Advanced Synthesis of Polycarbonyl Carbazole Derivatives for Commercial Scale-up
Advanced Synthesis of Polycarbonyl Carbazole Derivatives for Commercial Scale-up
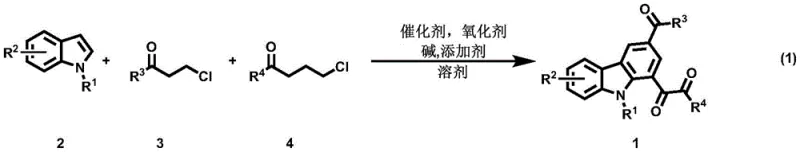
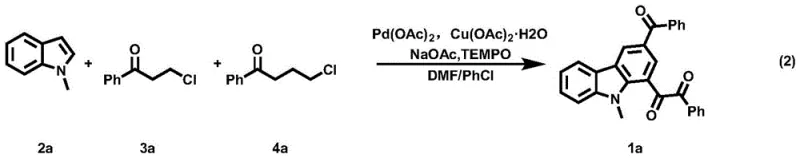
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex nitrogen-containing heterocycles, particularly carbazole derivatives, which serve as critical scaffolds in drug discovery and organic electronics. Patent CN115974761A introduces a groundbreaking preparation method for polycarbonyl carbazole derivatives that addresses longstanding challenges in synthetic efficiency and substrate versatility. This technology leverages a sophisticated domino reaction sequence involving indole derivatives and chloroketones, facilitated by a palladium-catalyzed system under mild alkaline conditions. For R&D directors and procurement managers alike, this innovation represents a significant leap forward, offering a streamlined pathway to high-value intermediates that were previously difficult to access economically. The ability to synthesize these structurally diverse molecules in a one-pot fashion not only simplifies the workflow but also enhances the overall atom economy of the process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbazole derivatives has relied on classical strategies such as the Graebe-Ullmann reaction, Borsche-Drechsel cyclization, and Fischer-Borsche synthesis. While these methods laid the foundation for heterocyclic chemistry, they are often plagued by severe operational drawbacks that hinder modern commercial application. These traditional routes typically necessitate harsh reaction conditions, including high temperatures and strong acidic or basic environments, which can degrade sensitive functional groups and limit the scope of applicable substrates. Furthermore, the multi-step nature of these conventional processes results in prolonged production cycles and accumulated waste, driving up the cost of goods sold. The requirement for pre-functionalized starting materials, such as biphenyl derivatives, further restricts structural diversity and increases the complexity of the supply chain, making it difficult for manufacturers to respond agilely to market demands for novel analogues.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN115974761A utilizes a highly efficient domino strategy that constructs the carbazole core from simple, readily available indole and chloroketone precursors. This novel approach eliminates the need for pre-functionalization and allows for the direct assembly of complex polycarbonyl architectures through a cascade of transformations including indole vinylation, Diels-Alder cycloaddition, dehydroaromatization, and C-H oxidation. The reaction proceeds smoothly under relatively mild thermal conditions, typically between 70°C and 130°C, using a combination of palladium catalysts and copper oxidants. This shift towards a convergent synthesis strategy significantly reduces the number of isolation steps required, thereby minimizing material loss and solvent consumption. By enabling the use of structurally diverse starting materials, this method empowers chemists to rapidly generate libraries of carbazole derivatives for biological screening or material science applications.
Mechanistic Insights into Pd-Catalyzed Domino Cyclization
The success of this synthetic route hinges on a meticulously orchestrated catalytic cycle that begins with the activation of the indole derivative. The palladium catalyst, optimally palladium acetate, facilitates the initial dehydrochlorination and vinylation of the indole with the beta-chloroketone derivative, generating a reactive alkenylindole intermediate in situ. This intermediate then undergoes a [4+2] Diels-Alder cycloaddition with the gamma-chloroketone derivative, forming the foundational carbocyclic ring of the carbazole system. Subsequent dehydroaromatization and C-H oxidation steps, driven by the copper oxidant and TEMPO additive, finalize the aromatic system and install the requisite carbonyl functionalities. Understanding this mechanistic pathway is crucial for process chemists aiming to optimize reaction parameters, as the balance between the rates of vinylation and cycloaddition determines the overall efficiency and impurity profile of the final product.
From an impurity control perspective, the mild alkaline conditions and the specific choice of additives play a pivotal role in ensuring high product purity. The use of TEMPO as a radical scavenger and oxidation mediator helps suppress side reactions that could lead to polymerization or over-oxidation of the sensitive ketone moieties. Additionally, the selection of a mixed solvent system, specifically N,N-dimethylformamide and toluene, enhances the solubility of the intermediates and stabilizes the transition states involved in the domino sequence. This careful tuning of the reaction environment minimizes the formation of by-products, reducing the burden on downstream purification processes such as column chromatography. For a reliable polycarbonyl carbazole derivative supplier, mastering these mechanistic nuances is essential to delivering consistent quality at scale, ensuring that the final API intermediates meet stringent regulatory specifications without requiring excessive reprocessing.
How to Synthesize Polycarbonyl Carbazole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized protocols established in the patent data to ensure reproducibility and safety. The process involves charging a pressure vessel with the indole substrate, the two distinct chloroketone coupling partners, and the catalytic system under an air atmosphere, which simplifies the operational setup by avoiding inert gas lines. The reaction mixture is then heated to the target temperature, typically around 120°C, and stirred for a duration of 12 to 24 hours to allow the domino cascade to reach completion. Detailed standard operating procedures regarding reagent addition order, temperature ramping rates, and workup protocols are critical for maintaining batch-to-batch consistency. For those interested in the precise stoichiometric ratios and specific handling instructions, the detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining indole derivatives, beta-chloroketone derivatives, and gamma-chloroketone derivatives in a solvent system of DMF and toluene.
- Add the palladium catalyst (preferably Pd(OAc)2), copper oxidant, TEMPO additive, and sodium acetate base to the mixture under air atmosphere.
- Heat the reaction mixture to 70-130°C for 12-24 hours, then isolate the product via silica gel column chromatography and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant cost reduction in pharmaceutical intermediate manufacturing achieved through the use of commodity chemicals. Indole derivatives and chloroketones are widely available from bulk chemical suppliers, eliminating the reliance on exotic or custom-synthesized starting materials that often create supply bottlenecks. Furthermore, the one-pot nature of the reaction drastically simplifies the manufacturing workflow, reducing the labor hours and equipment occupancy time associated with multi-step syntheses. This operational efficiency translates directly into lower overhead costs and improved throughput, allowing manufacturers to offer more competitive pricing to their downstream clients without compromising on margin.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps and the use of a robust palladium/copper catalyst system significantly lowers the raw material and processing costs. By avoiding the need for expensive protecting group strategies or harsh reagents, the overall cost of goods is optimized, making the final carbazole derivatives more accessible for large-scale drug development projects. The high atom economy of the domino reaction ensures that a greater proportion of the input mass is converted into valuable product, minimizing waste disposal costs and maximizing resource utilization efficiency.
- Enhanced Supply Chain Reliability: Utilizing commercially available and structurally diverse starting materials enhances the resilience of the supply chain against market fluctuations. Since the reaction tolerates a wide range of substituents on the indole and ketone rings, manufacturers can easily switch between different grades or suppliers of raw materials without needing to revalidate the entire process. This flexibility reduces lead time for high-purity intermediates, ensuring that production schedules can be maintained even when specific reagents face temporary shortages, thereby guaranteeing continuity of supply for critical pharmaceutical programs.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, operating at moderate temperatures and pressures that are compatible with standard stainless steel reactors found in most fine chemical facilities. The use of common solvents like DMF and toluene simplifies solvent recovery and recycling processes, aligning with modern green chemistry principles and environmental regulations. The simplified workup procedure, often requiring only filtration and recrystallization after chromatography, reduces the volume of hazardous waste generated, facilitating easier compliance with increasingly strict environmental discharge standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these polycarbonyl carbazole derivatives. The answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing pipelines. We encourage potential partners to review these insights to fully appreciate the strategic value this synthesis method brings to the table.
Q: What are the advantages of this domino reaction over traditional carbazole synthesis methods?
A: Unlike traditional methods like Graebe-Ullmann which require harsh conditions and long steps, this novel domino reaction operates under mild alkaline conditions with simple starting materials, offering higher yields and broader substrate scope.
Q: What catalyst system is optimal for this synthesis?
A: The patent identifies palladium acetate (Pd(OAc)2) combined with copper acetate hydrate as the oxidant and TEMPO as an additive as the optimal catalyst system, providing superior yields compared to other palladium salts.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available raw materials and standard solvents like DMF and toluene, with reaction temperatures between 70-130°C, making it highly scalable for industrial production without requiring extreme pressure or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycarbonyl Carbazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd-catalyzed domino reaction for the next generation of therapeutic agents and functional materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of carbazole derivatives we produce. We understand that in the fast-paced world of drug development, time is of the essence, and our team is committed to accelerating your timeline through expert process optimization and rapid scale-up capabilities.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific application needs. Whether you require custom analogues for SAR studies or metric tons of API intermediates for clinical trials, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your project. We are eager to provide you with specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can drive value and efficiency in your supply chain.
