Scalable Synthesis of TNNI3K Inhibitor Intermediates Using Water-Soluble Iridium Catalysts
Scalable Synthesis of TNNI3K Inhibitor Intermediates Using Water-Soluble Iridium Catalysts
The development of efficient synthetic routes for cardiac troponin kinase (TNNI3K) inhibitors represents a significant advancement in medicinal chemistry, particularly for treating ischemic injury and heart failure. As detailed in patent CN110857301B, a novel methodology has been established that leverages water-soluble bifunctional iridium complex catalysts to achieve high-efficiency N-methylation. This technological breakthrough addresses critical pain points in traditional pharmaceutical intermediate manufacturing, such as environmental toxicity and low atom economy. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this iridium-catalyzed pathway offers a compelling alternative to legacy processes. By utilizing methanol as both a reagent and solvent component in an aqueous system, the process aligns perfectly with modern green chemistry principles while maintaining the rigorous purity standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonamide-based kinase inhibitors has been plagued by the use of hazardous methylating agents and excessive organic solvents. Traditional protocols often require stoichiometric amounts of toxic reagents to introduce methyl groups onto the sulfonamide nitrogen, generating substantial chemical waste and complicating purification workflows. Furthermore, earlier reports indicate that existing methods suffer from poor atom economy, meaning a significant portion of the starting materials ends up as byproducts rather than the desired API intermediate. These inefficiencies not only drive up the cost of goods sold (COGS) but also impose heavy burdens on waste management systems, creating supply chain bottlenecks for manufacturers aiming to scale production. The reliance on harsh reaction conditions can also lead to the formation of difficult-to-remove impurities, jeopardizing the quality profile of the final drug substance.
The Novel Approach
In stark contrast, the innovative route disclosed in the patent utilizes a sophisticated iridium catalytic system to facilitate direct N-methylation using methanol. This approach transforms a typically wasteful process into a highly atom-economical transformation. The reaction proceeds in a mixed solvent system of methanol and water, drastically reducing the volume of volatile organic compounds (VOCs) released into the environment. As illustrated in the overall synthetic scheme below, the process flows smoothly from the starting bromobenzenesulfonamide to the final purine-substituted product through three distinct, high-yielding steps.
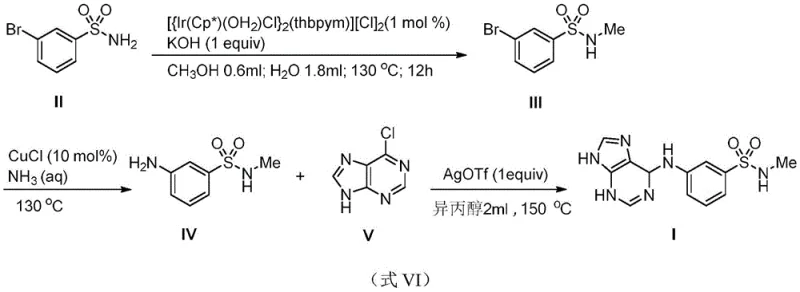
This streamlined workflow eliminates the need for pre-functionalized methyl donors and leverages the unique reactivity of the water-soluble catalyst to activate simple alcohols. For procurement managers focused on cost reduction in API manufacturing, this shift represents a move towards more sustainable and economically viable production models that minimize raw material waste and solvent disposal costs.
Mechanistic Insights into Iridium-Catalyzed N-Methylation
The cornerstone of this synthetic strategy is the employment of a specific water-soluble bifunctional iridium complex, structurally characterized as [{Ir(Cp*)(OH2)Cl}2(thbpym)][Cl]2. This catalyst operates through a borrowing hydrogen or hydrogen autotransfer mechanism, where the metal center temporarily dehydrogenates methanol to generate a reactive formaldehyde equivalent in situ. This electrophilic species then condenses with the sulfonamide nitrogen, followed by reduction via the borrowed hydrogen to yield the stable N-methyl product. The bifunctional nature of the ligand system enhances the catalyst's stability in aqueous media, allowing the reaction to tolerate the presence of water which is often detrimental to organometallic processes. This mechanistic elegance ensures that the reaction proceeds with high selectivity, minimizing over-alkylation or side reactions that could compromise the purity of the intermediate.
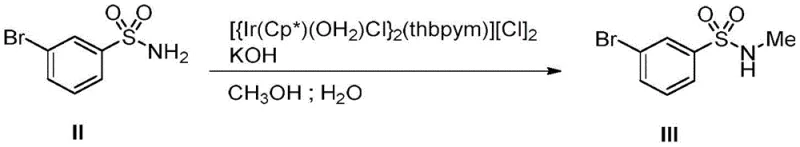
From an impurity control perspective, the mild basic conditions provided by potassium hydroxide (1 equiv) and the specific coordination environment of the iridium center prevent the degradation of the sensitive sulfonamide moiety. The reaction temperature of 130°C is sufficient to drive the kinetics without inducing thermal decomposition of the substrate. This precise control over the reaction parameters results in a clean crude profile, which significantly reduces the burden on downstream purification units like column chromatography or crystallization. For technical teams, understanding this mechanism highlights the robustness of the catalyst system, suggesting that it can be potentially recovered or optimized further for continuous flow applications, thereby enhancing the overall process mass intensity (PMI) of the manufacture.
How to Synthesize N-Methyl-3-(1H-purin-6-ylamino)benzenesulfonamide Efficiently
The execution of this synthesis requires careful attention to the sequential addition of reagents and the maintenance of specific thermal profiles to ensure optimal conversion. The process begins with the activation of the sulfonamide, followed by a copper-catalyzed amination, and concludes with a microwave-assisted coupling. Each step has been optimized to balance reaction rate with product integrity, ensuring that the final compound meets the stringent specifications required for biological testing and eventual therapeutic use. The following guide outlines the standardized operational parameters derived from the patent examples to assist process chemists in replicating this high-value transformation.
- Perform N-methylation of m-bromobenzenesulfonamide using a water-soluble bifunctional iridium complex catalyst (1 mol%) and KOH in a methanol/water mixture at 130°C for 12 hours.
- Convert the resulting N-methyl-3-bromobenzenesulfonamide to the amine derivative using aqueous ammonia and CuCl catalyst at 130°C.
- Couple the amine intermediate with 6-chloropurine using silver trifluoromethanesulfonate under microwave irradiation at 150°C to obtain the final TNNI3K inhibitor.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this iridium-catalyzed methodology offers profound strategic advantages for supply chain leaders and procurement officers managing the sourcing of complex heterocyclic intermediates. By shifting away from stoichiometric toxic reagents to a catalytic system, manufacturers can significantly reduce the regulatory burden associated with handling hazardous materials. This transition not only lowers compliance costs but also mitigates the risk of supply disruptions caused by stricter environmental regulations on chemical transport and storage. Furthermore, the use of water as a co-solvent simplifies the workup procedure, potentially reducing the consumption of extraction solvents like ethyl acetate and lowering the overall energy demand for solvent recovery.
- Cost Reduction in Manufacturing: The implementation of a catalytic cycle using inexpensive methanol as the carbon source fundamentally alters the cost structure of the synthesis. Unlike traditional methods that require expensive alkyl halides or sulfonates, this route utilizes a commodity chemical, leading to substantial raw material savings. Additionally, the high atom economy means less waste is generated per kilogram of product, which directly translates to lower waste disposal fees and reduced environmental levies. The elimination of complex protection-deprotection sequences further streamlines the operation, reducing labor hours and reactor occupancy time, which are critical drivers of manufacturing overhead.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as m-bromobenzenesulfonamide and 6-chloropurine, are commercially available bulk chemicals with stable supply chains. By avoiding exotic or highly regulated reagents, the risk of raw material shortages is minimized. The robustness of the iridium and copper catalysts ensures consistent batch-to-batch reproducibility, a key factor for maintaining steady production schedules. This reliability allows supply chain heads to forecast inventory needs more accurately and reduces the need for safety stock buffers, optimizing working capital utilization across the organization.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard heating and microwave equipment that is readily available in pilot and production plants. The aqueous nature of the initial methylation step aligns with global initiatives to reduce VOC emissions, making it easier to obtain environmental permits for new production lines. The simplified purification requirements, evidenced by the high yields reported in the patent examples, suggest that the process can be adapted for continuous manufacturing technologies, offering a pathway to even greater efficiency and reduced footprint in future commercial facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of the process capabilities and limitations for potential partners and licensees.
Q: What are the advantages of the water-soluble iridium catalyst in this synthesis?
A: The water-soluble bifunctional iridium complex allows the reaction to proceed in a mixed methanol/water solvent system, significantly reducing environmental pollution compared to traditional organic solvent-heavy methods. It offers high atom economy and operates under relatively mild conditions (130°C), facilitating easier downstream processing and waste treatment.
Q: How does this method improve upon previous synthetic routes for TNNI3K inhibitors?
A: Previous methods often relied on toxic chemicals and suffered from low atom economy. This novel approach utilizes methanol directly as a methylating agent in the presence of an efficient catalyst, eliminating the need for hazardous methylating reagents and simplifying the purification process through high-yield transformations.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process is designed with scalability in mind. The use of robust catalysts like the iridium complex and copper chloride, combined with standard heating and microwave techniques, allows for straightforward scale-up. The high yields reported in the examples (up to 90% for the amination step) indicate strong potential for cost-effective manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-3-(1H-purin-6-ylamino)benzenesulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic pathways in the development of next-generation cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this iridium-catalyzed route can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required by global regulatory bodies. Our commitment to green chemistry aligns perfectly with this patent's methodology, allowing us to offer sustainable manufacturing solutions that do not compromise on quality or delivery performance.
We invite R&D directors and procurement leaders to collaborate with us to evaluate the feasibility of this route for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic process. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is equipped with the most advanced and cost-effective chemical solutions available in the market.
