Advanced Synthesis of Chloroethynyl Cyclopropanecarboxylic Acid for Commercial Scale Pyrethroid Production
Advanced Synthesis of Chloroethynyl Cyclopropanecarboxylic Acid for Commercial Scale Pyrethroid Production
The global demand for high-efficiency, low-toxicity pyrethroids continues to drive innovation in the synthesis of their critical building blocks. Among these, (±)-2,2-dimethyl-3-chloroethynyl cyclopropanecarboxylic acid stands out as a pivotal intermediate for next-generation insecticides and acaricides. A groundbreaking approach detailed in patent CN102060694B offers a transformative pathway for producing this valuable compound, shifting away from traditional ester-based routes to a more direct and economically viable acid-based methodology. This technical insight report analyzes the strategic implications of this synthesis for R&D directors seeking purity, procurement managers targeting cost optimization, and supply chain leaders ensuring continuity. By leveraging a robust dehydrohalogenation strategy, manufacturers can now access a reliable agrochemical intermediate supplier network that prioritizes both operational simplicity and environmental compliance.
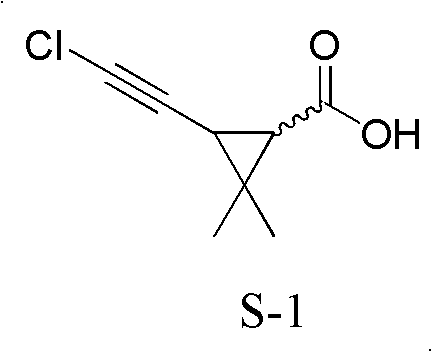
The structural integrity of the target molecule, as depicted above, relies on the precise installation of the chloroethynyl group onto the cyclopropane ring. Historically, achieving this transformation involved multi-step sequences that introduced unnecessary complexity and waste. The novel method described in the patent data circumvents these bottlenecks by utilizing (±)-2,2-dimethyl-3-(2,2-dichlorovinyl)cyclopropanecarboxylic acid, commonly known as permethric acid, as the direct starting material. This strategic choice eliminates the preliminary esterification and subsequent saponification steps that have long plagued conventional processes. For stakeholders in the fine chemical sector, this represents a significant leap forward in process intensification, allowing for higher throughput and reduced capital expenditure on reactor volume and processing time.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for this key pyrethroid intermediate have historically relied on the conversion of methyl or ethyl esters of permethric acid. As documented in earlier literature such as JP 52014748 and DE 2925570, these methods necessitate the use of strong alkoxides like sodium methoxide or potassium tert-butoxide to effect the elimination of hydrogen chloride. While chemically feasible, these protocols suffer from inherent inefficiencies that impact the bottom line. The requirement for stoichiometric amounts of expensive alkoxides drives up raw material costs significantly. Furthermore, the subsequent hydrolysis of the ester to the free acid adds an entire unit operation to the manufacturing schedule, increasing solvent consumption and energy usage. From a supply chain perspective, the reliance on moisture-sensitive alkoxides introduces logistical challenges regarding storage and handling, particularly in humid environments where reagent degradation can compromise batch consistency and yield.
The Novel Approach
In stark contrast, the methodology outlined in CN102060694B introduces a streamlined one-pot dehydrohalogenation directly on the carboxylic acid substrate. By employing inexpensive inorganic bases such as potassium hydroxide or sodium hydroxide in lower aliphatic alcohols, the process achieves the desired elimination without the need for prior ester protection. This shift not only reduces the molar equivalent cost of the base but also simplifies the reaction workup. The reaction proceeds smoothly under reflux conditions, typically between 80°C and 95°C, depending on the alcohol solvent chosen. The elimination of the ester hydrolysis step means that the final acidification yields the target product directly, bypassing the formation of intermediate salts that require separate cleavage. This direct route exemplifies the principles of green chemistry by minimizing step count and maximizing atom economy, providing a compelling value proposition for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Base-Catalyzed Dehydrohalogenation
The core chemical transformation involves the base-mediated elimination of hydrogen chloride from the 2,2-dichlorovinyl side chain to generate the chloroethynyl functionality. In this mechanism, the hydroxide ion acts as a potent base, abstracting a proton from the vinyl position while facilitating the departure of the chloride leaving group. The use of alcoholic solvents like ethanol or tert-butanol serves a dual purpose: they solubilize the organic acid substrate while also stabilizing the transition state through hydrogen bonding interactions. Unlike alkoxide bases which can induce transesterification side reactions if esters were present, the hydroxide/alcohol system is uniquely tuned for the free acid substrate. The reaction kinetics are favorable, with GC monitoring indicating complete conversion of the starting material within 4 to 12 hours. This robust kinetic profile ensures that the reaction can be driven to completion without excessive thermal stress that might degrade the sensitive cyclopropane ring.
Impurity control is another critical aspect where this mechanism excels. Traditional routes often struggle with over-elimination or polymerization of the acetylenic product under harsh basic conditions. However, the controlled reflux environment provided by the alcohol solvent acts as a thermal buffer, preventing localized hot spots that could trigger decomposition. Furthermore, the byproduct of the reaction is simply an inorganic salt (potassium chloride or sodium chloride), which precipitates out of the alcoholic solution upon cooling or can be easily filtered off. This physical separation of the byproduct prior to acidification ensures that the final organic phase is remarkably clean. The subsequent acidification step at low temperatures (-5°C to -15°C) further suppresses potential side reactions, promoting the crystallization of the pure acid. This mechanistic elegance translates directly into high-purity outputs, often exceeding 98% purity after a single recrystallization, meeting the stringent specifications required by pharmaceutical and agrochemical clients.
How to Synthesize (±)-2,2-Dimethyl-3-chloroethynyl Cyclopropanecarboxylic Acid Efficiently
Implementing this synthesis requires careful attention to reagent ratios and temperature control to maximize yield and safety. The process begins by preparing a homogeneous solution of the base in the alcohol solvent before introducing the substrate, ensuring immediate reaction onset upon addition. The molar ratio of base to substrate is optimized between 3.0:1 and 4.0:1 to drive the equilibrium towards the product while neutralizing the carboxylic acid proton. Following the reaction, the removal of solvent via reduced-pressure rotary evaporation concentrates the product salt, which is then subjected to acidic workup. The detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are critical for maintaining batch-to-batch reproducibility.
- Reflux the dichlorovinyl precursor with potassium or sodium hydroxide in a lower aliphatic alcohol solvent for 4 to 12 hours.
- Filter off the resulting salt byproduct and remove the solvent via reduced-pressure rotary evaporation to isolate the crude oil.
- Acidify the residue with dilute hydrochloric acid, cool to precipitate the solid, and purify via recrystallization in toluene and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The substitution of costly alkoxides with commodity chemicals like potassium hydroxide represents a fundamental shift in the cost structure of the intermediate. This change mitigates exposure to volatile pricing in the specialty reagent market and leverages the abundant global supply of inorganic bases. Additionally, the simplification of the process flow—removing the esterification and hydrolysis steps—drastically reduces the total cycle time per batch. This increased velocity allows manufacturing facilities to respond more agilely to market demand fluctuations, reducing lead time for high-purity agrochemical intermediates and improving overall asset utilization rates without requiring new capital investment.
- Cost Reduction in Manufacturing: The economic impact of replacing alkoxides with hydroxides cannot be overstated. Alkoxides are not only more expensive per kilogram but also require specialized packaging and inert atmosphere handling to prevent degradation. By switching to stable, shelf-stable hydroxides, the facility eliminates these overhead costs. Furthermore, the ability to recover and recycle the alcohol solvent through simple distillation adds another layer of savings. The elimination of column chromatography in favor of recrystallization significantly lowers the consumption of silica gel and elution solvents, which are major cost drivers in purification. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or retained as improved margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials. Permethric acid is a commodity intermediate produced at scale globally, ensuring a steady feedstock supply. The reagents—ethanol, KOH, HCl—are bulk chemicals with robust logistics networks, minimizing the risk of disruption due to single-source dependencies. The simplified workflow also reduces the number of quality control checkpoints required, accelerating the release of finished goods. This reliability is crucial for downstream formulators who depend on just-in-time delivery to maintain their own production schedules for final pesticide products.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is superior. Avoiding vacuum distillation of the final product reduces energy consumption and the risk of thermal runaway. The aqueous waste streams generated are primarily saline and can be treated using standard effluent protocols, unlike the complex organic waste associated with chromatographic purification. The process is inherently safer, operating at atmospheric pressure and moderate temperatures, which simplifies the engineering controls required for commercial scale-up of complex agrochemical intermediates. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chains. The following questions address common concerns regarding scalability, purity, and operational safety. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. By clarifying these points, we aim to bridge the gap between laboratory innovation and industrial application, ensuring that all parties have a clear understanding of the process capabilities.
Q: What is the primary advantage of using hydroxides over alkoxides in this synthesis?
A: Using sodium or potassium hydroxide significantly reduces raw material costs compared to expensive alkoxides like sodium methoxide, while simplifying storage and handling requirements for large-scale manufacturing.
Q: How does this method improve purification efficiency?
A: The process eliminates the need for energy-intensive vacuum distillation or complex column chromatography, relying instead on simple recrystallization to achieve high purity levels suitable for agrochemical applications.
Q: Can this route be scaled for industrial production?
A: Yes, the use of common solvents like ethanol and standard reflux conditions makes the process highly scalable, avoiding the safety hazards associated with handling large quantities of reactive alkoxides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloroethynyl Cyclopropanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and robust infrastructure. Our team has extensively analyzed the pathway described in CN102060694B and is fully equipped to execute this synthesis at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are designed to handle the specific thermal and mixing requirements of this dehydrohalogenation reaction, guaranteeing stringent purity specifications that exceed industry standards. With rigorous QC labs employing advanced analytical techniques, we validate every batch to ensure it meets the exacting demands of the global agrochemical market.
We invite you to collaborate with us to optimize your supply chain for pyrethroid intermediates. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this improved synthesis can impact your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
