Revolutionizing Monosulfide Production: A Scalable Selenium-Catalyzed Approach for Global Supply Chains
The landscape of fine chemical synthesis is constantly evolving, driven by the dual demands of higher purity and sustainable manufacturing practices. A pivotal advancement in this domain is detailed in patent CN102127038A, which introduces a novel method for synthesizing monosulfide compounds using a selenium-catalyzed carbonylation strategy. This technology represents a significant departure from classical thioether synthesis, offering a robust pathway for producing high-value pharmaceutical intermediates and agrochemical precursors. By leveraging carbon monoxide and water as key reagents alongside inexpensive selenium powder, this process achieves exceptional selectivity under mild atmospheric conditions. For global procurement leaders and R&D directors, understanding the mechanistic elegance and operational simplicity of this patent is crucial for securing a competitive edge in the supply of complex sulfur-containing molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of monosulfides has relied heavily on nucleophilic substitution reactions involving alkali metal sulfides, such as potassium sulfide or sodium sulfide, reacting with halogenated hydrocarbons in alcoholic solvents. While chemically straightforward, these legacy processes suffer from severe operational drawbacks that hinder modern manufacturing efficiency. The reaction conditions are often苛刻 (harsh), requiring strict anhydrous environments and elevated temperatures that pose safety risks and increase energy consumption. Furthermore, the stoichiometric use of inorganic sulfides generates substantial quantities of inorganic salt waste, creating a significant burden on wastewater treatment facilities and driving up disposal costs. The purification of the final thioether product is frequently complicated by the presence of polysulfide byproducts and unreacted starting materials, necessitating energy-intensive distillation or chromatography steps that erode overall process yield and profitability.
The Novel Approach
In stark contrast, the selenium-catalyzed method described in the patent data offers a transformative solution by shifting the paradigm from stoichiometric reagents to catalytic cycles. This approach utilizes disulfides and halogenated hydrocarbons as starting materials in the presence of carbon monoxide and water, mediated by elemental selenium. The reaction proceeds smoothly at atmospheric pressure within a temperature range of 20 to 100°C, significantly reducing the engineering controls required for high-pressure reactors. A defining feature of this novelty is the "one-pot" operational design, where the transformation occurs in a single vessel without the need for intermediate isolation. This streamlining not only accelerates the production timeline but also minimizes material loss during transfer operations. The use of readily available industrial solvents and the potential to utilize industrial tail gas containing carbon monoxide further underscores the economic and logistical viability of this method for large-scale applications.
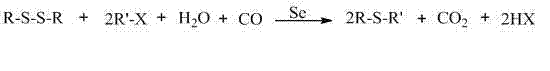
Mechanistic Insights into Selenium-Catalyzed Carbonylation
To fully appreciate the value proposition for R&D teams, one must delve into the unique catalytic behavior of selenium in this system. Unlike transition metal catalysts that often require complex ligand systems to maintain stability, elemental selenium acts as a versatile redox mediator. The proposed mechanism involves the initial activation of the disulfide bond by the selenium species, facilitated by the presence of a base. Carbon monoxide serves as a reducing agent and a carbonyl source, effectively inserting itself into the reaction coordinate to facilitate the cleavage of the S-S bond and the subsequent formation of the C-S bond. The presence of water is critical, acting as a proton source and participating in the hydrolysis steps that regenerate the active catalytic species. This intricate interplay ensures that the reaction maintains high turnover numbers while avoiding the accumulation of toxic or unstable intermediates that could compromise product quality.
Perhaps the most commercially attractive aspect of this mechanism is the phenomenon known as "quasi-homogeneous" or phase-transfer catalysis. The patent data highlights that while the selenium catalyst begins as an insoluble solid powder, it transforms into soluble active species during the reaction, ensuring efficient contact with the substrates in the liquid phase. Upon completion of the reaction and cooling, the catalyst spontaneously precipitates back into its solid elemental form. This self-separating characteristic is a game-changer for downstream processing. It eliminates the need for complex filtration through celite or the use of scavenger resins typically required to remove trace metal contaminants from pharmaceutical intermediates. Consequently, the final product exhibits a cleaner impurity profile, and the recovered selenium can be directly recycled into subsequent batches, driving down the effective cost of goods sold (COGS) over time.
How to Synthesize Furfuryl Sulfide Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The standard protocol involves charging a reactor with the specific disulfide substrate, such as furfuryl disulfide, and the corresponding alkyl halide. A catalytic amount of selenium powder is added along with a base, which can range from inorganic options like sodium acetate to organic amines like triethylamine, depending on the substrate's sensitivity. The reaction mixture is then subjected to a continuous flow of carbon monoxide at atmospheric pressure while being heated to the optimal temperature, typically around 90°C for many substrates. After the designated reaction time, the system is purged with air to oxidize any remaining reactive species before filtration. The detailed standardized synthesis steps for specific derivatives are outlined below.
- Charge a reactor with disulfide compounds, halogenated hydrocarbons, selenium powder, water, and an organic or inorganic base in a suitable solvent.
- Maintain a continuous flow of carbon monoxide at atmospheric pressure while heating the mixture to 20-100°C for 1 to 24 hours.
- Cool the reaction, switch gas flow to air to oxidize residual species, filter off the recovered solid selenium catalyst, and purify the filtrate via extraction and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this selenium-catalyzed technology translates into tangible strategic benefits beyond mere chemical curiosity. The shift away from hazardous high-pressure conditions and corrosive sulfide salts fundamentally alters the risk profile of the manufacturing site. By operating at atmospheric pressure, the capital expenditure (CAPEX) required for specialized pressure vessels and associated safety systems is drastically reduced. This lower barrier to entry allows for more flexible manufacturing networks, potentially enabling production closer to key markets to reduce logistics lead times. Furthermore, the simplified work-up procedure, characterized by the easy filtration of the solid catalyst, reduces the demand for labor-intensive purification steps and consumables, directly impacting the operational expenditure (OPEX).
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the use of low-cost, non-metallic selenium as the catalyst, avoiding the volatility associated with precious metal prices like palladium or platinum. The ability to recover and reuse the catalyst multiple times without significant loss of activity creates a closed-loop system that minimizes raw material waste. Additionally, the high selectivity of the reaction means that less starting material is lost to side reactions, improving the overall atom economy. These factors combine to offer a substantially lower cost structure compared to traditional sulfide alkylation methods, providing a buffer against raw material price fluctuations in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by the reliance on specialized reagents that have limited suppliers. This method utilizes commodity chemicals such as carbon monoxide, water, and common alkyl halides, which are widely available from multiple global sources. The robustness of the reaction conditions, tolerating a wide range of temperatures and solvent systems, ensures that production schedules are less likely to be disrupted by minor variations in utility supply or ambient conditions. This reliability is critical for maintaining consistent inventory levels for downstream pharmaceutical customers who operate on Just-In-Time manufacturing principles.
- Scalability and Environmental Compliance: As regulatory scrutiny on chemical manufacturing intensifies, the "green" credentials of this process offer a significant compliance advantage. The absence of heavy metal waste streams and the reduction of saline effluent simplify the permitting process for new production lines. The process is inherently scalable; the kinetics observed in laboratory settings translate well to pilot and commercial scales due to the lack of mass transfer limitations often seen in heterogeneous gas-liquid reactions. This scalability ensures that suppliers can rapidly ramp up production volumes to meet surging market demand for key intermediates without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium-catalyzed synthesis. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains.
Q: What are the primary advantages of using selenium over traditional sulfide sources?
A: Unlike traditional methods using potassium sulfide which require harsh conditions and generate significant waste, selenium catalysis operates under mild atmospheric pressure with high selectivity. Furthermore, the selenium catalyst exhibits a unique phase-transfer behavior, precipitating out as a solid post-reaction for easy recovery and reuse, drastically simplifying downstream processing.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It utilizes common industrial solvents and operates at atmospheric pressure, eliminating the need for expensive high-pressure autoclaves. The 'one-pot' nature of the reaction reduces equipment footprint and operational complexity, making it highly viable for commercial scale-up.
Q: How does this method impact the environmental footprint of thioether production?
A: This method is classified as a green chemical process. It avoids the generation of heavy metal waste associated with other catalytic systems and utilizes carbon monoxide efficiently to produce CO2 as a byproduct. The ability to recover and reuse the selenium catalyst further minimizes solid waste generation, aligning with strict modern environmental compliance standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monosulfide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven execution capabilities. Our team has extensively analyzed the selenium-catalyzed pathways described in recent intellectual property and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that moving from gram-scale optimization to multi-ton manufacturing involves complex engineering challenges, particularly in handling gaseous reagents like carbon monoxide safely. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of monosulfide intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of novel thioether derivatives or the scale-up of existing processes to improve margins, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing protocols can enhance your supply chain efficiency and product quality.
