Strategic Process Optimization for Quinoline-Based Angiogenesis Inhibitor Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic compounds, particularly those serving as potent angiogenesis inhibitors in oncology therapy. Patent CN102356063A discloses a groundbreaking method for preparing 6-(7-((1-aminocyclopropyl)methoxy)-6-methoxyquinolin-4-yloxy)-N-methyl-1-naphthamide, designated as Formula (I), and its pharmaceutically acceptable salts. This specific quinoline derivative represents a critical structural motif in modern cancer treatment protocols, necessitating a manufacturing process that guarantees both high chemical purity and operational safety. The disclosed innovation addresses significant limitations found in prior art by introducing a streamlined sequence that avoids hazardous intermediates while maintaining exceptional yield profiles. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented methodologies is essential for ensuring the consistent delivery of high-quality active pharmaceutical ingredients (APIs) to the global market. The following analysis delves into the technical superiority of this route, highlighting its potential to redefine cost structures and supply chain reliability for complex quinoline-based therapeutics.
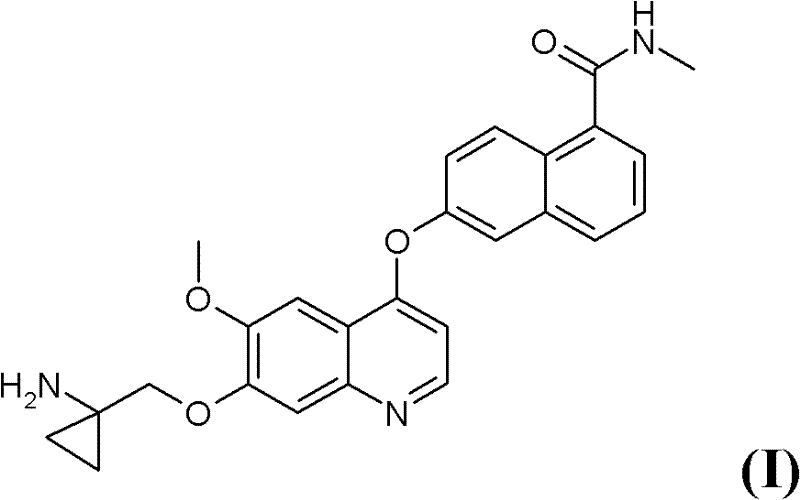
The development of this synthesis protocol was driven by the urgent need to overcome the inherent dangers associated with traditional methods for constructing the cyclopropyl-aminomethoxy side chain on the quinoline scaffold. Previous approaches, such as those referenced in WO 2008/112408 A1, relied heavily on the formation of acyl azide intermediates followed by a Curtius rearrangement to introduce the necessary nitrogen functionality. This classical transformation, while chemically effective on a small laboratory scale, presents unacceptable risks for industrial manufacturing due to the explosive nature of acyl azides and the stringent safety controls required for their handling. Furthermore, alternative alkylation strategies involving cycloalkyl-alkyl mesylates often suffer from harsh reaction conditions that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to purge. The limitations of these conventional methods create bottlenecks in production capacity and inflate the cost of goods sold due to specialized safety infrastructure and extensive purification requirements.
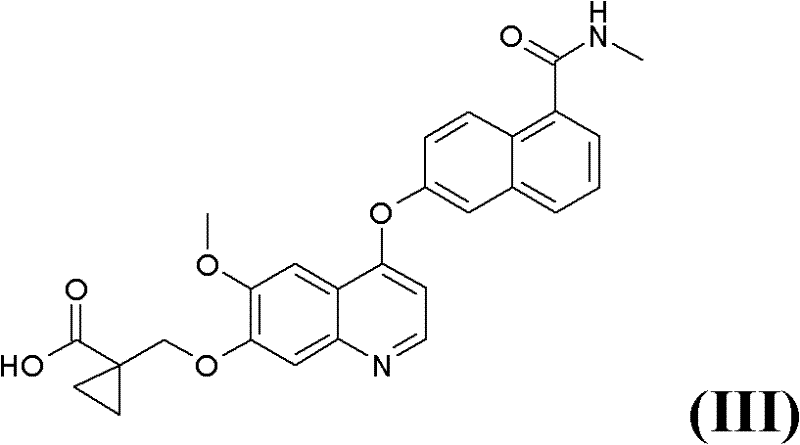
In stark contrast, the novel approach detailed in the patent utilizes a Mitsunobu reaction to establish the critical ether linkage early in the synthetic sequence, thereby circumventing the need for dangerous azide chemistry entirely. This strategic shift allows for the introduction of the protected 1-amino-1-cyclopropylmethyl moiety under mild, neutral conditions that preserve the integrity of the acetophenone precursor. By reacting a protected aminocyclopropanemethanol of Formula (VI) with 4-hydroxy-3-methoxyacetophenone (VII), the process generates the key intermediate (VIII) with high regioselectivity and minimal byproduct formation. This methodological pivot not only enhances the safety profile of the manufacturing plant but also simplifies the downstream processing steps, as the resulting intermediates are easier to isolate and purify. The elimination of the Curtius rearrangement step removes a major regulatory hurdle, facilitating smoother technology transfer and faster approval timelines for commercial production facilities aiming to become a reliable agrochemical intermediate supplier or pharma partner.
Mechanistically, the success of this synthesis hinges on the precise execution of the Mitsunobu etherification and the subsequent modified Leimgruber-Batcho reductive cyclization. The initial coupling between the alcohol (VI) and the phenol (VII) proceeds via the formation of an alkoxyphosphonium salt, which is then displaced by the phenoxide anion to form the ether bond with inversion of configuration, although stereochemistry is less critical for this specific achiral cyclopropyl system. The reaction is typically conducted in tetrahydrofuran at low temperatures, utilizing triphenylphosphine and diisopropyl azodicarboxylate (DIAD) to drive the equilibrium towards product formation. Following this, the nitration of the aromatic ring and subsequent condensation with N,N-dimethylformamide dimethyl acetal generate the beta-enaminoketone intermediate (X), which serves as the precursor for ring closure. The cyclization step employs iron powder in acetic acid, a cost-effective and environmentally friendlier reducing agent compared to catalytic hydrogenation, to reduce the nitro group and simultaneously close the quinoline ring to form the 4-hydroxyquinoline derivative (XI).
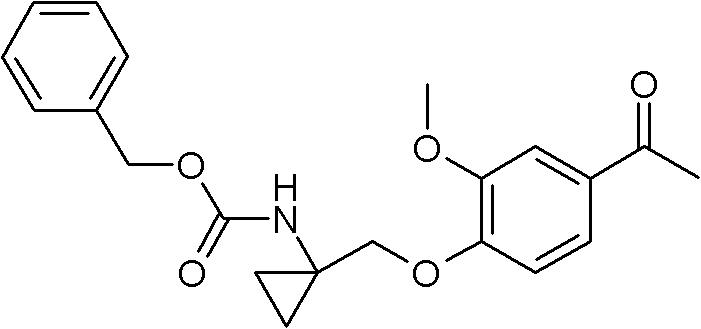
The control of impurities throughout this multi-step sequence is paramount for meeting the stringent quality standards required for oncology drugs. The use of iron powder for reduction, rather than precious metal catalysts, eliminates the risk of heavy metal contamination, which is a persistent challenge in API manufacturing that often requires expensive scavenging resins to resolve. Additionally, the stepwise construction of the molecule allows for intermediate isolation and purification, ensuring that impurities do not carry through to the final stages. For instance, the chlorination of the 4-hydroxyquinoline (XI) to the 4-chloroquinoline (XII) using phosphorus oxychloride is a well-understood transformation that proceeds with high conversion, minimizing the presence of unreacted starting material in the subsequent coupling step. This rigorous attention to chemical purity at every stage ensures that the final deprotection yields the target compound (I) with a purity exceeding 98%, as confirmed by LC-MS analysis in the provided examples, thereby satisfying the rigorous specifications demanded by regulatory bodies for clinical and commercial use.
How to Synthesize 6-(7-((1-aminocyclopropyl)methoxy)-6-methoxyquinolin-4-yloxy)-N-methyl-1-naphthamide Efficiently
The synthesis of this complex quinoline derivative requires a disciplined approach to reaction conditions and reagent stoichiometry to maximize yield and minimize waste. The process begins with the preparation of the ether-linked acetophenone intermediate via Mitsunobu coupling, followed by nitration and enaminone formation to set up the quinoline ring closure. Detailed standardized synthesis steps for this pathway are provided in the guide below, outlining the specific temperatures, solvents, and workup procedures necessary to replicate the high yields reported in the patent literature. Adhering to these protocols ensures reproducibility and safety, particularly when scaling from gram-scale laboratory experiments to kilogram or ton-scale commercial production runs.
- Perform Mitsunobu reaction between protected aminocyclopropanemethanol and 4-hydroxy-3-methoxyacetophenone to form the ether linkage.
- Execute nitration followed by enaminone formation using DMF-DMA to prepare the cyclization precursor.
- Conduct reductive cyclization using iron powder in acetic acid to construct the quinoline core, followed by chlorination and coupling.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers substantial strategic advantages over legacy methods, primarily driven by enhanced safety and operational efficiency. The elimination of explosive acyl azide intermediates drastically reduces the insurance premiums and specialized infrastructure costs associated with manufacturing hazardous chemicals, leading to significant cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the robustness of the Mitsunobu and reductive cyclization steps ensures consistent batch-to-batch quality, reducing the incidence of failed batches and the associated financial losses. This reliability is crucial for maintaining a steady supply of critical oncology intermediates, preventing disruptions that could delay clinical trials or commercial drug launches. By optimizing the synthetic pathway, manufacturers can achieve higher throughput with existing equipment, effectively increasing capacity without requiring massive capital expenditure on new reactors or safety systems.
- Cost Reduction in Manufacturing: The replacement of hazardous Curtius rearrangement chemistry with safer Mitsunobu coupling and iron-mediated reduction eliminates the need for expensive safety containment systems and specialized waste disposal protocols for azides. This shift inherently lowers the operational overhead and reduces the consumption of costly reagents, as the new pathway utilizes more economical starting materials and catalysts. Additionally, the high purity of the intermediates reduces the burden on downstream purification processes, saving on chromatography media and solvent consumption, which collectively contributes to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents, such as protected aminocyclopropanemethanols and common nitrating agents, ensures that the supply chain is not vulnerable to the shortages often associated with specialized or hazardous custom synthons. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, leading to more predictable lead times. This stability allows procurement managers to negotiate better terms with suppliers and maintain optimal inventory levels, securing the continuity of supply for high-purity pharmaceutical intermediates essential for global drug distribution networks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are easily transferred from pilot plants to large-scale commercial reactors. The avoidance of heavy metal catalysts in the reduction step aligns with increasingly strict environmental regulations regarding metal residues in APIs, simplifying the regulatory filing process. Moreover, the improved atom economy and reduced generation of hazardous waste streams support sustainability goals, making the manufacturing process more attractive to environmentally conscious stakeholders and reducing the long-term liability associated with chemical waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this quinoline derivative, based on the detailed disclosures within the patent documentation. These insights are intended to clarify the practical implications of the new methodology for stakeholders involved in process development and sourcing. Understanding these aspects is vital for making informed decisions about technology adoption and partnership opportunities in the competitive landscape of fine chemical manufacturing.
Q: Why is the new synthesis method safer than previous routes?
A: The novel process described in CN102356063A eliminates the use of hazardous acyl azide intermediates and the potentially explosive Curtius rearrangement, significantly reducing safety risks during large-scale production.
Q: What represents the key improvement in purity for this intermediate?
A: By utilizing mild Mitsunobu conditions for ether formation and efficient reductive cyclization, the process minimizes side reactions, resulting in intermediates with purity exceeding 95% and a final API purity greater than 98%.
Q: Is this process suitable for commercial scale-up?
A: Yes, the avoidance of explosive azides and the use of robust reagents like iron powder for reduction make this pathway highly amenable to commercial scale-up, ensuring supply chain continuity and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-(7-((1-aminocyclopropyl)methoxy)-6-methoxyquinolin-4-yloxy)-N-methyl-1-naphthamide Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN102356063A are executed with precision and efficiency. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for Mitsunobu couplings and reductive cyclizations, maintaining stringent purity specifications through our rigorous QC labs. We understand the critical nature of oncology intermediates and are committed to delivering materials that meet the highest international standards, providing our partners with the confidence needed to advance their drug development programs without supply chain interruptions.
We invite you to contact our technical procurement team to discuss your specific requirements for this quinoline intermediate. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall project costs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to support your long-term supply needs, ensuring a seamless integration of our high-quality intermediates into your production workflow.
