Scalable Iridium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral 3,4-Dihydroquinazolinone Intermediates
Scalable Iridium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral 3,4-Dihydroquinazolinone Intermediates
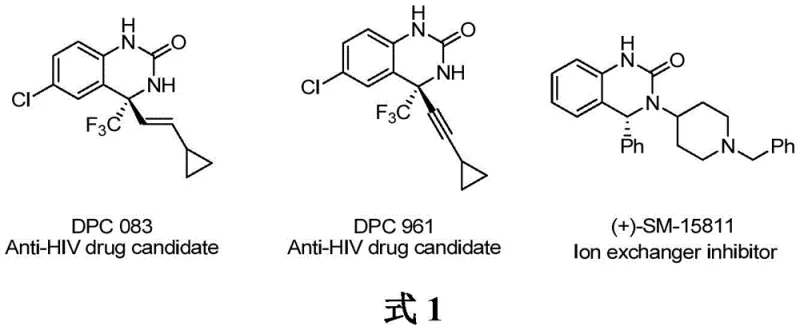
The pharmaceutical industry continuously demands robust and scalable synthetic routes for chiral heterocyclic scaffolds, particularly those exhibiting potent biological activity. Patent CN110776470B introduces a significant technological advancement in this domain by disclosing a highly efficient method for synthesizing chiral 3,4-dihydroquinazolinones via iridium-catalyzed asymmetric hydrogenation. This core structural motif is prevalent in numerous bioactive molecules, including second-generation non-nucleoside reverse transcriptase inhibitors like DPC 083 and DPC 961 used in HIV treatment, as well as ion exchange inhibitors such as SM-15811 for ischemic heart disease. The patented methodology addresses critical challenges in stereoselective synthesis, offering a pathway that combines mild reaction conditions with exceptional enantiocontrol, achieving enantiomeric excess values of up to 98%. For R&D directors and process chemists, this represents a viable alternative to traditional nucleophilic additions, providing a direct route to high-value API intermediates with superior atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the chiral center in 3,4-dihydroquinazolinone frameworks has relied heavily on the asymmetric addition of nucleophiles to quinazolinone precursors. While effective in specific contexts, these conventional strategies often suffer from significant drawbacks regarding substrate scope and operational complexity. Prior art, such as the palladium-catalyzed asymmetric hydrogenation reported by the Zhou group, demonstrated high enantioselectivity but was severely restricted in its applicability, functioning effectively only with trifluoromethyl or difluoromethyl substituted quinazolinones. This narrow substrate tolerance limits the utility of such methods for the diverse library synthesis required in modern drug discovery. Furthermore, traditional nucleophilic approaches frequently necessitate the use of stoichiometric chiral auxiliaries or harsh reaction conditions that generate substantial chemical waste, complicating downstream purification and increasing the environmental footprint of the manufacturing process.
The Novel Approach
The invention detailed in CN110776470B overcomes these historical bottlenecks by employing a transition metal iridium catalytic system tailored for broad-spectrum substrate compatibility. Unlike previous methods constrained by electronic properties of the substrate, this novel approach successfully hydrogenates quinazolinones bearing a wide array of substituents, including linear and branched alkyl groups, cyclic alkyls like cyclohexyl, and various substituted aryl rings. The introduction of hydroxyl groups or other functional moieties on the aromatic ring is shown to enhance substrate activity, facilitating smoother hydrogenation. By shifting from palladium to an optimized iridium-ligand complex, the process achieves not only higher yields but also maintains rigorous stereochemical control across diverse chemical spaces. This versatility allows for the synthesis of a multitude of chiral derivatives from a single generalized protocol, significantly streamlining the development of new pharmaceutical candidates.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this synthetic breakthrough lies in the precise orchestration of the iridium catalytic cycle, which is activated through the synergistic interaction between a metal precursor, a chiral diphosphine ligand, and a specific halogenated additive. The reaction initiates with the formation of an active cationic iridium species, generated in situ by mixing precursors such as [Ir(COD)Cl]2 with bulky, electron-rich chiral ligands like (R)-SegPhos, (R)-MeOBIPHEP, or (R)-DifluorPhos. The presence of additives such as bromochlorohydantoin (BCDMH), N-bromosuccinimide, or iodine is critical; these agents likely function to abstract chloride ligands from the iridium center, creating a coordinatively unsaturated cationic complex that is highly reactive towards hydrogen activation. This active species then coordinates with the C=N double bond of the quinazolinone substrate, delivering hydride and proton in a stereo-defined manner dictated by the chiral pocket of the ligand.

Impurity control in this system is inherently managed by the high chemoselectivity of the iridium catalyst. The mild conditions, typically ranging from 25°C to 80°C and hydrogen pressures between 100 to 1000 psi, minimize side reactions such as over-reduction or decomposition of sensitive functional groups often found in complex drug intermediates. The patent data indicates that the choice of solvent plays a pivotal role in maintaining catalyst stability and selectivity, with tetrahydrofuran (THF) emerging as a superior medium compared to methanol or dichloromethane in many instances. By optimizing the molar ratios of the metal precursor, ligand, and additive—typically keeping the metal loading as low as 1.0 mol%—the process ensures that the formation of racemic background products is suppressed. This mechanistic precision results in clean reaction profiles where the desired chiral amine is the predominant species, simplifying the isolation of high-purity material essential for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Chiral 3,4-Dihydroquinazolinone Efficiently
The operational protocol described in the patent is designed for practical implementation in both laboratory and pilot-scale settings, emphasizing ease of handling and reproducibility. The synthesis is divided into two distinct stages: the pre-formation of the catalyst complex and the subsequent hydrogenation of the substrate. This separation allows for quality control of the active catalytic species before introducing the valuable starting material. The process utilizes commercially available reagents and standard high-pressure equipment, removing barriers to adoption for contract manufacturing organizations. Detailed below is the strategic workflow for executing this transformation, ensuring optimal yield and enantiomeric purity.
- Prepare the active catalyst by mixing an iridium metal precursor, such as [Ir(COD)Cl]2, with a chiral diphosphine ligand like (R)-SegPhos in an organic solvent under nitrogen protection.
- Combine the prepared catalyst solution with the quinazolinone substrate and a halogenated additive (e.g., bromochlorohydantoin) in a high-pressure reactor.
- Charge the reactor with hydrogen gas at 100-1000 psi and stir at 25-80°C for 12-24 hours, followed by solvent removal and purification to isolate the chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this iridium-catalyzed hydrogenation route offers compelling economic and logistical benefits over legacy synthetic pathways. The primary advantage stems from the drastic simplification of the supply chain for chiral building blocks; rather than sourcing expensive, pre-functionalized chiral starting materials, manufacturers can utilize readily available achiral quinazolinone precursors. The catalytic nature of the process means that the expensive chiral information is imparted by a reusable or low-loading catalyst system, rather than stoichiometric chiral reagents that drive up the cost of goods significantly. This shift from stoichiometric to catalytic chirality represents a fundamental cost reduction in fine chemical manufacturing, allowing for more competitive pricing of the final API intermediate without compromising on quality or purity specifications.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the use of low catalyst loadings (as low as 1 mol%) significantly lower the raw material costs associated with chiral synthesis. Furthermore, the high atom economy of hydrogenation means that fewer atoms are wasted as byproducts, reducing the burden on waste treatment facilities and lowering disposal costs. The mild reaction temperatures also translate to reduced energy consumption for heating and cooling, contributing to a leaner overall manufacturing budget. By avoiding complex multi-step sequences often required to install chirality early in the synthesis, the overall process mass intensity is improved, leading to substantial cost savings in solvent usage and processing time.
- Enhanced Supply Chain Reliability: The reliance on commercially available iridium precursors and ligands, such as SegPhos and BINAP derivatives, ensures a stable and secure supply of critical reagents. Unlike proprietary enzymes or bespoke organocatalysts that may have long lead times or single-source risks, the components of this catalytic system are standard offerings from major chemical suppliers globally. This availability mitigates the risk of production delays caused by reagent shortages. Additionally, the robustness of the reaction conditions allows for flexibility in scheduling and batch sizing, enabling manufacturers to respond more agilely to fluctuating market demands for key pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently under pressures and temperatures that are standard in industrial hydrogenation reactors. The use of common solvents like THF and toluene facilitates straightforward solvent recovery and recycling programs, aligning with green chemistry principles. The high selectivity of the reaction minimizes the generation of difficult-to-separate diastereomers or regioisomers, which simplifies the purification train and reduces the volume of hazardous waste generated per kilogram of product. This environmental efficiency not only lowers compliance costs but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within CN110776470B, providing clarity on the operational parameters and expected outcomes for process development teams evaluating this route for commercial production.
Q: What is the maximum enantiomeric excess achieved in this iridium-catalyzed process?
A: According to patent CN110776470B, the method achieves excellent stereoselectivity with enantiomeric excess (ee) values reaching up to 98%, specifically demonstrated with 4-phenyl substituted substrates using optimized ligand systems.
Q: Which additives are critical for activating the iridium catalyst in this hydrogenation?
A: The patent identifies halogenated additives as essential for generating the active cationic iridium species. Effective additives include N-bromosuccinimide, bromochlorohydantoin (BCDMH), trichloroisocyanuric acid, and iodine, which significantly improve both conversion and enantioselectivity.
Q: Does this method support a broad range of substrate substituents?
A: Yes, unlike previous palladium-catalyzed methods limited to fluorinated groups, this iridium system accommodates diverse substituents including alkyl (methyl, isopropyl, cyclohexyl), aryl (phenyl, tolyl), and alkoxy groups, making it highly versatile for medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3,4-Dihydroquinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the iridium-catalyzed asymmetric hydrogenation pathway described in CN110776470B and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity observed in the lab is maintained at the plant scale. Our facility is outfitted with state-of-the-art high-pressure hydrogenation reactors and stringent purity specifications are enforced through our rigorous QC labs, guaranteeing that every batch of chiral 3,4-dihydroquinazolinone meets the exacting standards required for global pharmaceutical registration.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project needs. By leveraging our expertise in catalytic hydrogenation and process optimization, we can help you reduce lead time for high-purity pharmaceutical intermediates while optimizing your overall cost structure. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules, and let us demonstrate how our manufacturing capabilities can accelerate your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
