Advanced Pd-Catalyzed Synthesis of Allyl Phosphine Derivatives for Commercial Scale-Up
The landscape of organophosphine chemistry is undergoing a significant transformation driven by the demand for greener, more efficient synthetic routes for high-value intermediates. Patent CN115490728A, published in late 2022, introduces a groundbreaking methodology for the synthesis of allyl phosphine derivatives that addresses long-standing inefficiencies in traditional manufacturing. This technology leverages a palladium-catalyzed coupling reaction between readily available alkenyl carbonates and diarylphosphine hydrogens, operating under remarkably mild conditions. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this represents a pivotal shift towards sustainable fine chemical intermediates production. The process not only streamlines the synthetic pathway by eliminating harsh reagents but also ensures high selectivity and yield, making it an ideal candidate for the commercial scale-up of complex organophosphine compounds. By utilizing carbon dioxide as the sole byproduct, this method aligns perfectly with modern environmental compliance standards while maintaining the rigorous purity specifications required for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allyl phosphine derivatives has been plagued by significant operational and economic drawbacks that hinder large-scale adoption. The first conventional approach, often referred to as the direct method, relies on the reaction of diarylphosphine hydrogens with allyl alkanes containing leaving groups such as halides or esters. As illustrated in the reaction scheme below, this pathway frequently suffers from low reaction yields, poor selectivity, and the generation of substantial stoichiometric waste salts.

Furthermore, the second conventional strategy involves an indirect route where oxidized or sulfurized phosphine hydrogens react with dienes or allenes, followed by a necessary reduction step to obtain the final product. This multi-step sequence inherently increases the cost of goods sold (COGS) due to additional reagent consumption and extended processing times. The deprotection or reduction phase not only adds complexity to the workflow but also exacerbates environmental concerns through increased three-waste discharge. These legacy methods struggle to meet the increasing demands for cost reduction in pharmaceutical intermediates manufacturing, particularly when dealing with sensitive functional groups that may degrade under the harsh conditions often required for these transformations.
The Novel Approach
In stark contrast to these outdated techniques, the novel methodology disclosed in the patent utilizes alkenyl carbonates as the electrophilic coupling partners. This strategic substitution of leaving groups allows the reaction to proceed via a decarboxylative mechanism that is both atom-economical and environmentally benign. The core reaction scheme, depicted below, highlights the direct coupling of the carbonate substrate with the phosphine source in the presence of a palladium catalyst and a base.
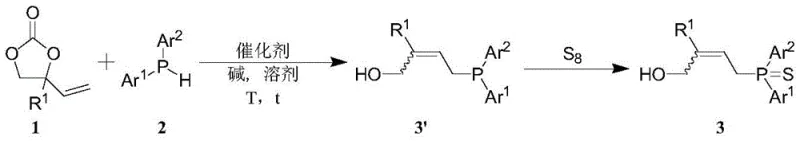
This innovative route operates effectively at room temperature (25°C), eliminating the need for energy-intensive heating protocols. The reaction exhibits exceptional functional group compatibility, tolerating a wide array of substituents including halogens, electron-withdrawing groups like trifluoromethyl, and electron-donating groups like methoxy. Following the coupling, a simple sulfurization treatment stabilizes the trivalent phosphorus intermediate into a robust phosphine sulfide derivative. This two-step one-pot potential significantly reduces the lead time for high-purity allyl phosphine derivatives, offering a streamlined alternative that enhances overall process efficiency and supply chain reliability for global chemical manufacturers.
Mechanistic Insights into Pd-Catalyzed Decarboxylative Allylation
The success of this synthesis hinges on the precise orchestration of a palladium-catalyzed cycle that facilitates the formation of the carbon-phosphorus bond. The mechanism likely initiates with the oxidative addition of the palladium catalyst to the alkenyl carbonate, followed by decarboxylation to generate a reactive pi-allyl palladium complex. This intermediate is then susceptible to nucleophilic attack by the diarylphosphine hydrogen, which acts as a soft nucleophile. The presence of a base, such as triethylamine, is crucial for deprotonating the phosphine species, thereby enhancing its nucleophilicity and driving the equilibrium towards product formation. The mild conditions prevent the isomerization of the double bond, ensuring high regioselectivity and preserving the structural integrity of the allyl moiety.
From an impurity control perspective, the use of alkenyl carbonates minimizes the formation of side products commonly associated with halide displacement reactions, such as homocoupling byproducts or elimination products. The subsequent sulfurization step serves a dual purpose: it stabilizes the air-sensitive P(III) species against oxidation during workup and provides a distinct handle for purification and characterization. The robustness of this catalytic system allows for the use of varied substrates without significant loss in efficiency, as evidenced by the consistent yields across different electronic environments on the aromatic rings. This mechanistic clarity provides R&D teams with the confidence to adapt the protocol for bespoke analogues, ensuring that high-purity organophosphine intermediates can be produced with minimal batch-to-batch variation.
How to Synthesize Allyl Phosphine Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of operation, requiring standard laboratory equipment and commercially available reagents. The process begins by charging a reaction vessel with the alkenyl carbonate substrate, the diarylphosphine hydrogen source, a palladium catalyst such as Pd(OAc)2, and an organic base in a solvent like tetrahydrofuran. The mixture is stirred at ambient temperature, allowing the coupling to reach completion within a few hours, monitored conveniently by TLC. Upon completion, elemental sulfur is introduced directly to the reaction mixture to effect sulfurization, followed by a standard aqueous workup and chromatographic purification.
- Combine alkenyl carbonate, diarylphosphine hydrogen, Pd(OAc)2 catalyst, triethylamine base, and THF solvent in a reaction vessel.
- Stir the mixture at room temperature (25°C) for approximately 5 hours to complete the coupling reaction.
- Add sulfur powder to the reaction mixture and stir for 20 minutes to stabilize the product via sulfurization before purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift from halide-based substrates to alkenyl carbonates represents a fundamental optimization of the raw material basket, leveraging cheaper and more abundant starting materials. This transition directly contributes to cost reduction in fine chemical intermediates manufacturing by removing the expense associated with pre-functionalized leaving groups and the disposal of halide waste streams. Furthermore, the ability to run the reaction at room temperature drastically lowers energy consumption, reducing the carbon footprint of the manufacturing process and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of the reduction step required in conventional indirect methods removes an entire unit operation from the production line. This simplification reduces labor costs, solvent usage, and reactor occupancy time. Additionally, the high atom economy of the decarboxylative coupling means less raw material is wasted, leading to substantial cost savings over large production volumes. The use of catalytic amounts of palladium, rather than stoichiometric reagents, further optimizes the cost structure, making the process economically viable for ton-scale production.
- Enhanced Supply Chain Reliability: Alkenyl carbonates and diarylphosphine hydrogens are widely available commodity chemicals, reducing the risk of supply bottlenecks associated with specialized reagents. The robustness of the reaction conditions ensures consistent output even with minor variations in raw material quality, enhancing supply continuity. By simplifying the synthetic route, the potential for process failures is minimized, ensuring that delivery schedules for critical pharmaceutical intermediates are met reliably. This stability is crucial for maintaining uninterrupted production lines in downstream drug manufacturing facilities.
- Scalability and Environmental Compliance: The generation of carbon dioxide as the primary byproduct simplifies waste management significantly compared to the saline waste generated by halide methods. This environmental advantage facilitates easier regulatory approval and reduces the cost of waste treatment infrastructure. The mild reaction conditions also enhance safety profiles, allowing for safer scale-up from kilogram to multi-ton batches. The process is inherently green, supporting the industry's move towards more sustainable chemical practices without compromising on yield or purity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities.
Q: What are the primary advantages of using alkenyl carbonates over traditional allyl halides in this synthesis?
A: Using alkenyl carbonates eliminates the need for pre-functionalized leaving groups like halides, reducing raw material costs and generating only carbon dioxide as a byproduct, which significantly simplifies waste treatment compared to halide salts.
Q: Why is a sulfurization step included in the final stage of the process?
A: The initial coupling product contains trivalent phosphorus which is air-sensitive; sulfurization converts it to a stable pentavalent phosphine sulfide derivative, facilitating easier handling, storage, and characterization without degradation.
Q: Can this method accommodate diverse functional groups on the aromatic rings?
A: Yes, the patent demonstrates excellent functional group tolerance, successfully synthesizing derivatives with fluoro, chloro, bromo, trifluoromethyl, methoxy, and thiomethyl substituents under mild room temperature conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Phosphine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in driving innovation within the pharmaceutical and agrochemical industries. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity allyl phosphine derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific requirements of palladium-catalyzed processes, including efficient metal scavenging and solvent recovery systems.
We invite potential partners to engage with our technical procurement team to discuss how this novel synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to optimize your production of complex organophosphine intermediates, ensuring quality, efficiency, and sustainability in every batch.
