Advanced Chiral Induction Strategy for Scalable Beta-Amino Acid Intermediate Production
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has long been hindered by the prohibitive costs and operational complexities associated with traditional asymmetric synthesis. Patent CN102212015A introduces a transformative methodology for preparing chiral beta-amino arylbutyric acid derivatives, specifically addressing the critical bottlenecks in producing key scaffolds for modern therapeutics. This technology leverages a sophisticated chiral auxiliary induction strategy that bypasses the need for expensive homogeneous transition metal catalysts, such as Ruthenium-BINAP complexes, which have historically dominated this chemical space. By shifting the stereocontrol mechanism to a robust organic induction pathway followed by heterogeneous hydrogenation, the process achieves exceptional optical purity exceeding 99.0% ee while drastically simplifying the operational workflow. For R&D directors and procurement strategists, this represents a pivotal shift towards more sustainable and economically viable manufacturing routes for complex amino acid derivatives.
The implications of this patent extend far beyond mere academic interest; it offers a tangible solution for the commercial scale-up of complex pharmaceutical intermediates. The method utilizes readily available starting materials, such as aryl acetoacetates and chiral amino alcohols, to construct the core carbon-nitrogen framework with precise stereochemical control. This approach not only mitigates the supply chain risks associated with scarce precious metals but also eliminates the stringent anaerobic conditions typically required for homogeneous asymmetric hydrogenation. Consequently, manufacturers can achieve consistent batch-to-batch reproducibility and significantly lower production costs, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships aiming to optimize their API supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino acids has relied heavily on homogeneous asymmetric hydrogenation, a technique fraught with significant industrial drawbacks. Traditional methods often employ costly chiral ligands coordinated with precious metals like Ruthenium or Rhodium, which not only inflate the raw material costs but also introduce severe challenges in downstream processing. The removal of trace metal residues to meet stringent pharmaceutical specifications requires additional purification steps, such as specialized scavenging or repeated recrystallization, which inevitably erodes overall yield and increases waste generation. Furthermore, these homogeneous catalytic systems are highly sensitive to oxygen and moisture, necessitating expensive reactor setups and rigorous inert atmosphere controls that complicate large-scale operations. Additionally, alternative chiral resolution methods, while effective in principle, suffer from a theoretical maximum yield of 50%, resulting in the wasteful discarding of the unwanted enantiomer and creating substantial environmental burdens.
The Novel Approach
In stark contrast, the methodology disclosed in CN102212015A employs a chiral amino alcohol-induced reduction strategy that fundamentally alters the economic and operational landscape of beta-amino acid synthesis. By condensing an aryl acetoacetate with a chiral amino alcohol, the process forms a cyclic intermediate that locks the stereochemistry through a rigid conformational constraint. This allows for highly diastereoselective reduction using inexpensive reagents like sodium cyanoborohydride, effectively transferring chirality without the need for exotic metal catalysts. The final step involves a straightforward heterogeneous hydrogenation to cleave the auxiliary, yielding the target molecule with outstanding optical purity. This route not only simplifies the reaction conditions to ambient pressure and temperature tolerances but also ensures that the final product is free from heavy metal contamination, streamlining the path to regulatory approval.
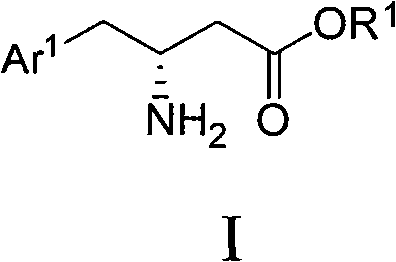
Mechanistic Insights into Chiral Amino Alcohol Induced Cyclization and Reduction
The core innovation of this technology lies in the formation and subsequent reduction of a seven-membered cyclic oxazepine intermediate, which serves as the template for stereochemical induction. When the aryl acetoacetate reacts with the chiral amino alcohol, it initially forms an enamine species that spontaneously cyclizes under basic conditions to create a rigid heterocyclic structure. This cyclization is crucial as it positions the prochiral double bond in a specific spatial orientation relative to the chiral center of the auxiliary. During the reduction phase, the hydride donor attacks the double bond from the less hindered face, dictated by the steric bulk of the phenyl group on the auxiliary. This facial selectivity ensures that the newly formed chiral center at the beta-position is established with high fidelity, resulting in a single dominant diastereomer.
Following the establishment of chirality, the removal of the auxiliary is achieved through catalytic hydrogenolysis, a process that is both chemoselective and efficient. The use of palladium hydroxide on carbon (Pd(OH)2/C) facilitates the cleavage of the benzyl-oxygen or benzyl-nitrogen bonds without affecting other sensitive functional groups on the aryl ring, such as fluorine substituents which are common in bioactive molecules. This step is particularly advantageous for R&D teams focused on impurity control, as heterogeneous catalysts are easily filtered off, preventing metal leaching into the final product. The mechanism ensures that the optical integrity established in the previous step is preserved, delivering a final product with an enantiomeric excess often surpassing 99%, thereby negating the need for further chiral purification steps that typically plague alternative synthetic routes.
How to Synthesize Chiral Beta-Amino Arylbutyric Acid Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable intermediates, emphasizing operational simplicity and high yield. The process begins with the condensation of the ketone substrate with the chiral amine, followed by cyclization and reduction to set the stereocenter, and concludes with hydrogenolytic cleavage. This sequence is designed to be scalable, utilizing common laboratory and plant equipment without the need for specialized high-pressure or cryogenic setups. The detailed标准化 synthesis steps见下方的指南 ensure that technical teams can replicate the high optical purity and yields reported in the patent examples, facilitating a smooth transition from bench-scale discovery to pilot plant production.
- Condense aryl acetoacetate with a chiral amino alcohol (e.g., S-phenylglycinol) to form an enamine intermediate.
- Cyclize the intermediate using a base to form a seven-membered cyclic oxazepine structure.
- Perform asymmetric induced reduction on the cyclic intermediate using sodium cyanoborohydride to set the chiral center.
- Execute catalytic hydrogenation using Pd(OH)2/C in methanol to remove the chiral auxiliary and yield the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chiral induction technology offers profound strategic benefits that directly impact the bottom line and operational resilience. By eliminating the dependency on homogeneous precious metal catalysts, manufacturers can insulate themselves from the volatile pricing of commodities like Ruthenium and Rhodium, leading to substantial cost savings in raw material procurement. The simplified reaction conditions reduce energy consumption and equipment maintenance costs, while the high yield and optical purity minimize waste disposal fees and maximize throughput. Furthermore, the use of stable, non-pyrophoric reagents enhances workplace safety and reduces the regulatory burden associated with handling hazardous materials, making the entire supply chain more robust and compliant with international safety standards.
- Cost Reduction in Manufacturing: The replacement of expensive homogeneous catalysts with affordable chiral amino alcohols and standard reducing agents significantly lowers the direct material cost per kilogram of the active intermediate. Since the process avoids the need for complex metal scavenging technologies and extensive recrystallization cycles to boost ee values, the overall processing time and utility consumption are drastically reduced. This efficiency translates into a more competitive pricing structure for the final API, allowing pharmaceutical companies to maintain healthy margins even in price-sensitive markets. The qualitative elimination of precious metal recovery steps further simplifies the cost model, removing a variable expense that often fluctuates with global metal markets.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium cyanoborohydride and palladium on carbon ensures a stable and diversified supply base, reducing the risk of production stoppages due to single-source supplier issues. Unlike specialized chiral ligands that may have long lead times and limited global availability, the reagents used in this process are widely produced and stocked by major chemical distributors. This accessibility guarantees continuous production schedules and shorter lead times for high-purity pharmaceutical intermediates, enabling drug developers to accelerate their clinical timelines without fear of material shortages. The robustness of the chemistry also means that technology transfer between different manufacturing sites is seamless, further securing the supply network.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily managed in large-scale reactors without the need for exotic engineering controls. The absence of toxic heavy metal residues in the final product simplifies environmental compliance and waste treatment, aligning with green chemistry principles and reducing the ecological footprint of the manufacturing process. High atom economy and the potential for solvent recycling contribute to a sustainable production model that meets the increasingly stringent environmental regulations imposed by global health authorities. This scalability ensures that the technology can support commercial volumes ranging from hundreds of kilograms to multi-ton annual production requirements effortlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology, derived directly from the patent's experimental data and claims. These insights are intended to clarify the feasibility and advantages of adopting this route for your specific project needs, ensuring that all stakeholders have a clear understanding of the process capabilities. Understanding these details is crucial for making informed decisions about process development and vendor selection.
Q: What optical purity can be achieved with this chiral induction method?
A: The patented method consistently achieves optical purity greater than 99.0% ee, eliminating the need for multiple recrystallizations often required in homogeneous catalysis.
Q: Does this process require expensive homogeneous metal catalysts?
A: No, the stereoselective step utilizes a chiral amino alcohol auxiliary and standard reducing agents like sodium cyanoborohydride, avoiding costly Ru or Rh complexes.
Q: Is the chiral auxiliary recoverable in this synthesis route?
A: The chiral auxiliary is removed during the final hydrogenation step. While the patent focuses on the forward synthesis, the use of robust auxiliaries often allows for potential recovery streams in optimized industrial processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Amino Arylbutyric Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of chiral intermediates for your drug development pipeline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral beta-amino arylbutyric acid derivatives meets the highest industry standards, including the >99.0% ee benchmark established by this advanced technology. Our commitment to excellence makes us a trusted partner for multinational pharmaceutical companies seeking to optimize their manufacturing strategies.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this technology offers compared to your current supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to move forward with confidence in your next clinical or commercial campaign. Let us help you transform complex chemical challenges into streamlined, cost-effective realities.
