Advanced Spirocyclic Bisphosphine Ligands for High-Efficiency Asymmetric Catalytic Hydrogenation
Advanced Spirocyclic Bisphosphine Ligands for High-Efficiency Asymmetric Catalytic Hydrogenation
The landscape of asymmetric synthesis is continually evolving, driven by the demand for higher purity and more efficient catalytic systems in the production of active pharmaceutical ingredients. A pivotal advancement in this field is documented in Chinese Patent CN1439643A, which introduces a novel class of spirocyclic bisphosphine ligands characterized by a rigid spiroindane backbone. This structural motif imparts axial chirality, resulting in distinct dextrorotatory and levorotatory isomers that serve as highly effective chiral inducers. The patent details a robust synthetic pathway starting from spirocyclic diphenols, utilizing trifluoromethanesulfonic anhydride esterification followed by palladium-catalyzed coupling and trichlorosilane reduction. These ligands, particularly when complexed with ruthenium and chiral diamines, demonstrate exceptional performance in the asymmetric catalytic hydrogenation of prochiral ketones, achieving enantiomeric excess (e.e.) values as high as 99.5% alongside remarkable catalytic activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial application of asymmetric hydrogenation has relied heavily on established chiral bisphosphine ligands such as BINAP, DIOP, and DuPHOS, which possess C2 symmetry axes. While these ligands have facilitated the synthesis of critical drugs like Naproxen and Ibuprofen, they are not without significant operational drawbacks that impact manufacturing efficiency. A primary concern with many conventional ligands is their inherent instability under ambient conditions; they often exhibit poor stability towards air and moisture, necessitating the use of rigorously degassed solvents and strictly inert gas atmospheres throughout the reaction and handling processes. This requirement for specialized handling equipment and stringent environmental controls adds substantial complexity and cost to the manufacturing workflow. Furthermore, while effective, some traditional systems may struggle to maintain both high activity and high stereoselectivity simultaneously across a broad substrate scope, often requiring high catalyst loadings to achieve acceptable conversion rates, which is economically inefficient for large-scale production.
The Novel Approach
The spirocyclic bisphosphine ligand described in the patent represents a strategic evolution in ligand design, addressing the stability and efficiency gaps left by earlier generations. By incorporating a spiroindane structure, the ligand achieves a rigid conformation that enhances its thermal and chemical stability, thereby mitigating the need for extreme exclusion of air and moisture during certain processing stages. This structural rigidity also translates to superior stereocontrol, as evidenced by the ability to form matched bisphosphine-bisamine-ruthenium complexes that deliver outstanding enantioselectivity. The synthetic route outlined allows for the modular introduction of various substituents (R groups) on the phosphine aryl rings, enabling fine-tuning of the electronic and steric properties to optimize performance for specific substrates. This flexibility, combined with the inherent stability of the spiro-framework, offers a reliable pharmaceutical intermediate supplier with a versatile tool for tackling challenging asymmetric reductions that were previously cost-prohibitive or technically difficult to scale.
Mechanistic Insights into Palladium-Catalyzed Coupling and Reduction
The synthesis of these high-performance ligands relies on a sophisticated sequence of organometallic transformations that ensure high fidelity in the installation of the phosphine moieties. The process initiates with the activation of the spirocyclic diphenol precursor through esterification with trifluoromethanesulfonic anhydride in the presence of pyridine, generating a reactive bis-triflate intermediate. This activated species then undergoes a palladium-catalyzed cross-coupling reaction with diaryl phosphine oxides. Utilizing catalysts such as palladium acetate coordinated with ligands like dppb (1,4-bis(diphenylphosphino)butane) in dimethyl sulfoxide (DMSO) at elevated temperatures (50-150°C), the carbon-phosphorus bond is formed with high regioselectivity. This step is critical as it establishes the chiral environment around the phosphorus atoms, which is essential for subsequent asymmetric induction.
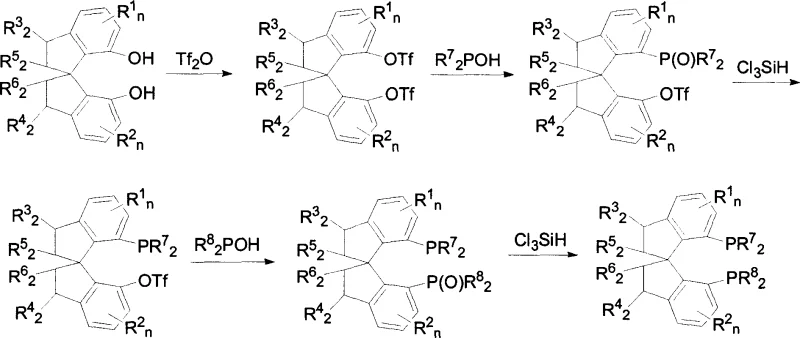
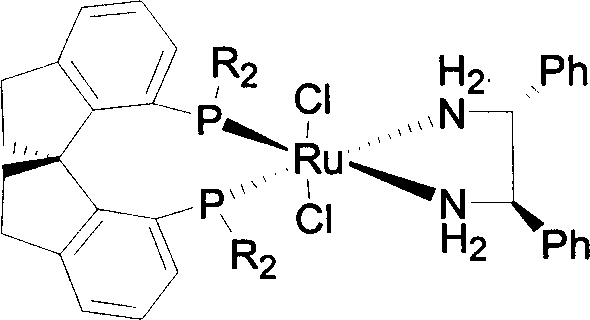
Following the coupling, the phosphine oxide intermediates must be reduced to the corresponding phosphines to activate the ligand for metal coordination. This is achieved using trichlorosilane (Cl3SiH) in the presence of a base like diisopropylethylamine in toluene under reflux conditions. The reduction step is chemically demanding but crucial, as it converts the stable oxide into the nucleophilic phosphine capable of coordinating with transition metals. The resulting ligand can then be complexed with ruthenium precursors, such as [(C6H6)RuCl2]2, and chiral diamines to form the active catalytic species. The rigidity of the spiro-backbone ensures that the chiral information is effectively transmitted from the ligand to the metal center, facilitating the highly stereoselective hydride transfer to the prochiral ketone substrate.
How to Synthesize Spirocyclic Bisphosphine Ligand Efficiently
The preparation of these ligands involves a multi-step sequence that requires precise control over reaction parameters to maximize yield and optical purity. The process begins with the activation of the chiral diol, followed by sequential phosphination and reduction cycles to install the phosphine groups. Each step utilizes standard organic synthesis techniques but demands careful attention to stoichiometry and temperature to prevent side reactions. For detailed operational protocols, including specific molar ratios, solvent volumes, and workup procedures derived directly from the patent examples, please refer to the standardized synthesis guide below.
- Esterify spirocyclic diphenol with trifluoromethanesulfonic anhydride in dichloromethane at 0-50°C to form bis-triflate.
- Perform palladium-catalyzed coupling with diaryl phosphine oxide in DMSO at 100°C using diisopropylethylamine as base.
- Reduce the phosphine oxide intermediate using trichlorosilane in toluene at reflux temperatures to yield the final phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this spirocyclic ligand technology offers compelling economic and logistical benefits that extend beyond mere technical performance. The primary driver for cost optimization lies in the exceptional catalytic efficiency of the resulting ruthenium complexes. With reported turnover numbers (S/C) reaching up to 100,000, the amount of expensive precious metal catalyst required per kilogram of product is drastically minimized. This reduction in catalyst loading directly translates to significant cost reduction in chiral ligand manufacturing, as the burden of purchasing and recovering high-value ruthenium and phosphine materials is substantially lowered. Furthermore, the high conversion rates observed reduce the need for extensive recycling loops or secondary processing to recover unreacted starting materials, streamlining the overall production timeline.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the high stability of the ligand precursors and intermediates. Unlike sensitive ligands that degrade upon exposure to ambient conditions, leading to batch failures and material waste, the spirocyclic framework offers robustness that simplifies storage and handling requirements. This durability reduces the need for specialized containment infrastructure and minimizes losses due to degradation, ensuring that the raw material investment yields maximum product output. Additionally, the use of relatively common reagents such as trichlorosilane and palladium acetate, combined with standard solvents like toluene and DMSO, ensures that the supply chain for inputs remains stable and cost-effective, avoiding reliance on exotic or single-source specialty chemicals.
- Enhanced Supply Chain Reliability: From a logistics perspective, the scalability of this synthesis route is a major advantage. The reaction conditions, such as refluxing in toluene or heating in DMSO, are readily adaptable to large-scale reactor systems without requiring cryogenic temperatures or ultra-high pressures that often bottleneck production capacity. This adaptability ensures that supply can be ramped up quickly to meet market demand without the need for capital-intensive equipment upgrades. The robustness of the chemistry also implies a lower risk of batch-to-batch variability, which is critical for maintaining consistent supply to downstream pharmaceutical manufacturers who require strict adherence to quality specifications for regulatory compliance.
- Scalability and Environmental Compliance: The environmental footprint of the manufacturing process is optimized through high atom economy and efficient workup procedures. The synthesis generates manageable byproducts, and the use of recyclable solvents like toluene aligns with green chemistry principles increasingly demanded by global regulatory bodies. The high selectivity of the catalyst reduces the formation of unwanted stereoisomers, thereby minimizing the waste associated with purification and separation processes. This efficiency not only lowers waste disposal costs but also facilitates easier compliance with environmental regulations, making the technology a sustainable choice for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spirocyclic ligand technology in industrial settings. These insights are derived from the specific experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the operational benefits and performance metrics associated with this advanced catalytic system.
Q: What are the stability advantages of this spirocyclic ligand compared to BINAP?
A: Unlike traditional ligands like BINAP which often require degassed solvents and inert atmospheres due to poor stability, this spirocyclic bisphosphine ligand exhibits superior stability, allowing for more convenient operation without stringent exclusion of air or moisture in many processing steps.
Q: What level of enantioselectivity can be achieved with this catalyst system?
A: When matched with appropriate chiral diamines to form a ruthenium complex, this ligand system achieves exceptionally high enantioselectivity, with e.e. values reaching up to 99.5% in the asymmetric hydrogenation of prochiral ketones.
Q: How does the turnover number (S/C) impact production costs?
A: The catalyst system demonstrates a high conversion number (S/C) of up to 100,000, which significantly reduces the required loading of expensive precious metal catalysts, thereby lowering the overall cost of goods for large-scale pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirocyclic Bisphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral ligands play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical purity. Our capability to handle complex organometallic syntheses allows us to provide high-purity spirocyclic bisphosphine ligands that meet the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be integrated into your specific production workflows. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is equipped with the most efficient and reliable catalytic solutions available in the market.
