Advanced Spirocyclic Phosphine-Oxazoline Ligands for High-Efficiency Asymmetric Hydrogenation
The landscape of asymmetric catalysis is continually evolving, driven by the relentless demand for higher purity and efficiency in the production of chiral pharmaceutical intermediates. Patent CN1884290A introduces a groundbreaking advancement in this field through the development of novel chiral spirocyclic phosphine-oxazoline ligands and their corresponding ionic iridium complexes. This technology addresses a critical bottleneck in organic synthesis: the asymmetric hydrogenation of imines, a reaction class that has historically struggled to achieve both high activity and high stereoselectivity simultaneously. By leveraging a unique spirodihydroindene backbone, these ligands offer a robust solution for generating chiral amines, which are ubiquitous structural motifs in active pharmaceutical ingredients (APIs) and agrochemicals. For R&D directors and process chemists, understanding the structural nuances of this innovation is key to unlocking more efficient synthetic pathways.
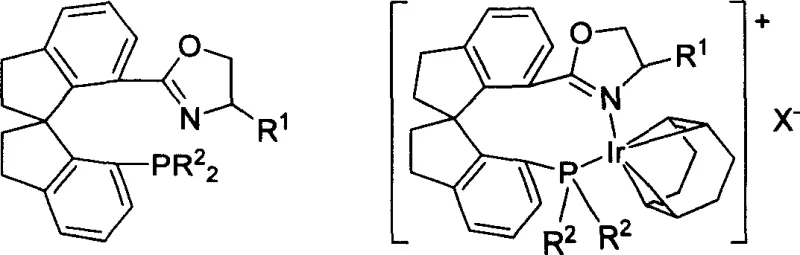
Traditional methods for asymmetric hydrogenation often rely on ligands that, while effective for olefins, falter when applied to the more challenging carbon-nitrogen double bonds found in imines. Conventional phosphine-oxazoline ligands, such as those developed by Pfaltz and Helmchen in the early 1990s, represented a significant step forward but were frequently limited by moderate enantioselectivity, often failing to exceed 90% ee in difficult substrates. Furthermore, many existing catalytic systems necessitate harsh reaction conditions, including high hydrogen pressures and elevated temperatures, which complicate reactor design and increase operational risks. The lack of rigidity in flexible ligand backbones can lead to multiple competing transition states, diluting the optical purity of the final product and necessitating costly downstream purification steps like recrystallization or chiral chromatography.
The novel approach detailed in the patent data fundamentally alters this paradigm by introducing a rigid spirocyclic framework that locks the ligand into a specific chiral conformation. This structural rigidity minimizes conformational freedom during the catalytic cycle, ensuring that the chiral information is transferred with maximum fidelity from the ligand to the substrate. The resulting ionic iridium complexes demonstrate exceptional performance, achieving enantiomeric excess (ee) values as high as 97% while maintaining high catalytic activity even under normal atmospheric pressure. This combination of high selectivity and mild operating conditions represents a substantial leap forward, offering a reliable pharma intermediate supplier pathway that drastically simplifies process development and reduces the environmental footprint of chiral synthesis.
Mechanistic Insights into Spirocyclic Ligand Coordination and Stereoselection
The superior performance of these catalysts stems from the intricate interplay between the spirodihydroindene backbone and the metal center. The ligand possesses two distinct chiral elements: axial chirality arising from the spiro junction and central chirality within the oxazoline ring. This dual-chirality system creates a highly defined chiral pocket around the iridium atom. When the precursor [Ir(COD)Cl]2 reacts with the ligand in the presence of a sodium salt (NaX), it forms a cationic iridium complex where the phosphine and the nitrogen of the oxazoline coordinate to the metal. The rigid spiro structure prevents the ligand from adopting non-productive conformations that typically lead to racemic background reactions. Instead, it forces the imine substrate to approach the metal hydride species from a single, energetically favored trajectory.
Furthermore, the electronic properties of the phosphine moiety can be finely tuned by varying the aryl substituents (R2 groups), ranging from phenyl to substituted phenyls with electron-donating or withdrawing groups. This tunability allows chemists to optimize the electron density at the iridium center, balancing the oxidative addition of hydrogen and the migratory insertion of the imine. The patent data highlights that the ionic nature of the complex, stabilized by non-coordinating anions like hexafluorophosphate or BARF, enhances solubility in organic solvents such as dichloromethane and prevents the formation of inactive dimeric species. This mechanistic robustness ensures consistent batch-to-batch reproducibility, a critical factor for commercial scale-up of complex pharmaceutical additives where impurity profiles must be strictly controlled.
How to Synthesize Spirocyclic Phosphine-Oxazoline Ligands Efficiently
The synthesis of these high-value ligands follows a logical, multi-step sequence starting from commercially available or easily prepared optically pure 1,1'-spirodihydroindene-7,7'-diphenol. The process begins with the activation of the phenolic hydroxyl groups via triflation, followed by a palladium-catalyzed cross-coupling to install the phosphine oxide functionality. Subsequent reduction converts the phosphine oxide to the active phosphine, which is then carried forward through carbonylation, hydrolysis, and amidation steps to construct the oxazoline ring. Each step has been optimized to maximize yield and preserve optical purity, ensuring that the final ligand is suitable for demanding catalytic applications. The detailed standardized synthesis steps are outlined in the guide below.
- Triflation of 1,1'-spirodihydroindene-7,7'-diphenol using trifluoromethanesulfonic anhydride and pyridine to form the bis-triflate intermediate.
- Palladium-catalyzed coupling with diaryl phosphine oxide followed by reduction with trichlorosilane to install the phosphine moiety.
- Carbonylation to introduce the ester group, followed by hydrolysis, amidation with chiral aminoethanol, and final cyclization to form the oxazoline ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere technical performance. The ability to conduct hydrogenation reactions at atmospheric pressure eliminates the need for specialized high-pressure autoclaves, significantly reducing capital expenditure (CAPEX) and maintenance costs associated with high-pressure equipment. This simplification of the hardware requirements also enhances plant safety by removing the risks associated with storing and handling large volumes of compressed hydrogen gas. Moreover, the high enantioselectivity achieved (up to 97% ee) means that the crude product requires minimal purification, leading to substantial cost savings in solvent usage, energy consumption for crystallization, and waste disposal. These factors collectively contribute to a more lean and agile manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal scavenging steps is a major driver for cost optimization. Because the catalyst exhibits high turnover numbers and the ligand structure is robust, the residual iridium levels in the final API can be kept low without aggressive purification protocols. Additionally, the use of common solvents like dichloromethane and methanol, rather than exotic fluorinated solvents, keeps raw material costs predictable and low. The streamlined synthesis of the ligand itself, utilizing standard palladium coupling chemistry, ensures that the catalyst precursor can be produced at a competitive price point compared to bespoke chiral ligands.
- Enhanced Supply Chain Reliability: The synthetic route relies on readily available starting materials such as spirodihydroindene derivatives and common phosphine oxides, reducing the risk of supply bottlenecks associated with rare natural products or complex custom syntheses. The robustness of the catalytic system against moisture and air (to a reasonable extent during handling) simplifies logistics and storage requirements. This reliability ensures continuous production schedules, preventing costly downtime that can occur when waiting for specialized reagents or when dealing with unstable catalyst batches that degrade during shipping.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-kilogram pilot runs without significant re-engineering. The reaction conditions are mild, typically operating between 0°C and 40°C, which reduces the energy load for heating or cooling large reactors. From an environmental perspective, the high atom economy of the hydrogenation reaction and the reduced need for chromatographic purification align with green chemistry principles, helping manufacturers meet increasingly stringent regulatory standards for waste generation and solvent emissions in fine chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spirocyclic ligand technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on performance expectations and operational parameters. Understanding these details is crucial for integrating this catalyst into existing production workflows.
Q: What represents the primary advantage of this spiro-ligand over conventional Ph-Ox ligands?
A: Unlike conventional phosphine-oxazoline ligands which often yield only moderate enantioselectivity, this novel spirocyclic backbone provides superior rigidity, achieving ee values up to 97% in imine hydrogenation.
Q: Does this catalytic system require high-pressure hydrogenation equipment?
A: No, a significant operational advantage is that the ionic iridium complexes derived from these ligands exhibit high reactivity, allowing hydrogenation to be completed effectively under normal atmospheric pressure.
Q: What types of substrates are compatible with this catalyst system?
A: The catalyst demonstrates broad substrate scope, effectively hydrogenating various N-aryl ketimines with different electronic properties, including those with electron-donating and electron-withdrawing groups on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirocyclic Phosphine-Oxazoline Ligand Supplier
As the global demand for chiral intermediates continues to surge, partnering with a technically proficient manufacturer is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the synthesis of complex ligands and catalysts with stringent purity specifications, ensuring that every batch meets the rigorous quality standards required by top-tier pharmaceutical companies. Our rigorous QC labs utilize advanced analytical techniques to verify enantiomeric excess and metal residue levels, guaranteeing the performance of our products in your critical synthesis steps.
We invite you to collaborate with us to evaluate the feasibility of this advanced hydrogenation technology in your specific process. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production volume and purity requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By leveraging our expertise in asymmetric catalysis, we can help you accelerate your development timelines and secure a stable, cost-effective supply of high-performance chiral building blocks for your next-generation therapeutics.
