Advanced Pd-Catalyzed Synthesis of 1,1-Diarylethene Derivatives for Commercial API Manufacturing
Advanced Pd-Catalyzed Synthesis of 1,1-Diarylethene Derivatives for Commercial API Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for high-value intermediates, particularly stilbene derivatives which serve as critical scaffolds for antiestrogen medications and herbicide antidotes. A significant technological breakthrough in this domain is detailed in Chinese Patent CN108586211B, which discloses a novel synthesis method for 1,1-diarylethene derivatives via palladium-catalyzed decarboxylative coupling. This innovation represents a paradigm shift from traditional, operationally complex methodologies to a more streamlined, industrially viable process. By utilizing readily available cinnamic acid derivatives and aryl triethoxysilanes as primary building blocks, the patented route eliminates the need for cryogenic conditions and unstable organometallic reagents. For R&D directors and procurement managers alike, this development signals a potential for substantial cost reduction in API manufacturing and enhanced supply chain reliability for complex organic intermediates.
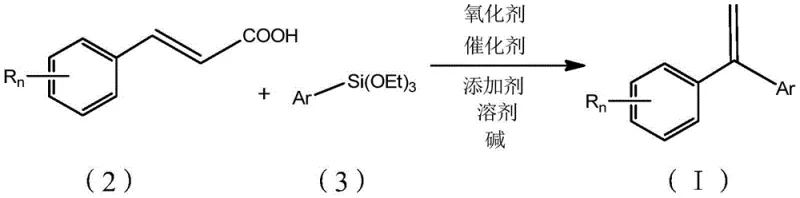
The core of this invention lies in the strategic selection of reactants that balance reactivity with commercial availability. The process involves mixing a cinnamic acid derivative with an aryl triethoxysilane in the presence of a specific catalytic system comprising a palladium source, a copper oxidant, a specialized nitrogen ligand, and a fluoride base. The reaction proceeds efficiently in polar aprotic solvents like DMSO under an inert argon atmosphere at temperatures ranging from 100 to 160 degrees Celsius. This thermal window is significantly more accessible than the minus 78 degrees Celsius required for competing Grignard-based routes, thereby lowering the barrier for commercial scale-up. The resulting 1,1-diarylethene structure is formed through a sophisticated mechanism involving oxidative addition, transmetallation, and decarboxylation, offering a versatile platform for generating diverse substituted stilbenes essential for modern drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,1-stilbene derivatives has been plagued by severe operational constraints and safety hazards that hinder large-scale production. Traditional methods often rely on the reaction of phenyl magnesium bromide with 1,1-diphenylethanol, a process that demands rigorous cryogenic control at minus 78 degrees Celsius to manage the exothermic nature of the Grignard reagent and prevent side reactions. Such extreme low-temperature requirements impose a massive energy burden on manufacturing facilities and necessitate specialized equipment that increases capital expenditure. Furthermore, alternative routes involving the reaction of bromobenzene with acetophenone frequently suffer from poor selectivity, generating significant amounts of side products that complicate downstream purification and reduce overall atom economy. More recent approaches utilizing iodobenzene and N-toluenesulfonylhydrazone introduce expensive reagents like tamoxifen precursors, drastically inflating the raw material costs and rendering the process economically unfeasible for commodity chemical production.
The Novel Approach
In stark contrast, the methodology described in patent CN108586211B offers a transformative solution by leveraging the stability and low cost of cinnamic acid derivatives and aryl triethoxysilanes. This new approach bypasses the need for hazardous organometallic reagents and cryogenic cooling, operating instead at moderate to high temperatures between 100 and 160 degrees Celsius. The use of aryl triethoxysilanes is particularly advantageous as these organosilicon compounds are chemically stable, easy to handle, and environmentally benign compared to their tin or zinc counterparts. The reaction is driven by a palladium catalyst system that facilitates the cleavage of the carbon-carbon double bond and the subsequent coupling with the aryl silane moiety. This decarboxylative strategy not only simplifies the experimental operation but also utilizes cheap and easily obtainable substrates, making it highly attractive for industrial development. The transition from harsh, low-yield protocols to this温和 (mild) yet efficient catalytic cycle represents a major step forward in sustainable chemical manufacturing.
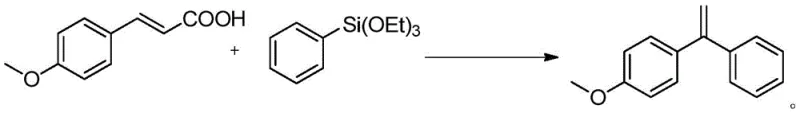
Mechanistic Insights into Pd-Catalyzed Decarboxylative Coupling
The success of this synthesis hinges on a finely tuned catalytic cycle involving palladium and copper species. The mechanism likely initiates with the oxidative addition of the palladium catalyst to the unsaturated carboxylic acid derivative, followed by decarboxylation to generate a reactive vinyl-palladium intermediate. Simultaneously, the aryl triethoxysilane is activated by the fluoride base, typically cesium fluoride (CsF), which enhances the nucleophilicity of the silicon-bound aryl group for transmetallation. The choice of ligand is critical; screening data indicates that bidentate nitrogen ligands, particularly 4,4'-di-tert-butyl-2,2'-bipyridine (L7), significantly outperform phenanthroline derivatives. The steric bulk of the tert-butyl groups in ligand L7 likely prevents the formation of inactive palladium black aggregates, thereby maintaining a high concentration of the active catalytic species throughout the reaction duration. This stabilization effect is crucial for achieving the reported yields of up to 58 percent under optimized conditions.
Furthermore, the role of the oxidant cannot be overstated in this redox-neutral coupling. While various copper salts were screened, copper(II) fluoride (CuF2) emerged as the superior oxidant, providing a 24 percent yield in initial screenings compared to negligible results with cuprous salts or other cupric variants. The fluoride ion from the oxidant may also synergistically assist the cesium fluoride base in activating the silicon reagent. The reaction environment is further optimized by the addition of p-benzoquinone (BQ) as an additive, which helps to re-oxidize the palladium species and sustain the catalytic turnover. Solvent selection also plays a pivotal role, with dimethyl sulfoxide (DMSO) proving far superior to DMF or DMA, likely due to its high polarity and ability to stabilize the charged intermediates involved in the decarboxylation step. Understanding these mechanistic nuances allows process chemists to troubleshoot and adapt the protocol for diverse substrate classes.
How to Synthesize 1,1-Diarylethene Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters identified during the patent's extensive screening phase. The procedure begins with the thorough degassing of the reaction vessel to establish an inert argon atmosphere, which is essential for preventing the oxidation of the sensitive palladium catalyst. Reagents are added in a specific sequence to ensure proper mixing and activation before heating. The following guide outlines the standardized operational steps derived from the patent examples, ensuring reproducibility and safety.
- Mix cinnamic acid derivative, aryl triethoxysilane, Pd catalyst, CuF2 oxidant, L7 ligand, CsF base, and BQ additive in DMSO solvent under argon protection.
- Heat the reaction mixture to 100-160°C (optimally 150°C) and stir for 2-24 hours (optimally 5 hours) to facilitate the coupling reaction.
- Filter the reaction mixture through celite, extract with ethyl acetate or dichloromethane, dry over magnesium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive and unstable reagents like phenyl magnesium bromide or iodobenzene-hydrazone complexes with commodity chemicals like cinnamic acid and triethoxysilanes, companies can significantly reduce their exposure to volatile market prices and supply disruptions. Cinnamic acid is a widely produced unsaturated carboxylic acid with a robust global supply chain, ensuring consistent availability for large-scale manufacturing campaigns. Moreover, the elimination of cryogenic requirements removes the need for specialized low-temperature reactors and the associated high energy consumption for cooling, leading to substantial operational cost savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The economic profile of this process is enhanced by the use of earth-abundant catalysts and inexpensive oxidants. While palladium is a precious metal, the optimized loading of 10 mol% combined with the high turnover facilitated by the L7 ligand ensures efficient metal utilization. More importantly, the avoidance of stoichiometric amounts of expensive organometallic reagents and the reduction in purification steps due to fewer side products contribute to a leaner cost structure. The ability to run the reaction at 150 degrees Celsius in standard glass-lined or stainless steel reactors without specialized cooling jackets further lowers the capital intensity of the manufacturing process, making it accessible for both batch and continuous flow production modes.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the chemical stability of the key starting materials. Aryl triethoxysilanes are known for their shelf stability and resistance to moisture compared to Grignard reagents, which require strict anhydrous conditions and immediate use. This stability allows for bulk purchasing and long-term storage, mitigating the risk of production delays caused by reagent degradation. Additionally, the reaction tolerates a variety of functional groups, meaning a single robust protocol can be applied to synthesize a library of derivatives for different drug candidates. This flexibility reduces the need for developing unique, bespoke processes for each new analog, accelerating the timeline from bench scale to commercial supply and ensuring a steady flow of high-purity intermediates to downstream formulation teams.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with green chemistry principles. The use of DMSO as a solvent, while requiring recovery, is preferable to large volumes of ether or THF often used in Grignard reactions. The reaction generates minimal hazardous waste, as the byproducts are primarily inorganic salts and silica species that are easier to dispose of than heavy metal sludge from other coupling methods. The simplicity of the workup procedure, involving filtration through celite and standard extraction, facilitates easy scale-up from gram to kilogram quantities without complex engineering changes. This straightforward scalability ensures that the technology can meet the demands of clinical trial material production as well as full commercial launch volumes without bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these details is crucial for process development teams evaluating the feasibility of this route for their specific target molecules.
Q: What are the critical reaction conditions for maximizing yield in this synthesis?
A: Optimal results are achieved using Pd(OAc)2 (10 mol%) as the catalyst, CuF2 (1.5 equiv) as the oxidant, and the bulky ligand L7 (15 mol%) in DMSO at 150°C for 5 hours.
Q: Why is this method superior to traditional Grignard reactions for stilbene derivatives?
A: Unlike Grignard reactions requiring harsh cryogenic conditions (-78°C), this method operates at elevated temperatures (100-160°C) using stable, commercially available cinnamic acids and organosilanes, significantly reducing energy costs and operational complexity.
Q: Does this protocol tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates good tolerance for various substituents including methoxy, halogens (Cl, F), cyano, and trifluoromethyl groups, although electron-withdrawing groups like fluorine may result in lower yields compared to chloro-substituted substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Diarylethene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN108586211B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise to translate such patented methodologies into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,1-diarylethene derivatives meets the highest quality standards required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of palladium catalysis and decarboxylative coupling with confidence.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you require custom synthesis of specific stilbene analogs or scale-up of the standard protocol, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can drive efficiency and reliability in your supply chain.
