Advanced Manufacturing of 2-Nitroimidazole: A Cost-Efficient Route for Radiosensitizer Intermediates
The pharmaceutical industry's demand for efficient radiosensitizers has driven significant innovation in the synthesis of key intermediates like 2-nitroimidazole. Patent CN111018788A introduces a transformative preparation method that fundamentally shifts the synthetic paradigm from expensive, specialized precursors to abundant bulk chemicals. By utilizing imidazole as the starting material and employing a novel imidazole-type ionic liquid system, this technology achieves high regioselectivity in the bromination step, effectively bypassing the traditional limitations associated with direct nitration. This breakthrough not only addresses the critical issue of raw material availability but also streamlines the production workflow, offering a robust solution for manufacturers seeking to optimize their supply chains for oncology drug intermediates. The strategic integration of green chemistry principles ensures that the process remains environmentally compliant while delivering high-purity outputs essential for downstream pharmaceutical applications.
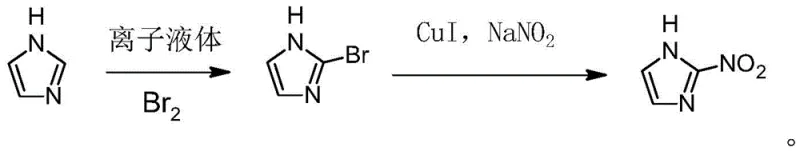
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-nitroimidazole has been plagued by significant economic and operational inefficiencies inherent in legacy methodologies. Prior art, such as the methods described in J. Am. Chem. Soc. (1965) and subsequent patents, predominantly relies on 2-aminoimidazole sulfate as the foundational building block. This starting material is not only prohibitively expensive due to its complex synthesis but also suffers from inconsistent market availability, creating bottlenecks for large-scale production. Furthermore, these conventional routes necessitate the use of hazardous reagents like fluoboric acid for diazotization, posing severe safety risks and requiring specialized containment infrastructure. The reliance on stoichiometric amounts of copper sulfate catalysts generates substantial volumes of heavy metal-contaminated wastewater, imposing heavy burdens on waste treatment facilities and increasing the overall environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN111018788A pioneers a streamlined two-step sequence that leverages the abundance and low cost of bulk imidazole. The core innovation lies in the use of a functionalized imidazole-type ionic liquid which acts as both a solvent and a directing agent, facilitating the selective introduction of a bromine atom at the 2-position of the imidazole ring. This regioselective bromination creates 2-bromoimidazole, a versatile intermediate that is subsequently converted to the target nitro compound via a copper-catalyzed nitrosation reaction. By eliminating the need for the scarce 2-aminoimidazole sulfate and removing dangerous fluoboric acid from the equation, this new route drastically simplifies the supply chain logistics. The process operates under milder conditions for the initial halogenation and utilizes catalytic rather than stoichiometric amounts of copper species in the second step, representing a significant leap forward in atom economy and process sustainability.
Mechanistic Insights into Ionic Liquid Mediated Regioselective Bromination
The success of this synthetic route hinges on the unique physicochemical properties of the imidazole-type ionic liquid employed in the first step. Unlike traditional molecular solvents, this ionic medium interacts specifically with the imidazole substrate to stabilize the transition state leading to 2-substitution, effectively suppressing the thermodynamically favored 4(5)-bromination that typically dominates in standard conditions. The mechanism involves the formation of a reddish-brown tribromoimidazole ionic liquid intermediate upon the addition of bromine, which then reacts with free imidazole to release the desired 2-bromoimidazole product. This controlled environment ensures that the bromine atom is installed precisely where needed, setting the stage for the subsequent nucleophilic substitution. The structural integrity of the ionic liquid, characterized by specific alkyl substituents on the nitrogen atoms, is crucial for maintaining this selectivity throughout the reaction course.
Following the isolation of the bromo-intermediate, the transformation to 2-nitroimidazole proceeds through a copper-catalyzed nitrosation mechanism using sodium nitrite. In this step, the cuprous iodide catalyst facilitates the displacement of the bromine atom by the nitro group in an aprotic polar solvent such as DMSO or DMF. The reaction temperature is carefully maintained between 120°C and 150°C to ensure complete conversion while minimizing side reactions. From an impurity control perspective, the use of a discrete intermediate (2-bromoimidazole) allows for purification before the final step, preventing the carryover of unreacted starting materials or regioisomers into the final API intermediate. This staged approach provides multiple checkpoints for quality assurance, ensuring that the final product meets the stringent purity specifications required for pharmaceutical grade materials, typically exceeding 98.5% content as demonstrated in the patent examples.
How to Synthesize 2-Nitroimidazole Efficiently
The practical implementation of this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins with the careful addition of bromine to the ionic liquid at controlled temperatures to manage exothermicity, followed by the introduction of imidazole to generate the brominated intermediate. After extraction and isolation, the second stage involves heating the intermediate with sodium nitrite and the copper catalyst in a polar aprotic solvent. For a comprehensive understanding of the specific operating procedures, safety protocols, and workup details, please refer to the standardized synthesis guide below.
- Selective Bromination: React imidazole-type ionic liquid with bromine at 20-30°C to form a tribromo-intermediate, then add bulk imidazole to generate 2-bromoimidazole with high regioselectivity.
- Extraction and Isolation: Extract the reaction mixture using organic solvents like toluene or chlorobenzene to isolate the 2-bromoimidazole intermediate from the ionic liquid phase.
- Catalytic Nitrosation: React the isolated 2-bromoimidazole with sodium nitrite in DMSO or DMF using cuprous iodide (CuI) as a catalyst at 120-150°C to yield high-purity 2-nitroimidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers profound strategic advantages rooted in raw material security and operational efficiency. By shifting the feedstock base from a niche, high-cost specialty chemical to a ubiquitous bulk commodity, manufacturers can insulate themselves from the volatility associated with specialized precursor markets. The elimination of fluoboric acid not only reduces the cost of hazardous material handling and storage but also simplifies regulatory compliance regarding transportation and worker safety. Furthermore, the reduction in copper usage from stoichiometric to catalytic levels significantly lowers the cost of waste treatment and disposal, contributing to a leaner and more sustainable cost structure. These factors combined create a more resilient supply chain capable of sustaining long-term production schedules without the risk of raw material shortages.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substitution of expensive 2-aminoimidazole sulfate with inexpensive bulk imidazole. Since the starting material represents a fundamental input cost, switching to a commodity chemical drastically lowers the baseline production expense. Additionally, the recovery and potential reuse of the ionic liquid solvent further enhance the economic viability of the process by reducing solvent consumption costs. The avoidance of large quantities of copper sulfate also translates to savings in both reagent procurement and the downstream costs associated with heavy metal waste remediation.
- Enhanced Supply Chain Reliability: Relying on bulk imidazole ensures a stable and continuous supply of raw materials, as it is produced globally at massive scales for various industrial applications. This contrasts sharply with the supply constraints often faced with specialized amino-imidazole derivatives, which may have limited suppliers and longer lead times. By decoupling production from these bottleneck materials, manufacturers can guarantee consistent delivery schedules to their pharmaceutical clients, thereby strengthening business relationships and market reputation. The simplified reagent list also reduces the complexity of vendor management and inventory control.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard unit operations such as liquid-liquid extraction and crystallization that are easily adapted from pilot to commercial scale. The significant reduction in hazardous waste generation, particularly the avoidance of fluorine-containing waste streams and excessive copper sludge, aligns perfectly with modern environmental regulations. This green profile facilitates easier permitting for new production lines and reduces the risk of operational shutdowns due to environmental non-compliance, ensuring uninterrupted supply for critical oncology medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing method.
Q: Why is the ionic liquid method superior to traditional 2-aminoimidazole routes?
A: Traditional methods rely on expensive and hard-to-source 2-aminoimidazole sulfate and hazardous fluoboric acid. The ionic liquid method uses cheap, bulk imidazole and eliminates dangerous reagents, significantly lowering raw material costs and EHS risks.
Q: How does this process improve regioselectivity for the 2-position?
A: Direct nitration of imidazole typically yields the 4(5)-nitro isomer. This patent utilizes a unique imidazole-type ionic liquid that directs bromination specifically to the 2-position, which is then converted to the nitro group, ensuring the correct isomer for radiosensitizer synthesis.
Q: What are the environmental benefits of this manufacturing process?
A: The process avoids the massive consumption of copper sulfate seen in older diazotization methods, reducing heavy metal wastewater. Additionally, replacing fluoboric acid with safer reagents improves operational safety and reduces hazardous waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitroimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving radiosensitizers. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative ionic liquid-mediated synthesis described in CN111018788A can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-nitroimidazole meets the exacting standards required for pharmaceutical synthesis. Our commitment to process excellence means we can deliver this complex intermediate with the consistency and reliability that global supply chains demand.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product's competitiveness in the global market.
