Advanced Pd(II)-Catalyzed Synthesis of Coumarin-Pyrrole Compounds for Commercial Scale-Up
Advanced Pd(II)-Catalyzed Synthesis of Coumarin-Pyrrole Compounds for Commercial Scale-Up
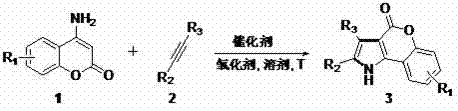
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly pyrrole derivatives which serve as critical scaffolds in numerous bioactive molecules. A significant breakthrough in this domain is detailed in patent CN103232462A, which discloses a novel synthetic method for coumarin-pyrrole compounds. This technology leverages a Palladium(II)-catalyzed oxidative cycloaddition strategy, utilizing 4-aminocoumarin compounds and disubstituted acetylene compounds as primary building blocks. By employing a unique 'lock'-auxiliary trans-enamine approach, this process circumvents the need for complex directing groups often required in traditional C-H activation strategies. For R&D directors and procurement specialists, this represents a pivotal shift towards more atom-economical and operationally simple pathways for generating high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrole derivatives has relied heavily on classical named reactions such as the Knorr, Paal-Knorr, and Hantzsch reactions. While these methods have served the industry for decades, they are increasingly viewed as suboptimal for modern green chemistry standards due to several inherent drawbacks. Traditional routes often necessitate severe reaction conditions, including high temperatures and strong acidic or basic environments, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, these classical methods frequently suffer from low atom economy and require multi-step sequences to install necessary substituents, thereby increasing waste generation and processing time. In the context of transition metal-catalyzed approaches, earlier iterations often depended on Rhodium(III) or Ruthenium(III) catalysts coupled with specific directing groups (DG) to achieve regioselectivity, adding significant cost and synthetic complexity to the overall manufacturing process.
The Novel Approach
The methodology presented in the referenced patent introduces a paradigm shift by utilizing a 'lock'-auxiliary strategy that eliminates the need for pre-installed directing groups. This innovative approach facilitates a direct oxidative coupling between the active C-H bond of the aromatic ring and the alkyne substrate. As illustrated in the comparative scheme below, this bypasses the laborious steps associated with DG installation and removal.
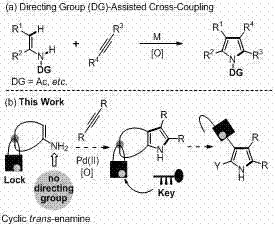
By employing 4-aminocoumarins as the starting material, the reaction inherently possesses the necessary structural features to guide the cyclization without external auxiliaries. This results in a streamlined one-step synthesis that significantly reduces the number of unit operations required. The reaction conditions are notably mild, often proceeding at room temperature or with moderate heating, which enhances the safety profile and energy efficiency of the process. This simplification not only accelerates the timeline from bench to pilot plant but also drastically improves the overall yield and purity of the final coumarin-pyrrole products, addressing key pain points for supply chain managers focused on efficiency.
Mechanistic Insights into Pd(II)-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the efficient Pd(II) catalytic cycle that drives the oxidative annulation. The reaction initiates with the coordination of the palladium catalyst to the 4-aminocoumarin substrate, activating the proximal C-H bond for nucleophilic attack on the disubstituted alkyne. This is followed by a migratory insertion and subsequent reductive elimination to form the new C-C and C-N bonds characteristic of the pyrrole ring. Crucially, the presence of an oxidant, such as Copper(II) acetate or molecular oxygen, serves to regenerate the active Pd(II) species from the reduced Pd(0) state, ensuring the catalytic turnover continues efficiently. This redox-neutral or oxidative nature of the cycle allows for high turnover numbers and minimizes the accumulation of palladium black, a common issue in cross-coupling reactions that can complicate downstream purification.
From an impurity control perspective, the mildness of the reaction conditions plays a vital role in maintaining product integrity. Harsh conditions often promote polymerization of alkynes or decomposition of the coumarin lactone ring; however, this protocol operates effectively in solvents like DMSO at temperatures ranging from room temperature to 120°C. The specificity of the 'lock' mechanism ensures that the cyclization occurs regioselectively, minimizing the formation of isomeric byproducts that are difficult to separate. For quality control teams, this translates to a cleaner crude reaction mixture, reducing the burden on purification columns and crystallization steps. The broad substrate scope demonstrated in the patent, accommodating various electron-donating and electron-withdrawing groups on both the coumarin and alkyne moieties, further underscores the robustness of this mechanistic pathway for diverse chemical libraries.
How to Synthesize Coumarin-Pyrrole Compounds Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and atmospheric conditions to maximize yield and reproducibility. The standard protocol involves dissolving the 4-aminocoumarin and the disubstituted alkyne in a polar aprotic solvent such as DMSO, followed by the addition of the palladium catalyst and the copper oxidant. The reaction is typically conducted under an oxygen atmosphere to facilitate the re-oxidation of the catalyst. Detailed operational parameters, including specific molar ratios and workup procedures, are critical for achieving the high yields reported in the patent embodiments. For a comprehensive understanding of the standardized synthesis steps required for laboratory or pilot-scale execution, please refer to the structured guide below.
- Dissolve 4-aminocoumarin compounds and disubstituted acetylene compounds in a reaction solvent such as DMSO.
- Add palladium catalyst (e.g., Pd(OAc)2) and an oxidant (e.g., Cu(OAc)2) to the reaction mixture under an oxygen atmosphere.
- Stir the mixture at temperatures ranging from room temperature to 120°C until completion, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage stems from the accessibility and cost-effectiveness of the raw materials. 4-Aminocoumarins and disubstituted alkynes are commercially available commodities or can be synthesized via established, low-cost pathways. This abundance ensures a stable supply base, mitigating the risks associated with sourcing exotic or proprietary starting materials that often plague niche pharmaceutical projects. Furthermore, the elimination of multi-step protecting group manipulations and directing group installations significantly compresses the manufacturing timeline, allowing for faster response to market demands and reduced inventory holding costs.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the simplification of the process flow. By consolidating what would traditionally be a multi-step sequence into a single oxidative coupling event, manufacturers can realize substantial reductions in labor, solvent consumption, and waste disposal costs. The use of earth-abundant copper salts as co-oxidants alongside palladium further optimizes the reagent cost profile compared to systems relying solely on precious metals or stoichiometric amounts of expensive oxidants. Additionally, the high atom economy means that a greater proportion of the input mass is converted into the desired product, directly improving the cost-per-kilogram metric for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The ability to run the reaction at room temperature or with moderate heating reduces the dependency on specialized high-pressure or cryogenic equipment, making the process transferable to a wider range of CDMO partners globally. The tolerance of the catalyst system to various functional groups implies that supply disruptions for specific substituted precursors can often be managed by switching to alternative analogs without redesigning the entire synthetic route. This flexibility is crucial for maintaining continuity of supply in the face of raw material volatility.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, the process aligns well with green chemistry principles. The reduction in synthetic steps inherently lowers the E-factor (mass of waste per mass of product), simplifying wastewater treatment and solid waste management. The use of oxygen or air as a terminal oxidant in some variations produces water as the only byproduct, significantly reducing the toxicological burden of the effluent stream. This cleaner profile facilitates easier regulatory approval for commercial manufacturing sites and supports corporate sustainability goals, making the technology attractive for long-term production contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this coumarin-pyrrole synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, aiming to clarify feasibility for potential licensees or manufacturing partners. Understanding these nuances is essential for accurate project planning and risk assessment.
Q: What are the primary advantages of this Pd-catalyzed method over traditional pyrrole synthesis?
A: Unlike traditional Knorr or Paal-Knorr reactions which often suffer from harsh conditions and low atom economy, this method utilizes a 'lock'-auxiliary trans-enamine strategy allowing for direct oxidative coupling under mild conditions with high yields.
Q: Which catalysts and oxidants are compatible with this synthetic route?
A: The process effectively employs Palladium(II) acetate as the catalyst. Compatible oxidants include Copper(II) acetate, Silver acetate, benzoquinone, DDQ, or molecular oxygen, providing flexibility for process optimization.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method features cheap and readily available raw materials, simple operational procedures, and mild reaction conditions, making it highly suitable for commercial scale-up and industrial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin-Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the Pd(II)-mediated oxidative coupling described in CN103232462A. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates, guaranteeing that every batch of coumarin-pyrrole compound delivered meets the highest global standards for quality and consistency.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthetic route for their drug discovery programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and cost-effective supply chain for your critical nitrogen-heterocycle needs.
