Advanced Non-C2 Symmetric Ruthenium Catalysts for Commercial Scale Chiral Alcohol Production
The landscape of asymmetric synthesis has been fundamentally transformed by the innovations detailed in Chinese Patent CN1166672C, which introduces a sophisticated class of metal complexes featuring non-C2 symmetric axial chiral bisphosphine ligands. This technological breakthrough addresses critical challenges in the production of optically active alcohols, which serve as indispensable building blocks for a vast array of active pharmaceutical ingredients (APIs) and fine chemicals. Unlike traditional catalysts that rely on rigid C2 symmetry, this novel system leverages the unique steric and electronic properties of modified H4-BINAP ligands coordinated with divalent ruthenium and chiral diamines. For R&D directors and process chemists, this represents a significant leap forward in catalyst design, offering a versatile platform that can be finely tuned to achieve exceptional enantioselectivity across a broader range of challenging substrates. The patent outlines a robust methodology that not only enhances catalytic performance but also simplifies the synthetic route, making it an attractive option for industrial scale-up where consistency and purity are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of prochiral ketones has relied heavily on rhodium-based catalysts or early-generation ruthenium-BINAP systems, which, while effective, present distinct limitations in modern manufacturing contexts. Traditional rhodium catalysts, such as those derived from Wilkinson's catalyst, often suffer from high costs due to the precious metal content and can exhibit limited substrate scope, particularly when dealing with sterically hindered or electronically deactivated ketones. Furthermore, conventional C2 symmetric ligands, though successful in many cases, sometimes lack the necessary conformational flexibility to induce high enantioselectivity in complex molecular architectures, leading to suboptimal optical purity that requires costly downstream purification. These inefficiencies translate directly into higher production costs and longer lead times for pharmaceutical intermediates, creating bottlenecks in the supply chain for critical drug substances. Additionally, the sensitivity of some older catalyst systems to air and moisture necessitates stringent handling protocols, further complicating the operational workflow in large-scale reactors.
The Novel Approach
The innovative approach presented in this patent overcomes these historical barriers by employing a non-C2 symmetric ruthenium complex that combines the stability of ruthenium with the enhanced stereocontrol of tailored bisphosphine-diamine ligands. This new catalytic system demonstrates remarkable versatility, successfully hydrogenating a wide spectrum of ketones, including functionalized aryl alkyl ketones that were previously difficult to process with high fidelity. As illustrated in the reaction schemes below, the catalyst achieves near-quantitative conversion rates and excellent enantiomeric excess (ee) values, often exceeding 90% and reaching up to 98% in optimized conditions. 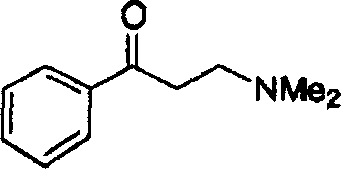 This level of performance significantly reduces the burden on chiral separation processes, thereby streamlining the overall manufacturing workflow. By shifting the focus to non-C2 symmetric ligands, the technology unlocks new possibilities for cost reduction in pharmaceutical intermediates manufacturing, allowing producers to access high-purity chiral alcohols more efficiently and reliably than ever before.
This level of performance significantly reduces the burden on chiral separation processes, thereby streamlining the overall manufacturing workflow. By shifting the focus to non-C2 symmetric ligands, the technology unlocks new possibilities for cost reduction in pharmaceutical intermediates manufacturing, allowing producers to access high-purity chiral alcohols more efficiently and reliably than ever before.
Mechanistic Insights into Ru-H4-BINAP-Diamine Catalytic Hydrogenation
The exceptional performance of this catalyst system can be attributed to its sophisticated mechanistic pathway, which operates through a metal-ligand bifunctional catalysis mechanism characteristic of Noyori-type hydrogenations. In this arrangement, the ruthenium center acts as the hydride source, while the NH moiety of the coordinated chiral diamine facilitates proton transfer, creating a concerted six-membered transition state that ensures precise stereochemical control. The non-C2 symmetric nature of the H4-BINAP derivative introduces a unique chiral environment around the metal center, allowing for finer discrimination between the enantiotopic faces of the ketone substrate compared to symmetric analogues. This structural asymmetry is crucial for accommodating diverse substituents on the ketone, as it prevents unfavorable steric clashes that might otherwise lower the reaction rate or selectivity. 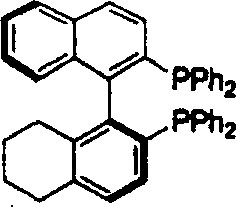 The synthesis of the active complex involves the careful coordination of the divalent ruthenium precursor with the bisphosphine ligand followed by the introduction of the diamine, resulting in a stable yet highly reactive species capable of operating under mild conditions.
The synthesis of the active complex involves the careful coordination of the divalent ruthenium precursor with the bisphosphine ligand followed by the introduction of the diamine, resulting in a stable yet highly reactive species capable of operating under mild conditions.
From an impurity control perspective, this mechanism offers significant advantages by minimizing side reactions such as over-reduction or racemization, which are common pitfalls in less selective catalytic systems. The robust nature of the ruthenium-diamine bond ensures that the catalyst remains intact throughout the reaction cycle, preventing the leaching of metal ions that could contaminate the final product. This is particularly vital for high-purity pharmaceutical intermediates where residual metal levels are strictly regulated. The ability to operate at relatively low temperatures and moderate hydrogen pressures further contributes to the stability of sensitive functional groups on the substrate, preserving the integrity of the molecule while achieving the desired transformation. Consequently, the resulting product profile is exceptionally clean, reducing the need for extensive chromatographic purification and enhancing the overall economic viability of the process.
How to Synthesize Non-C2 Symmetric Ruthenium Catalyst Efficiently
The preparation of this advanced catalyst is designed to be straightforward and scalable, utilizing readily available starting materials and standard laboratory techniques that can be easily adapted for industrial production. The process typically begins with the activation of a divalent ruthenium dimer, such as [RuCl2(C6H6)]2, in a polar organic solvent like DMF or toluene under an inert atmosphere to prevent oxidation. The H4-BINAP ligand is then introduced to form the initial phosphine-ruthenium complex, followed by the addition of the chiral diamine to complete the coordination sphere. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction vessel under inert atmosphere and combine divalent ruthenium precursor with the H4-BINAP ligand in a polar organic solvent.
- Heat the mixture to facilitate initial coordination, then cool to room temperature before introducing the chiral diamine component.
- Stir the final mixture to complete complexation, followed by solvent removal and purification via precipitation to isolate the active catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this non-C2 symmetric ruthenium catalyst technology offers compelling strategic benefits that extend beyond mere technical performance. The shift from expensive rhodium-based systems to robust ruthenium complexes inherently lowers the raw material cost basis, as ruthenium is generally more abundant and cost-effective while delivering superior longevity and turnover numbers. This transition supports significant cost reduction in fine chemical manufacturing by eliminating the need for excessive catalyst loading and reducing the frequency of catalyst replenishment in continuous processes. Furthermore, the simplified workup procedures described in the patent, which often involve simple filtration and solvent evaporation, drastically reduce processing time and energy consumption, contributing to a leaner and more agile production schedule.
- Cost Reduction in Manufacturing: The utilization of ruthenium instead of rhodium provides a direct material cost advantage, while the high catalytic activity ensures that minimal amounts of the precious metal are required per batch. This efficiency translates into substantial savings on precious metal recovery and waste management, as the lower metal load reduces the environmental burden and associated disposal costs. Additionally, the high selectivity of the catalyst minimizes the formation of byproducts, maximizing the yield of the desired chiral alcohol and reducing the loss of valuable starting materials. These factors combined create a highly economical process that improves the overall margin structure for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The synthetic route for the catalyst ligands and precursors relies on commodity chemicals and established synthetic pathways, ensuring a stable and secure supply chain that is less susceptible to geopolitical disruptions or market volatility. The robustness of the catalyst allows for longer storage life and easier transportation, reducing the risk of degradation during logistics. By implementing this technology, manufacturers can secure a consistent supply of high-quality chiral intermediates, reducing lead time for high-purity pharmaceutical intermediates and ensuring uninterrupted production flows for downstream API synthesis. This reliability is crucial for meeting the rigorous delivery schedules demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in batch reactors with straightforward scale-up parameters that do not require exotic equipment or extreme conditions. The use of common solvents and the absence of hazardous reagents simplify compliance with environmental, health, and safety (EHS) regulations, facilitating smoother regulatory approvals. The high atom economy of the hydrogenation reaction aligns with green chemistry principles, minimizing waste generation and supporting corporate sustainability goals. This environmental compatibility makes the technology an ideal choice for modern chemical facilities aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this non-C2 symmetric ruthenium catalyst technology, based on the specific data and embodiments provided in the patent documentation. These insights are intended to clarify the operational capabilities and strategic value of the system for potential partners and stakeholders evaluating this innovation for their own production pipelines.
Q: What are the primary advantages of this non-C2 symmetric catalyst over traditional BINAP systems?
A: The non-C2 symmetric architecture allows for greater structural tunability and often provides superior enantioselectivity for bulky or functionalized ketone substrates that traditional C2 symmetric ligands struggle to differentiate effectively.
Q: Is this catalyst system suitable for large-scale industrial hydrogenation?
A: Yes, the synthesis method described in the patent utilizes robust ruthenium precursors and standard solvents, making it highly scalable for commercial production while maintaining high conversion rates and optical purity.
Q: What types of ketone substrates are compatible with this technology?
A: The system demonstrates broad substrate scope, effectively hydrogenating various aryl alkyl ketones, including those with electron-withdrawing or electron-donating groups, as well as heteroaromatic and functionalized ketones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Non-C2 Symmetric Ruthenium Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in Patent CN1166672C and are fully equipped to bring this advanced catalytic solution to your production lines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle sensitive organometallic chemistry with the utmost precision, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee the highest quality standards for every batch of catalyst and intermediate we produce.
We invite you to collaborate with us to unlock the full commercial potential of this asymmetric hydrogenation technology for your specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing processes, demonstrating exactly how this novel catalyst can optimize your operations. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to jointly develop a supply strategy that meets your volume requirements and quality expectations.
