Advanced Metal-Free Synthesis of p-Aryl Azophenol Derivatives for Commercial Scale-Up
Introduction to Novel p-Aryl Azophenol Synthesis Technology
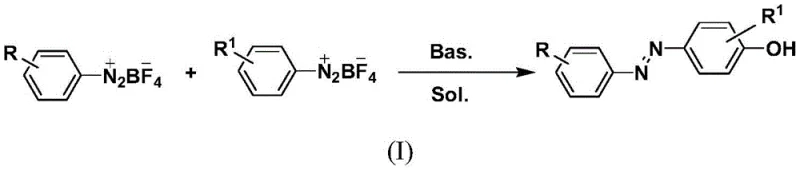
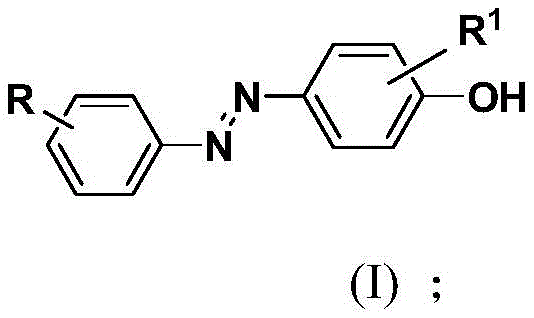
The landscape of organic synthesis for functional aromatic compounds is undergoing a significant transformation driven by the demand for greener, more efficient manufacturing processes. Patent CN113105357B introduces a groundbreaking methodology for the synthesis of p-aryl azophenol derivatives, utilizing a dual aryl diazonium salt strategy that operates under remarkably mild conditions. This technology represents a pivotal shift away from traditional heavy metal catalysis and harsh reaction environments, offering a robust pathway for producing high-value intermediates used in pharmaceuticals, dyes, and smart materials. By leveraging a one-pot tandem reaction in water-mixed solvents, this approach not only simplifies the operational workflow but also aligns with modern sustainability goals essential for today's chemical industry. For R&D directors and procurement specialists, understanding the nuances of this patent is critical for securing a reliable supply chain of high-purity azophenol derivatives.
The core innovation lies in the bifunctionalization of aryl diazonium salts with water, facilitated by specific additives such as sodium acetate or organic bases. Unlike conventional methods that often struggle with explosive hazards associated with diazonium species or require stringent temperature controls, this disclosed method maintains stability at room temperature. The versatility of the substrate scope allows for various substituents including alkyl, alkoxy, halogen, and nitro groups, making it adaptable for diverse molecular architectures. As we delve deeper into the technical specifics, it becomes evident that this synthesis route offers substantial advantages in terms of cost reduction in pharmaceutical intermediates manufacturing and enhanced process safety. The ability to generate complex multifunctionalized scaffolds efficiently positions this technology as a key enabler for next-generation material science and drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-substituted azophenol compounds has relied on established protocols such as the Mills condensation, the Wallach reaction, or traditional diazotization coupling, each carrying significant operational burdens. The Mills reaction typically necessitates glacial acetic acid as a solvent and involves the condensation of p-nitrosophenol with aniline, often requiring excessive oxidizing agents that generate substantial volumes of toxic waste. Similarly, the Wallach reaction is a multi-step process involving the reduction of nitrobenzene to azoxybenzene followed by acidic rearrangement, which complicates the workflow and lowers overall atom economy. Perhaps most critically, traditional diazotization coupling reactions involve the in situ generation of diazonium salts, which are notoriously unstable and potentially explosive, demanding rigorous temperature control and specialized safety infrastructure. These legacy methods frequently result in low yields, difficult purification processes, and environmental compliance challenges that drive up the total cost of ownership for manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113105357B utilizes pre-formed aryl diazonium tetrafluoroborate salts as stable starting materials, reacting them directly in a mixed solvent system of water and organic co-solvents like methanol or ethanol. This approach eliminates the need for transition metal catalysts, thereby removing the costly and time-consuming steps associated with heavy metal scavenging and residual analysis. The reaction proceeds smoothly at temperatures ranging from 0°C to 40°C, preferably at room temperature, which drastically reduces energy consumption and thermal hazards. By employing additives such as sodium acetate or triethylamine, the system facilitates a controlled hydrolysis and coupling sequence that minimizes side reactions and maximizes product purity. This streamlined process not only enhances the safety profile of the manufacturing operation but also significantly simplifies the downstream processing, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Metal-Free Diazonium Coupling
The mechanistic pathway of this novel synthesis is elegantly simple yet highly effective, relying on the intrinsic reactivity of the diazonium cation in the presence of a mild base and water. Initially, the aryl diazonium salt undergoes a base-promoted decomposition where the nitrogen molecule is expelled, generating a highly reactive aryl carbocation intermediate. This electrophilic species is immediately trapped by water molecules present in the solvent mixture, leading to the formation of a phenol intermediate through a hydrolysis step. Subsequently, under the basic conditions provided by additives like sodium acetate, the newly formed phenol is deprotonated to yield a nucleophilic phenoxide anion. This anion then attacks a second equivalent of the aryl diazonium salt in an electrophilic aromatic substitution-like manner, forming the characteristic azo bond at the para-position relative to the hydroxyl group. This cascade sequence ensures high regioselectivity and avoids the random coupling often seen in less controlled environments.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining a clean product profile. Traditional strong acid or base conditions can lead to over-oxidation, polymerization, or decomposition of sensitive functional groups, but this buffered system preserves the integrity of substituents such as esters, nitriles, and halogens. The use of stable tetrafluoroborate salts further mitigates the risk of uncontrolled exotherms that could degrade product quality. By avoiding transition metals, the process inherently prevents metal-induced side reactions and eliminates the risk of metal contamination in the final API or intermediate. This level of control is essential for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical applications. The mechanistic clarity allows process chemists to fine-tune parameters such as additive stoichiometry and solvent ratios to optimize yields, which have been demonstrated to reach as high as 89% in specific embodiments.
How to Synthesize p-Aryl Azophenol Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the diazonium salts and the choice of additives to ensure optimal conversion. The general protocol involves dissolving the aryl diazonium tetrafluoroborate precursors in a methanol-water mixture, followed by the addition of a base such as sodium acetate at room temperature. The reaction mixture is stirred for approximately 3 hours, during which time the transformation proceeds to completion as monitored by thin-layer chromatography. Workup typically involves extraction with dichloromethane, drying over anhydrous magnesium sulfate, and purification via silica gel column chromatography using petroleum ether and ethyl acetate gradients. This straightforward procedure minimizes the need for specialized equipment and allows for rapid iteration during process development. For detailed operational parameters and specific embodiment data, please refer to the standardized synthesis steps outlined below.
- Dissolve two equivalents of aryl diazonium tetrafluoroborate salts in a mixed solvent system comprising methanol and water.
- Add a stoichiometric amount of an additive such as sodium acetate or triethylamine to the reaction mixture at room temperature.
- Stir the reaction for approximately 3 hours, monitor by TLC, and isolate the product via extraction and silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts removes a significant cost center associated with purchasing expensive palladium or copper reagents and the subsequent validation required to prove their removal from the final product. Furthermore, the use of water as a co-solvent reduces the reliance on volatile organic compounds (VOCs), aligning manufacturing processes with increasingly strict environmental regulations and reducing waste disposal costs. The mild reaction conditions mean that standard stainless steel reactors can be used without the need for exotic lining or extreme cooling capabilities, lowering capital expenditure for scale-up. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive and readily available aryl diazonium salts and common bases like sodium acetate instead of precious metal catalysts. By operating at room temperature, the energy footprint of the reaction is drastically simplified, eliminating the need for cryogenic cooling or high-pressure heating systems. The high yields reported in the patent, such as 89% for unsubstituted derivatives and 86% for chloro-substituted analogs, indicate excellent atom economy which directly lowers the cost of goods sold. Additionally, the simplified workup procedure reduces labor hours and solvent consumption during purification, further enhancing the overall economic viability of the process for large volume production.
- Enhanced Supply Chain Reliability: Sourcing stable aryl diazonium tetrafluoroborate salts is generally more reliable than managing the logistics of hazardous in situ diazotization reagents. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification results. Since the method does not rely on scarce noble metals, it is immune to the geopolitical supply fluctuations that often plague catalyst-dependent syntheses. This stability allows for better long-term planning and inventory management, ensuring that downstream customers receive their orders of critical intermediates on time without interruption.
- Scalability and Environmental Compliance: The aqueous nature of the solvent system significantly reduces the generation of hazardous organic waste, simplifying effluent treatment and ensuring compliance with green chemistry principles. The absence of explosive hazards associated with traditional diazonium handling allows for safer scale-up from kilogram to tonne quantities without requiring specialized blast-proof facilities. This safety profile facilitates faster regulatory approval for manufacturing sites and reduces insurance premiums associated with chemical production. Consequently, manufacturers can expand capacity more rapidly to meet market demand while maintaining a strong environmental, social, and governance (ESG) profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages of the metal-free, water-compatible system described in CN113105357B.
Q: What are the primary advantages of this synthesis method over traditional Mills or Wallach reactions?
A: This method eliminates the need for excessive oxidizing or reducing agents required in Mills and Wallach reactions, significantly reducing toxic by-products. Furthermore, it operates under mild, transition metal-free conditions using water-compatible solvents, enhancing environmental safety and simplifying purification.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes stable aryl diazonium salts and operates at room temperature (0-40°C) without inert atmosphere requirements. These mild conditions, combined with the use of common solvents like methanol and water, make it highly suitable for large-scale commercial manufacturing.
Q: How does the reaction mechanism ensure high regioselectivity for the para-position?
A: The mechanism involves the formation of a phenol intermediate via hydrolysis of the diazonium salt, followed by deprotonation to a phenoxide anion. This nucleophile selectively couples with the second diazonium salt at the para-position due to electronic activation and steric factors, ensuring high purity of the target azophenol derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Aryl Azophenol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis methods disclosed in patent CN113105357B for producing high-quality azophenol intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical and specialty chemical applications. We are committed to leveraging this advanced metal-free technology to deliver superior products that enhance your supply chain efficiency.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener methodology. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Contact our technical procurement team today to discuss your requirements and secure a reliable supply of these critical intermediates for your future projects.
