Revolutionizing Tetrahydroquinoxaline Production: A Safe, Water-Mediated Rhodium Catalytic Strategy for Commercial Scale-Up
Revolutionizing Tetrahydroquinoxaline Production: A Safe, Water-Mediated Rhodium Catalytic Strategy for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for safer, more sustainable, and cost-effective synthetic routes. A pivotal advancement in this domain is documented in Chinese Patent CN110483420B, which discloses a highly efficient preparation method for tetrahydroquinoxaline compounds. This technology represents a paradigm shift from traditional high-pressure hydrogenation or stoichiometric reduction methods to a sophisticated rhodium-catalyzed transfer hydrogenation protocol. By utilizing water as the ultimate hydrogen source in conjunction with zinc powder and a rhodium catalyst, this invention addresses critical pain points regarding safety, atom economy, and operational simplicity. For global procurement leaders and R&D directors seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this patented methodology is essential for securing supply chains that are both resilient and compliant with modern green chemistry standards.
The core innovation lies in the ability to reduce the aromatic quinoxaline ring system under remarkably mild conditions, typically ranging from 40°C to 80°C, without the need for specialized high-pressure reactors. This technical breakthrough not only lowers the barrier to entry for manufacturing these valuable scaffolds but also opens new avenues for synthesizing deuterated drugs by simply substituting regular water with deuterium oxide. As we delve deeper into the technical specifics, it becomes evident that this process offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring that production volumes can be increased from kilogram to multi-ton scales without compromising on safety or quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydroquinoxaline derivatives has relied heavily on catalytic hydrogenation using molecular hydrogen gas or chemical reductions employing hydride reagents. These conventional approaches are fraught with significant logistical and safety challenges that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. The use of high-pressure hydrogen gas necessitates expensive infrastructure, rigorous safety protocols, and specialized equipment capable of withstanding extreme pressures, which drastically increases capital expenditure and operational overhead. Furthermore, alternative methods utilizing formic acid as a hydrogen donor often generate substantial amounts of waste byproducts, leading to complex downstream purification processes and environmental compliance issues. The harsh reaction conditions associated with these traditional methods can also lead to poor selectivity, resulting in the formation of unwanted byproducts that compromise the purity of the final API intermediate.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110483420B introduces a transformative approach that leverages a rhodium-zinc-water system to achieve the desired reduction with exceptional efficiency. This novel route operates under atmospheric pressure or slight positive pressure of inert gas, completely eliminating the risks associated with high-pressure hydrogenation. The use of water as the hydrogen source is particularly groundbreaking, as it is the cheapest, safest, and most abundant proton donor available, fundamentally altering the economics of the synthesis. By combining this with inexpensive zinc powder as a sacrificial reductant and a highly active rhodium catalyst, the process achieves high conversion rates while maintaining a clean reaction profile. This shift not only simplifies the operational workflow but also aligns perfectly with the industry's move towards greener, more sustainable manufacturing practices, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates.

The general reaction scheme illustrates the versatility of this method, accommodating a wide range of substituents on the quinoxaline ring, including alkyl, alkoxy, and halogen groups. This broad substrate scope ensures that the technology can be applied to the synthesis of diverse libraries of bioactive molecules, providing a flexible platform for drug discovery and process development teams.
Mechanistic Insights into Rhodium-Catalyzed Transfer Hydrogenation
At the heart of this innovative synthesis is a sophisticated catalytic cycle mediated by rhodium complexes, such as [Rh(COD)Cl]2 or [Cp*RhCl2]2. The mechanism involves the activation of the rhodium center by the ligand and the subsequent interaction with zinc powder, which serves to regenerate the active low-valent rhodium species in situ. Water plays a dual role in this system: it acts as the proton source for the reduction of the nitrogen-containing heterocycle and participates in the hydrolysis steps necessary to release the product. The presence of additives like potassium hydroxide further facilitates the reaction by modulating the pH and enhancing the solubility of intermediates, ensuring a smooth progression of the catalytic cycle. Understanding these mechanistic details is crucial for R&D directors aiming to optimize reaction parameters for specific substrates, as slight modifications in ligand choice—such as switching from Bpy to BINAP—can significantly influence the stereoselectivity and yield of the transformation.
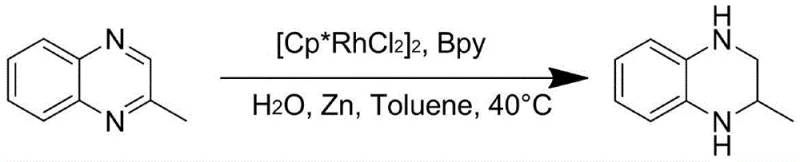
Impurity control is another critical aspect where this method excels. The mild thermal conditions (40-80°C) prevent the thermal degradation of sensitive functional groups that might otherwise decompose under the harsher conditions of traditional hydrogenation. Additionally, the high chemoselectivity of the rhodium catalyst ensures that only the quinoxaline ring is reduced, leaving other reducible groups on the molecule intact. This precision minimizes the formation of structural analogs and over-reduced byproducts, thereby simplifying the purification process and enhancing the overall purity of the final product. For manufacturers targeting high-purity pharmaceutical intermediates, this level of control is indispensable for meeting stringent regulatory requirements and ensuring the safety of the final drug product.
How to Synthesize Tetrahydroquinoxaline Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to standard laboratory and pilot plant setups. The procedure generally involves the preparation of a catalytic mixture under an inert atmosphere, followed by the sequential addition of the substrate and water. Reaction monitoring is easily achieved using thin-layer chromatography (TLC), allowing for real-time assessment of conversion. Once the reaction is complete, standard workup procedures involving filtration and column chromatography yield the target compound in high purity. For a comprehensive, step-by-step breakdown of the standardized synthesis protocol, please refer to the detailed guide below.
- Prepare the catalytic system by dissolving rhodium metal catalyst (e.g., [Rh(COD)Cl]2), zinc powder, and an additive (e.g., KOH) or ligand in a dry solvent like toluene under an inert nitrogen or argon atmosphere.
- Add the quinoxaline-based reaction substrate to the mixture, followed by the addition of water, ensuring the molar ratio of substrate to water is maintained between 1: 10 and 1:30.
- Heat the reaction mixture to 40-80°C and monitor progress via TLC until the substrate is fully consumed, then purify the residue using silica gel column chromatography to isolate the target tetrahydroquinoxaline.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this rhodium-catalyzed water-mediated reduction strategy offers profound benefits for procurement managers and supply chain heads. The primary advantage lies in the drastic simplification of the supply chain logistics; by replacing hazardous hydrogen gas and expensive hydride reagents with benign water and zinc powder, companies can significantly reduce their dependency on specialized gas suppliers and mitigate the risks associated with transporting dangerous chemicals. This shift not only enhances the reliability of the supply chain but also reduces the administrative burden related to safety compliance and hazardous waste management. Furthermore, the use of earth-abundant zinc and water as key reagents ensures that raw material costs remain stable and predictable, shielding the manufacturing process from the volatility often seen in the pricing of specialty chemical reagents.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the substitution of expensive reducing agents with low-cost zinc and water lead to substantial operational savings. The simplified downstream processing, driven by the high selectivity of the reaction, further reduces the consumption of solvents and silica gel during purification, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: By utilizing reagents that are globally available and non-hazardous, the risk of supply disruptions due to regulatory restrictions or transportation incidents is minimized. This robustness ensures a continuous flow of materials, allowing manufacturers to maintain consistent production schedules and meet delivery deadlines with greater confidence.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic byproducts make this process inherently scalable and environmentally friendly. The reduced generation of chemical waste aligns with increasingly strict environmental regulations, facilitating easier permitting and long-term sustainability goals for large-scale manufacturing facilities.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial viability of this synthesis method, we have compiled a set of frequently asked questions based on the patent data. These answers provide clarity on the operational parameters, safety profiles, and scalability of the technology, helping stakeholders make informed decisions about integrating this process into their manufacturing portfolios.
Q: What are the primary advantages of using water as a hydrogen source in this synthesis?
A: Using water as a hydrogen source eliminates the need for high-pressure hydrogen gas cylinders or hazardous formic acid, significantly improving operational safety and reducing waste disposal costs associated with traditional reduction methods.
Q: How does this rhodium-catalyzed method compare to conventional hydrogenation in terms of selectivity?
A: The rhodium-catalyzed transfer hydrogenation operates under mild temperatures (40-80°C), which minimizes side reactions and over-reduction often seen in harsh catalytic hydrogenation, thereby ensuring higher purity and better control over the impurity profile.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes cheap and readily available reagents like zinc powder and water, and avoids extreme pressures, making it highly scalable and economically viable for the commercial production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoxaline Supplier
As the demand for high-quality heterocyclic intermediates continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is crucial for success. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the lab bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of tetrahydroquinoxaline meets the highest industry standards. Whether you require custom synthesis or large-volume supply, our team is dedicated to delivering excellence and reliability.
We invite you to leverage our technical expertise to optimize your supply chain and reduce costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your drug development goals and ensure a secure, high-quality supply of critical intermediates.
