbO2) in dichloromethane to generate the final stable nitroxide diradical.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers distinct advantages that align with the strategic goals of modern chemical procurement. The elimination of transition metal catalysts in the final oxidation step, relying instead on stoichiometric lead dioxide, simplifies the removal of metal impurities, which is often a costly and time-consuming bottleneck in pharmaceutical and electronic grade chemical production. This simplification leads to substantial cost savings by reducing the number of unit operations and the volume of solvents required for purification. Furthermore, the use of commodity chemicals as starting materials insulates the supply chain from the volatility associated with specialized reagents, ensuring a more predictable and reliable sourcing strategy for long-term projects.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by streamlining the synthetic sequence. By avoiding the need for expensive chiral catalysts or cryogenic conditions, the operational expenditure (OPEX) is drastically lowered. The ability to skip intermediate purification steps for the imidazolidine precursor further reduces labor and material costs, allowing for a leaner manufacturing model that maximizes margin potential without compromising on the quality of the final diradical product.
- Enhanced Supply Chain Reliability: The reliance on bulk chemicals such as 2-nitropropane, bromine, and zinc powder ensures that raw material availability is rarely a constraint. Unlike processes dependent on bespoke ligands or rare earth metals, this route leverages a mature global supply base. This robustness translates to shorter lead times and greater flexibility in production scheduling, enabling suppliers to respond rapidly to fluctuating market demands for advanced electronic materials and magnetic precursors.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot production. The use of standard solvents like methanol and dichloromethane allows for established recovery and recycling protocols, minimizing environmental impact. While lead dioxide is used, its heterogeneous nature facilitates easy filtration and disposal or recycling, adhering to stringent environmental regulations while maintaining high throughput capabilities for industrial clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this diradical compound. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation. Understanding these nuances is essential for R&D teams evaluating this material for inclusion in new molecular magnet architectures or spintronic devices.
Q: What is the primary application of this diradical compound?
A: This compound serves as a crucial building block for synthesizing molecular ferromagnets. When coordinated with transition metals like Co(II), it forms chain structures exhibiting magnetic ordering, which is vital for developing next-generation spintronic and memory storage devices.
Q: How does this synthesis method improve upon traditional radical production?
A: Unlike traditional methods that often yield unstable single radicals or require complex multi-step protections, this patented route utilizes a direct condensation and oxidation strategy. It significantly simplifies the workflow by allowing the intermediate to proceed to the next step without rigorous purification, thereby enhancing overall throughput.
Q: Is the final product stable under standard conditions?
A: Yes, the resulting Ullman-type nitroxide radical demonstrates exceptional stability at room temperature and in polar solvents. This stability overcomes a major limitation of typical free radicals, facilitating easier handling, storage, and integration into downstream coordination chemistry processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(tetramethylimidazoline) Phenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the development of next-generation technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market deployment is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4,4'-bis(4,4,5,5-tetramethylimidazoline-3-oxide-1-oxyl) phenyl ether meets the exacting standards required for molecular ferromagnet synthesis. Our commitment to quality assurance means you can rely on us for consistent supply continuity.
We invite you to collaborate with us to optimize your supply chain for advanced materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your innovation goals while driving down overall project costs.
