Advanced PIFA-Mediated Synthesis of 3-Selenocoumarins for Commercial Pharmaceutical Applications
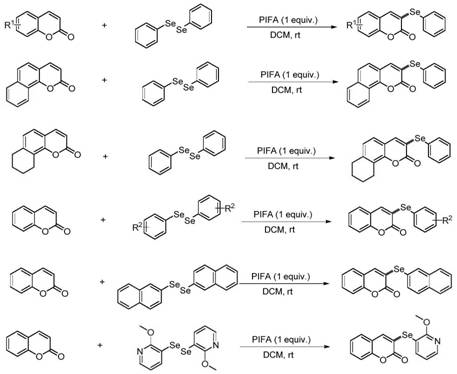
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing carbon-heteroatom bonds, particularly for bioactive scaffolds like coumarins. Patent CN110483460B introduces a groundbreaking preparation method for 3-selenocoumarin compounds, utilizing phenyliodine(III) bis(trifluoroacetate) (PIFA) as a mild yet powerful oxidant. This technology represents a significant leap forward in organic synthesis, enabling the direct regioselective selenization of coumarin backbones under exceptionally mild conditions. Unlike traditional approaches that often demand harsh environments or precious metal catalysts, this novel pathway operates efficiently at room temperature in dichloromethane (DCM) solvent. The strategic implementation of PIFA facilitates the generation of reactive selenium species that selectively attack the C-3 position of the coumarin ring, yielding high-purity intermediates essential for drug discovery. For R&D directors and procurement specialists alike, this patent offers a compelling value proposition: a streamlined, cost-effective route to valuable selenium-containing heterocycles that possess immense potential as anti-inflammatory, antioxidant, and anticancer agents.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted coumarins has been fraught with significant technical and economic challenges that hinder large-scale adoption. Conventional strategies typically rely on pre-functionalized reactant coupling or transition metal-catalyzed cross-dehydrocoupling, which introduce substantial complexity into the manufacturing workflow. Methods involving palladium complexes often necessitate the use of expensive ligands and additives, driving up the raw material costs significantly while complicating the purification process due to residual metal contamination. Furthermore, radical-based approaches using cobalt, copper, or iron catalysts frequently require elevated temperatures and excess oxidants to generate the necessary reactive species, leading to higher energy consumption and safety concerns in a plant setting. Another critical limitation of prior art is the restricted substrate scope; many existing protocols are only applicable to coumarins with specific substituents at the 4-position, severely limiting the chemical diversity accessible to medicinal chemists. These inefficiencies result in prolonged lead times, increased waste generation, and ultimately, a higher cost of goods sold for the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy techniques, the PIFA-mediated selenization method described in CN110483460B offers a streamlined, atom-economical solution that directly addresses these pain points. By employing PIFA as a hypervalent iodine oxidant, the reaction proceeds smoothly at room temperature under an air atmosphere, eliminating the need for inert gas protection or energy-intensive heating systems. This metal-free approach not only simplifies the operational procedure but also ensures that the final product is free from toxic heavy metal residues, a critical quality attribute for pharmaceutical intermediates. The methodology demonstrates exceptional versatility, accommodating a wide range of coumarin substrates with various electronic properties, including electron-donating and electron-withdrawing groups, without compromising yield or selectivity. Moreover, the use of readily available diselenides as selenium sources further enhances the economic viability of the process, making it an attractive option for cost-conscious procurement managers looking to optimize their supply chain for complex heterocyclic building blocks.
Mechanistic Insights into PIFA-Catalyzed Regioselective Selenization
Understanding the underlying mechanistic pathway is crucial for R&D teams aiming to replicate or adapt this synthesis for specific analog libraries. The reaction initiates with the interaction between PIFA and the diselenide compound, leading to the homolytic cleavage of the Se-Se bond and the generation of phenylselenol radicals alongside trifluoroacetoxyiodobenzene radicals. These highly reactive selenium radicals then undergo electrophilic attack at the electron-rich C-3 position of the coumarin double bond, forming a transient seleno-radical intermediate. This step is the key determinant of regioselectivity, ensuring that functionalization occurs exclusively at the desired position rather than other potential sites on the aromatic ring. Subsequently, the seleno-radical intermediate is oxidized by the trifluoroacetoxyiodobenzene radical species to produce a seleno-cation intermediate, which is a pivotal high-energy state in the catalytic cycle. Finally, a deprotonation event restores aromaticity and stability to the system, delivering the final 3-selenocoumarin product with high fidelity. This radical-cation manifold avoids the need for external metal catalysts, relying instead on the redox potential of the hypervalent iodine reagent to drive the transformation efficiently.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the risk of metal-induced side reactions or catalyst poisoning, which can often lead to difficult-to-remove impurities in the final batch. The mild reaction conditions also minimize thermal degradation of sensitive functional groups, preserving the integrity of complex substrates that might otherwise decompose under harsher regimes. Furthermore, the stoichiometry of the reaction is well-defined, with a molar ratio of coumarin to diselenide optimized at 1:1.2 to maximize conversion while minimizing excess reagent waste. The specificity of the PIFA oxidant ensures that over-oxidation side products are kept to a minimum, resulting in a cleaner crude reaction profile that simplifies downstream purification. For quality assurance teams, this translates to more consistent batch-to-batch reproducibility and easier compliance with stringent regulatory standards regarding impurity profiles in drug substances.
How to Synthesize 3-Selenocoumarin Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to precise procedural guidelines that leverage the unique reactivity of the PIFA system. The process begins with the dissolution of the coumarin substrate and the corresponding diselenide in dry dichloromethane, ensuring a homogeneous reaction medium that facilitates efficient mass transfer. Upon addition of the PIFA oxidant at room temperature, the reaction mixture should be stirred vigorously to maintain suspension and contact between reagents, with progress monitored closely via thin-layer chromatography (TLC) to prevent over-reaction. Once conversion is complete, typically within a short timeframe ranging from 30 minutes to a few hours depending on the substrate, the workup involves a standard aqueous extraction to remove iodine byproducts and acid residues. The organic layer is then dried and concentrated, followed by purification via column chromatography using a petroleum ether and ethyl acetate gradient to isolate the target 3-selenocoumarin in high purity. Detailed standardized synthesis steps are provided below for technical reference.
- Prepare the reaction mixture by combining coumarin substrates and diselenide compounds in dichloromethane (DCM) solvent under air atmosphere.
- Add PIFA (phenyliodine(III) bis(trifluoroacetate)) as the oxidant at room temperature to initiate the regioselective selenization process.
- Monitor reaction progress via TLC, then perform aqueous workup and column chromatography to isolate high-purity 3-selenocoumarin products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this PIFA-mediated synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures and improved throughput. By eliminating the requirement for expensive transition metal catalysts such as palladium or copper, companies can achieve significant cost reduction in pharmaceutical intermediate manufacturing, as there is no longer a need to source, store, or recover these precious materials. Additionally, the removal of metal catalysts obviates the need for specialized metal scavenging steps during purification, which are often time-consuming and add considerable cost to the overall production cycle. This streamlining of the workflow allows for faster turnaround times from synthesis to final product release, enhancing the agility of the supply chain in responding to market demands.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound, driven primarily by the substitution of costly catalysts with inexpensive hypervalent iodine reagents. Since PIFA is commercially available in bulk quantities at a fraction of the cost of noble metals, the raw material expenditure per kilogram of product is substantially lowered. Furthermore, the reaction proceeds at room temperature, which eliminates the energy costs associated with heating or cooling reactors to extreme conditions, contributing to a lower carbon footprint and reduced utility bills. The high yields reported in the patent data, often exceeding 90% for a broad range of substrates, mean that less starting material is wasted, improving the overall material efficiency of the process. These factors combine to create a highly competitive cost structure that allows suppliers to offer better pricing to downstream pharmaceutical clients without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of readily available and stable reagents like coumarins, diselenides, and PIFA, which are not subject to the same geopolitical supply constraints as rare earth metals or specialized ligands. The robustness of the reaction conditions, which tolerate air and moisture better than many organometallic processes, reduces the risk of batch failures due to environmental fluctuations, ensuring consistent delivery schedules. This reliability is critical for maintaining continuous production lines for API intermediates, where interruptions can have cascading effects on drug development timelines. By adopting a synthesis route that relies on commodity chemicals rather than bespoke catalysts, procurement teams can diversify their supplier base and mitigate the risk of single-source bottlenecks, thereby securing a more stable flow of materials.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the simplicity of the operation, which does not require specialized high-pressure equipment or complex inert atmosphere setups. The use of DCM as a solvent, while requiring proper handling, is well-established in industrial settings with existing recovery infrastructure, making the transition to large-scale production seamless. From an environmental perspective, the absence of heavy metals simplifies waste treatment protocols, as effluent streams do not require extensive processing to meet strict regulatory limits for metal content. This ease of compliance reduces the administrative and financial burden associated with environmental health and safety (EHS) management, allowing facilities to operate more sustainably. The combination of high atom economy and mild conditions makes this method an ideal candidate for green chemistry initiatives, aligning corporate manufacturing goals with global sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenization technology, derived directly from the patent specifications and practical application scenarios. These insights are designed to clarify the operational parameters and benefits for stakeholders evaluating this method for integration into their existing portfolios. Understanding these details is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of using PIFA over traditional metal catalysts for selenization?
A: Using PIFA eliminates the need for expensive transition metal catalysts like palladium or copper, thereby removing complex metal removal steps and reducing heavy metal contamination risks in the final pharmaceutical intermediate.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction operates at room temperature in common solvents like DCM with short reaction times, making it highly suitable for scale-up without requiring specialized high-pressure or high-temperature equipment.
Q: What is the regioselectivity profile of this selenization method?
A: The method achieves direct regioselective selenization at the 3-position of the coumarin backbone, overcoming limitations of previous methods that were restricted to 4-substituted coumarins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Selenocoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates like 3-selenocoumarins to accelerate drug discovery and development programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop innovation to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-selenocoumarin delivered meets the highest industry standards for potency and impurity control. We understand that consistency is key in pharmaceutical supply, and our dedicated technical team is committed to providing uninterrupted support throughout the lifecycle of your product.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods through the implementation of this advanced PIFA-mediated synthesis. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us help you secure a reliable source of high-purity 3-selenocoumarins that empowers your research and commercial success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
