Advanced Synthesis of Rigid Chiral Catalysts for High-Purity Pharmaceutical Manufacturing
Introduction to Next-Generation Chiral Organocatalysts
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for higher purity and more robust catalytic systems in the production of active pharmaceutical ingredients. Patent CN103450073A introduces a significant breakthrough in this domain by detailing the synthesis and application of a novel R-diphenylprolinol chiral organic small molecule compound featuring a rigid cyclopropane structure. Unlike traditional prolinol derivatives which often suffer from conformational flexibility leading to variable stereoselectivity, this new catalyst design incorporates a fused cyclopropane ring to lock the molecular geometry. This structural modification is not merely an academic exercise but a practical solution to the longstanding challenges of controlling enantioselectivity and diastereoselectivity in complex organic transformations. By enhancing the rigidity of the catalyst backbone, the invention provides a reliable platform for executing asymmetric Michael additions with superior precision, addressing the critical needs of R&D directors seeking consistent high-performance reagents for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
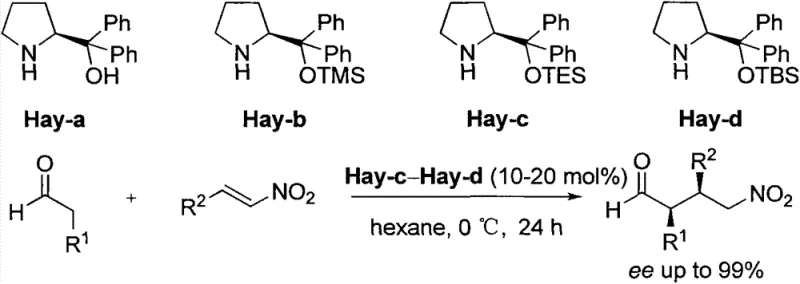
Prior to this innovation, the synthesis of chiral proline derivatives with cyclopropane motifs relied heavily on methodologies that were fraught with inefficiencies and safety concerns. As illustrated in the historical context of S-proline synthesis by the H. Stephen group, conventional routes often utilized toxic and expensive cyclization reagents such as trimethyltin methylide (Me3SnCH2). Furthermore, these legacy processes struggled significantly with stereocontrol during the ring-closing steps. The existing literature and prior art indicate that traditional Simmons-Smith type reactions on similar substrates often resulted in poor diastereomeric ratios, typically yielding mixtures where the desired isomer was not the major product. For instance, previous attempts yielded enantiomeric mixtures with ratios as low as 1:4, necessitating extensive and yield-depleting chiral separation processes. This lack of intrinsic selectivity not only inflated the cost of goods but also introduced substantial environmental burdens due to the generation of unwanted isomeric waste and the use of hazardous heavy metal reagents, rendering such methods unsuitable for modern, green chemical manufacturing standards.
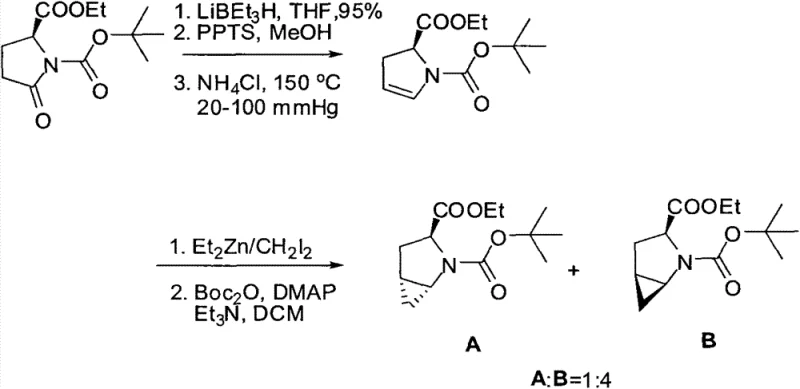
The Novel Approach
The methodology disclosed in CN103450073A represents a paradigm shift by optimizing the Simmons-Smith cyclopropanation conditions to favor the desired stereochemistry intrinsically. By carefully tuning the reaction parameters, specifically utilizing diethylzinc (ZnEt2) and chloroiodomethane (CH2ClI) at controlled low temperatures between -20°C and -15°C, the process achieves a remarkable diastereomeric ratio exceeding 25:1. This high level of selectivity is further enhanced by a specific post-reaction treatment involving aqueous ethylenediaminetetraacetic acid (EDTA) and amine solutions, which effectively purifies the crude mixture without the need for chromatographic resolution of enantiomers at this early stage. The subsequent steps involve a robust Grignard reaction with phenylmagnesium bromide followed by a mild acidic deprotection using trifluoroacetic acid. This streamlined three-step sequence eliminates the need for toxic tin reagents entirely and bypasses the costly resolution steps required by older methods, thereby offering a direct, high-yielding pathway to optically pure catalyst precursors that is both economically and environmentally superior.
Mechanistic Insights into Rigidity-Enhanced Asymmetric Catalysis
The core innovation of this technology lies in the strategic introduction of the cyclopropane ring fused to the pyrrolidine backbone, which fundamentally alters the conformational landscape of the catalyst. In traditional organocatalysis using Hayashi-type prolinol ethers, the flexibility of the five-membered ring can sometimes lead to multiple competing transition states, resulting in compromised enantioselectivity, especially with challenging substrates. The cyclopropane moiety acts as a rigid constraint, locking the nitrogen lone pair and the adjacent stereocenters into a fixed spatial arrangement. This increased rigidity amplifies the steric differentiation between the Re and Si faces of the incoming electrophile during the catalytic cycle. When applied to asymmetric Michael additions, such as the reaction between aldehydes and nitroalkenes, this structural preorganization ensures that the enamine intermediate adopts a single, highly favored conformation. Consequently, the nucleophilic attack occurs with high fidelity, translating the catalyst's chirality into the product with exceptional efficiency. This mechanistic advantage allows the catalyst to maintain high performance even under varied conditions, providing a broader substrate scope compared to its flexible counterparts.
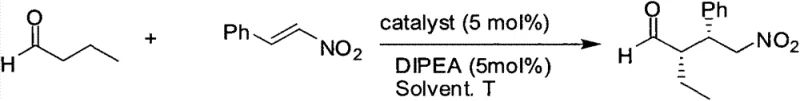
Furthermore, the impurity profile of reactions catalyzed by this rigid system is significantly cleaner due to the suppression of non-selective background pathways. The patent data demonstrates that under optimized conditions, specifically at -20°C, the catalyst can achieve enantiomeric excess (ee) values up to 91% with yields reaching 86%. This performance is a marked improvement over standard S-proline catalysts which, under similar comparative conditions, yielded only 75% ee. The ability to operate effectively at low temperatures without losing activity suggests a low activation energy barrier for the desired transition state relative to the undesired ones. For process chemists, this means that the reaction can be run with confidence that side products and opposite enantiomers will be minimized, simplifying downstream purification. The combination of high conversion and high stereoselectivity reduces the burden on analytical teams and ensures that the final API intermediates meet stringent regulatory specifications for chiral purity without requiring recursive recrystallization steps.
How to Synthesize R-Diphenylprolinol Efficiently
The synthesis of this high-value chiral catalyst is designed for operational simplicity while maintaining rigorous control over stereochemical outcomes. The process begins with the cyclopropanation of a protected pyrrole ester, followed by carbon-carbon bond formation via Grignard addition, and concludes with deprotection. Each step has been optimized to maximize yield and minimize the formation of difficult-to-remove impurities. The detailed standardized synthetic steps, including precise molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility at scale.
- Perform Simmons-Smith cyclopropanation on (R)-1-N-Boc-2,3-dihydro-2-pyrrolecarboxylate using ZnEt2 and CH2ClI at -20°C to achieve >25: 1 diastereoselectivity.
- Conduct a Grignard reaction with phenylmagnesium bromide at 65-75°C to introduce the diphenyl hydroxymethyl group.
- Execute acidic deprotection using trifluoroacetic acid (TFA) at 5°C to yield the final R-diphenylprolinol catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere technical performance. The elimination of toxic organotin reagents and the avoidance of chiral resolution steps translate directly into a more resilient and cost-effective supply chain. By securing a manufacturing process that relies on commodity chemicals like diethylzinc and chloroiodomethane rather than exotic, regulated substances, companies can mitigate supply risks associated with raw material availability. Moreover, the high intrinsic selectivity of the process means that less raw material is wasted on unwanted isomers, effectively increasing the overall mass efficiency of the production line. This efficiency gain is crucial for maintaining competitive pricing in the global market for chiral intermediates, where margin pressure is constant.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic reduction in processing costs associated with purification. Traditional methods that produce 1:4 isomeric mixtures require expensive chiral chromatography or multiple recrystallizations to isolate the active enantiomer, often resulting in a theoretical maximum yield loss of over 50%. In contrast, this new method achieves a >25:1 selectivity ratio directly from the reactor. This qualitative leap in selectivity means that the majority of the reacted material is the desired product, significantly reducing the volume of solvents and adsorbents needed for purification. Additionally, the removal of toxic tin reagents eliminates the costly waste disposal fees and specialized handling protocols required for heavy metal contaminants, further lowering the operational expenditure per kilogram of catalyst produced.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for complex chiral building blocks. This synthesis route utilizes widely available starting materials such as ethyl pyrrole carboxylate and standard Grignard reagents, which are produced by multiple chemical manufacturers globally. This diversification of the raw material base reduces the risk of supply disruptions caused by geopolitical issues or production outages at a single vendor. Furthermore, the robustness of the reaction conditions, which do not require ultra-low cryogenic temperatures below -20°C or high-pressure equipment, allows for production in a wider range of manufacturing facilities. This flexibility enables companies to dual-source production or shift manufacturing locations more easily, ensuring a steady flow of critical chiral catalysts to support uninterrupted API production schedules.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, the ability to scale a process without generating hazardous waste is a key competitive advantage. This method replaces toxic tin-based cyclization agents with zinc-based reagents, which are significantly less harmful and easier to manage in wastewater treatment systems. The high atom economy resulting from the high selectivity also means less chemical waste is generated per unit of product. From a scalability perspective, the exothermic nature of the Grignard and cyclopropanation steps is manageable within standard stainless steel reactors equipped with conventional cooling jackets. This compatibility with existing infrastructure allows for seamless scale-up from pilot plant quantities to multi-ton commercial production without the need for capital-intensive equipment upgrades, facilitating rapid response to market demand surges.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral catalyst technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational parameters and expected outcomes for potential users and partners.
Q: What is the key advantage of the cyclopropane-fused prolinol catalyst over traditional Hayashi catalysts?
A: The introduction of a rigid cyclopropane ring significantly increases steric hindrance and structural rigidity, solving the issue of poor diastereoselectivity found in flexible prolinol derivatives and achieving ee values up to 91%.
Q: How does this synthesis method improve cost efficiency compared to prior art?
A: By achieving a diastereomeric ratio greater than 25:1 directly, the process eliminates the need for costly and yield-loss-inducing chiral resolution steps required by older methods which typically produced 1:4 mixtures.
Q: Is this catalyst suitable for large-scale industrial asymmetric Michael additions?
A: Yes, the synthesis avoids toxic tin reagents and uses scalable conditions like Simmons-Smith cyclopropanation and standard Grignard reactions, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Diphenylprolinol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent examples can be reliably reproduced on an industrial scale. We understand that for pharmaceutical applications, consistency is paramount; therefore, our stringent purity specifications and rigorous QC labs are designed to guarantee that every batch of R-diphenylprolinol catalyst meets the highest standards of optical purity and chemical integrity required for GMP environments.
We invite you to collaborate with us to leverage this advanced catalytic technology for your next drug development project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes, demonstrating how switching to this high-selectivity catalyst can optimize your overall process economics. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and efficient supply of high-performance chiral intermediates for your global operations.
