Advanced Synthesis of Optically Pure Cis-Phosphine Ligands for Commercial Catalysis
Advanced Synthesis of Optically Pure Cis-Phosphine Ligands for Commercial Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly specific chiral ligands that can unlock new reactivities in pharmaceutical and fine chemical synthesis. A significant breakthrough in this domain is detailed in patent CN111484522B, which discloses a novel preparation method for optically pure cis-2-(diphenylphosphino)-1-cyclohexanecarboxylic acid. This compound represents a critical advancement over previous art, which was largely limited to racemic trans-isomers. The ability to access the cis-configuration with high optical purity opens new avenues for designing asymmetric Lewis acid and transition metal catalysts, directly addressing the needs of R&D teams seeking to optimize enantioselective transformations. The structural integrity and stereochemical definition of this molecule are paramount for its function as a ligand.
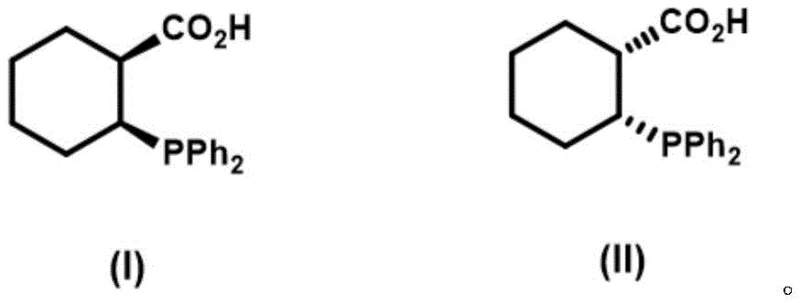
For procurement managers and supply chain heads, the introduction of this specific chiral building block signifies a move towards more reliable pharmaceutical intermediate supplier capabilities. The synthesis route described eliminates the need for difficult resolution steps often associated with racemic mixtures, thereby streamlining the production workflow. By establishing a direct asymmetric synthesis pathway, manufacturers can achieve substantial cost savings in fine chemical manufacturing through improved atom economy and reduced waste generation. Furthermore, the robustness of the reaction conditions suggests that the commercial scale-up of complex phosphine ligands is feasible without requiring exotic or prohibitively expensive infrastructure, ensuring a stable supply for downstream applications in drug discovery and process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthetic landscape for cyclohexyl-based phosphine carboxylic acids was dominated by the work of Stelzer et al. (2001), which reported the preparation of racemic trans-2-(diphenylphosphino)-1-cyclohexanecarboxylic acid. The reliance on racemic mixtures presents a fundamental limitation for modern asymmetric catalysis, where the precise spatial arrangement of donor atoms is crucial for inducing chirality in the substrate. Separating enantiomers from a racemic trans-mixture often involves cumbersome resolution processes using chiral resolving agents, which inherently caps the maximum theoretical yield at 50% and generates significant chemical waste. Additionally, the trans-geometry imposes different steric constraints compared to the cis-isomer, potentially limiting the scope of catalytic reactions where a specific bite angle or pocket size is required for optimal turnover and selectivity.
The Novel Approach
The methodology outlined in patent CN111484522B fundamentally shifts the paradigm by enabling the direct construction of the optically pure cis-isomer. This approach bypasses the thermodynamic pitfalls of isomerization and the inefficiencies of resolution. By utilizing a chiral magnesium-BINOL complex to catalyze the initial conjugate addition, the process installs the chiral center with exceptional fidelity right from the start. This strategic design ensures that every molecule produced contributes to the desired catalytic activity, effectively doubling the potential utility of the raw materials compared to racemic routes. For industry stakeholders, this translates to a more sustainable and economically viable pathway for producing high-purity OLED material precursors or specialized agrochemical intermediates that rely on precise chiral phosphine environments.
Mechanistic Insights into Asymmetric Conjugate Addition and Reduction
The core of this synthetic strategy lies in a sophisticated three-step cascade that meticulously controls stereochemistry. The process initiates with an asymmetric conjugate addition between methyl 1-cyclohexene-1-carboxylate and diphenylphosphine oxide. This step is mediated by a chiral catalyst system generated in situ from dibutylmagnesium, water, and (R) or (S)-H8-BINOL in tetrahydrofuran. The coordination of the magnesium species to the BINOL ligand creates a rigid chiral pocket that directs the nucleophilic attack of the phosphine oxide to one specific face of the cyclohexene double bond. This results in the formation of the first intermediate with 100% enantiomeric excess (ee) and diastereomeric ratio (dr), locking the cis-relationship between the phosphine oxide and the ester group. The reaction is conducted at controlled low temperatures (-30 to -15°C) to maximize selectivity and minimize side reactions.
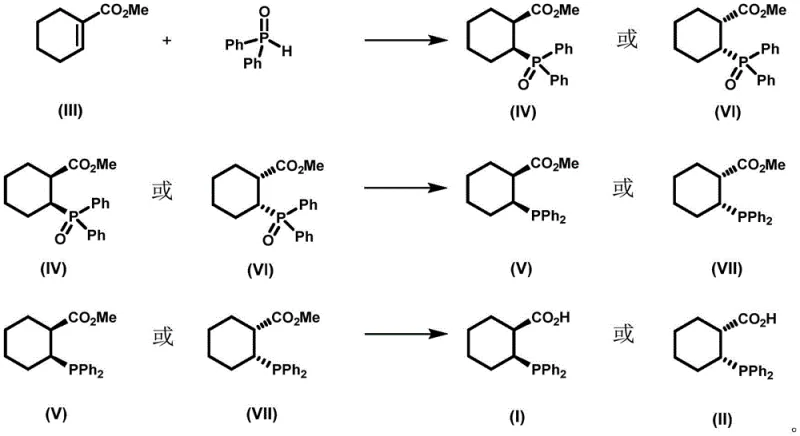
Following the establishment of chirality, the second phase involves the reduction of the phosphine oxide moiety to the free phosphine. This is achieved using a composite reducing system of trichlorosilane and triethylamine in toluene at elevated temperatures (90-110°C). This specific reagent combination is chosen for its ability to reduce the P=O bond efficiently without affecting the ester functionality or causing epimerization at the chiral center. The final step is the hydrolysis of the methyl ester to the carboxylic acid using aluminum trichloride in dimethyl sulfide. This Lewis acid-mediated hydrolysis is mild enough to preserve the sensitive phosphine group while ensuring complete conversion to the target acid. The entire sequence demonstrates a deep understanding of chemoselectivity, ensuring that the final product retains its optical purity throughout the transformation.
How to Synthesize cis-2-(diphenylphosphino)-1-cyclohexanecarboxylic Acid Efficiently
To implement this synthesis effectively, operators must adhere to strict anhydrous conditions and precise temperature controls, particularly during the initial catalytic addition step. The protocol requires the sequential addition of reagents to maintain the integrity of the active magnesium catalyst species. Detailed standard operating procedures regarding stoichiometry, quenching methods, and purification techniques are essential for reproducing the high yields and purity reported in the patent examples. The following guide outlines the critical operational parameters required to achieve successful outcomes in a laboratory or pilot plant setting.
- Perform asymmetric conjugate addition of methyl 1-cyclohexene-1-carboxylate with diphenylphosphine oxide using a Bu2Mg/(R or S)-H8-BINOL catalyst system at low temperature (-30 to -15°C) to establish chirality.
- Reduce the resulting phosphine oxide intermediate using a composite reducing agent of trichlorosilane and triethylamine in toluene at elevated temperatures (90-110°C) to obtain the phosphine species.
- Hydrolyze the ester group of the second intermediate using aluminum trichloride in dimethyl sulfide to yield the final optically pure cis-2-(diphenylphosphino)-1-cyclohexanecarboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers distinct advantages that align with the goals of cost reduction and supply chain resilience. The elimination of resolution steps not only improves yield but also simplifies the downstream processing requirements, reducing the load on purification units and solvent recovery systems. The reagents employed, such as trichlorosilane, triethylamine, and aluminum trichloride, are commodity chemicals available from multiple global sources, mitigating the risk of single-source supply bottlenecks. This accessibility ensures that the reducing lead time for high-purity pharmaceutical intermediates is minimized, allowing for faster response to market demands. Furthermore, the avoidance of precious metal catalysts in the synthesis of the ligand itself keeps the raw material costs predictable and stable.
- Cost Reduction in Manufacturing: The direct asymmetric synthesis route significantly lowers the cost of goods sold by maximizing the utilization of starting materials. Unlike resolution processes that discard half of the material, this method converts the majority of the input into the desired isomer. The use of standard solvents like THF and toluene, which are easily recycled, further contributes to operational expenditure savings. Additionally, the high selectivity reduces the formation of hard-to-remove impurities, lowering the cost associated with extensive chromatographic purification.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. The process does not rely on ultra-low cryogenic temperatures (below -40°C) or air-sensitive reagents that require specialized handling, making it easier to transfer technology between manufacturing sites. This flexibility allows for diversified production strategies, ensuring continuity of supply even if one facility faces operational challenges. The stability of the intermediates also allows for potential telescoping of steps, further streamlining logistics.
- Scalability and Environmental Compliance: The synthesis is designed with scalability in mind, utilizing reaction vessels and conditions compatible with standard chemical engineering practices. The waste profile is manageable, primarily consisting of magnesium and aluminum salts which can be treated using standard effluent protocols. By avoiding heavy metal catalysts in the ligand synthesis, the environmental footprint is reduced, facilitating compliance with increasingly stringent global environmental regulations and supporting green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chiral ligand. These answers are derived from the specific technical disclosures and beneficial effects highlighted in the patent documentation, providing clarity on the feasibility and advantages of adopting this new synthetic route for industrial applications.
Q: What is the primary advantage of this cis-phosphine ligand over existing trans-isomers?
A: Unlike the previously reported racemic trans-isomers, this optically pure cis-configuration offers a distinct steric environment that significantly enhances the performance of asymmetric Lewis acid and transition metal catalysts, filling a critical gap in chiral ligand availability.
Q: How is high enantiomeric purity maintained during the synthesis?
A: The process utilizes a highly stereoselective magnesium-BINOL catalytic system in the initial addition step, achieving 100% ee and dr values, which ensures that the chirality is locked in early and preserved through subsequent reduction and hydrolysis steps.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route employs robust, commercially available reagents such as trichlorosilane and aluminum trichloride, and avoids extreme cryogenic conditions (operating at -20°C rather than lower), making it highly suitable for commercial scale-up and consistent supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable cis-2-(diphenylphosphino)-1-cyclohexanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands in accelerating drug discovery and process optimization. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative chemistries like the one described in CN111484522B can be seamlessly transitioned from the bench to the plant. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of cis-2-(diphenylphosphino)-1-cyclohexanecarboxylic acid meets the exacting standards required for asymmetric catalysis.
We invite you to collaborate with us to leverage this technology for your specific projects. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient synthesis route can impact your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey towards more efficient and sustainable chemical manufacturing.
