Advanced Zinc-Catalyzed Synthesis of Allyl Organophosphorus Compounds for Commercial Scale-Up
Advanced Zinc-Catalyzed Synthesis of Allyl Organophosphorus Compounds for Commercial Scale-Up
The chemical industry is constantly seeking more sustainable and cost-effective pathways for synthesizing high-value intermediates, and the technology disclosed in patent CN113201015B represents a significant leap forward in the preparation of allyl organophosphorus compounds. This innovative method utilizes readily available allyl alcohol derivatives and P-H compounds as starting materials, employing inexpensive zinc salts as catalysts under mild inert gas conditions. Unlike traditional methods that rely on precious transition metals, this zinc-catalyzed approach offers a greener, more economical alternative that maintains high atom economy while drastically simplifying the purification process. For R&D directors and procurement managers alike, this development signals a potential shift towards more robust supply chains for critical organophosphorus intermediates used in medicinal chemistry, agricultural science, and material synthesis. The ability to achieve yields as high as 97.1% under relatively mild thermal conditions underscores the practical viability of this route for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allyl organophosphorus compounds has been plagued by significant technical and economic hurdles associated with the use of noble metal catalysts. Conventional protocols often require transition metals such as ruthenium, cadmium, silver, or palladium to facilitate the coupling reactions between alkynols or MBH alcohols and phosphine sources. While these methods can achieve reasonable conversion, they introduce severe downstream processing challenges, primarily due to the difficulty of removing trace heavy metal residues to meet stringent pharmaceutical purity standards. Furthermore, catalysts like tris(pentafluorophenyl)borane, while metal-free, are prohibitively expensive and tend to form mixtures with the product that complicate isolation. The reliance on these scarce and costly reagents not only inflates the production cost but also creates supply chain vulnerabilities, making the consistent manufacturing of high-purity intermediates a persistent bottleneck for fine chemical producers.
The Novel Approach
The methodology outlined in CN113201015B fundamentally disrupts this status quo by replacing expensive transition metals with earth-abundant zinc salts, specifically zinc chloride or zinc acetate. This novel approach leverages the unique Lewis acidic properties of zinc to activate the reaction between allyl alcohol derivatives and P-H compounds without the need for harsh conditions or exotic ligands. By operating at moderate temperatures ranging from 20°C to 100°C, preferably between 50°C and 100°C, the process minimizes energy consumption and thermal degradation of sensitive functional groups. The compatibility of zinc salts with common organic solvents like acetonitrile, DMF, or toluene further enhances the flexibility of the process, allowing for easy adaptation to existing reactor infrastructure. Most critically, the incompatibility of the zinc catalyst with the final product system facilitates a much cleaner workup, reducing the number of purification steps required and thereby increasing the overall throughput and economic efficiency of the manufacturing line.
Mechanistic Insights into Zinc Salt-Catalyzed Allylation
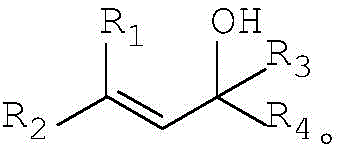
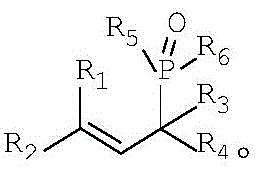
The core of this technological advancement lies in the efficient activation of the P-H bond and the subsequent nucleophilic attack on the allylic system, mediated by the zinc catalyst. In this mechanism, the zinc salt acts as a Lewis acid to coordinate with the hydroxyl group of the allyl alcohol derivative, thereby enhancing the leaving group ability of the hydroxyl moiety and generating a reactive allylic species in situ. Simultaneously, the zinc center may assist in deprotonating or activating the P-H compound, increasing its nucleophilicity towards the electrophilic allylic intermediate. This dual activation pathway ensures high regioselectivity and minimizes side reactions such as polymerization or elimination, which are common pitfalls in allylic substitutions. The broad substrate tolerance is evident in the structural variability allowed for the R groups, where R1 through R4 can encompass a wide range of alkyl, alkoxy, aryl, or aryloxy substituents, including halogenated variants.
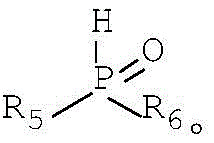
The versatility of the P-H component is equally impressive, accommodating diverse electronic and steric environments defined by the R5 and R6 groups. Whether utilizing diphenyl phosphine oxide or substituted variants like di-p-tolylphosphine oxide, the zinc catalyst maintains high activity, driving the reaction to completion with excellent yields. The structural integrity of the final allyl organophosphorus product is preserved, with the phosphorus moiety successfully installed at the allylic position without scrambling or isomerization. This mechanistic robustness is crucial for producing high-purity intermediates where even minor impurities can compromise downstream biological activity or material performance. The reaction proceeds cleanly to generate water as the primary byproduct alongside the target molecule, aligning with green chemistry principles by maximizing atom economy and minimizing waste generation.
Furthermore, the impurity profile of this zinc-catalyzed process is significantly cleaner compared to transition metal counterparts. Since zinc salts do not tend to form stable organometallic complexes that persist through workup, the risk of metal contamination in the final API or intermediate is drastically reduced. This is a critical advantage for pharmaceutical manufacturers who must adhere to strict ICH guidelines regarding elemental impurities. The ability to tune the reaction by adjusting the molar ratio of reactants—typically keeping the allyl alcohol derivative to P-H compound ratio between 1:1 and 1:2—allows for precise control over conversion and selectivity. With catalyst loadings as low as 0.01 to 0.1 equivalents, the process remains economically viable even for large-scale batches, ensuring that the cost of goods sold (COGS) remains competitive in the global market.
How to Synthesize Allyl Organophosphorus Compounds Efficiently
To implement this synthesis effectively, operators must adhere to specific procedural guidelines that maximize yield and safety while minimizing waste. The process begins with the careful selection of high-purity starting materials and the establishment of an inert atmosphere to prevent oxidation of the phosphorus species. Detailed standardized synthetic steps see the guide below for precise execution parameters regarding temperature ramps and quenching procedures.
- Under inert gas protection (nitrogen or helium), combine allyl alcohol derivatives, P-H compounds, and a zinc salt catalyst (such as zinc chloride) in an organic solvent like acetonitrile.
- Heat the reaction mixture to a temperature between 50°C and 100°C, preferably around 80°C, and maintain stirring for a duration of 1 to 10 hours to ensure complete conversion.
- Upon completion, cool the system to room temperature, concentrate under reduced pressure, and purify the crude product using column chromatography to isolate the high-purity allyl organophosphorus compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-catalyzed technology translates into tangible strategic benefits that extend far beyond simple reaction chemistry. The shift away from precious metals immediately alleviates pressure on sourcing budgets, as zinc salts are commodity chemicals with stable pricing and abundant global availability compared to volatile markets for ruthenium or palladium. This stability allows for more accurate long-term cost forecasting and reduces the risk of production stoppages due to catalyst shortages. Moreover, the simplified downstream processing means that manufacturing facilities can achieve higher throughput with existing equipment, effectively increasing capacity without requiring significant capital expenditure on new purification trains or metal scavenging units.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated heavy metal removal steps leads to a substantial decrease in overall production costs. By utilizing zinc chloride, a low-cost and widely available reagent, manufacturers can avoid the premium pricing associated with noble metals while simultaneously reducing the consumption of specialized scavenging resins or activated carbon typically required for metal cleanup. This streamlined workflow reduces solvent usage and labor hours dedicated to purification, resulting in a leaner, more cost-efficient manufacturing process that enhances profit margins for high-volume intermediates.
- Enhanced Supply Chain Reliability: Relying on earth-abundant zinc salts rather than scarce transition metals significantly de-risks the supply chain against geopolitical fluctuations and mining constraints. The raw materials for this process, including various allyl alcohol derivatives and phosphine oxides, are commercially accessible from multiple vendors, ensuring a continuous flow of inputs for production schedules. This diversification of the supply base prevents single-source bottlenecks and allows procurement teams to negotiate better terms, securing a steady supply of high-purity intermediates essential for maintaining uninterrupted pharmaceutical or agrochemical production lines.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common organic solvents make this process highly scalable from kilogram to multi-ton quantities without encountering significant engineering hurdles. The generation of water as a benign byproduct aligns with increasingly stringent environmental regulations, reducing the burden on wastewater treatment facilities and lowering the environmental footprint of the manufacturing site. This compliance advantage facilitates faster regulatory approvals and supports corporate sustainability goals, making the technology an attractive option for companies aiming to reduce their ecological impact while maintaining high production standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zinc-catalyzed synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industrial application.
Q: What are the primary advantages of using zinc salts over transition metal catalysts in this synthesis?
A: Using zinc salts, particularly zinc chloride, eliminates the need for expensive and toxic transition metals like ruthenium, palladium, or silver. This significantly reduces raw material costs and simplifies post-treatment by avoiding complex heavy metal removal processes, resulting in higher purity products suitable for pharmaceutical applications.
Q: What is the substrate scope for the allyl alcohol derivatives and P-H compounds in this method?
A: The method demonstrates broad versatility, accommodating allyl alcohol derivatives where R1-R4 can be hydrogen, alkyl, alkoxy, aryl, or aryloxy groups. Similarly, the P-H compounds allow for varied R5-R6 substituents including substituted or unsubstituted alkyl and aryl groups, enabling the synthesis of a diverse library of organophosphorus intermediates.
Q: What are the optimal reaction conditions for maximizing yield in this process?
A: Optimal results are achieved using a molar ratio of allyl alcohol derivative to P-H compound to zinc salt of approximately 1:1-2:0.01-0.05. The reaction is typically conducted in solvents like acetonitrile at temperatures between 50°C and 100°C for 5 to 10 hours, yielding products with efficiencies exceeding 90% in many examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Organophosphorus Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this zinc-catalyzed technology for the production of high-value fine chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of batch size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every shipment of allyl organophosphorus compounds meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging this innovative chemistry to deliver superior value to our global partners.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthesis method. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your project timelines and enhance your competitive edge in the market.
