Advanced Rhodium-Catalyzed Synthesis of 1-Aminoisoquinoline Skeletons for Pharmaceutical Applications
Advanced Rhodium-Catalyzed Synthesis of 1-Aminoisoquinoline Skeletons for Pharmaceutical Applications
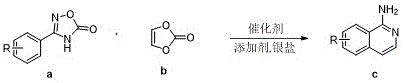
The pharmaceutical industry continuously seeks robust and efficient methodologies for constructing privileged scaffolds, particularly the 1-aminoisoquinoline core, which is prevalent in numerous bioactive molecules including thrombin inhibitors and anti-inflammatory agents. A groundbreaking approach detailed in patent CN113735770B introduces a novel rhodium-catalyzed strategy that utilizes 4-phenyl oxadiazolone compounds and vinylene carbonate to generate these valuable heterocycles. This technology represents a significant leap forward in synthetic efficiency, addressing long-standing challenges regarding reagent safety and regioselectivity. By leveraging transition metal catalysis, specifically rhodium, this method achieves high atom economy and simplifies the overall synthetic workflow. The process effectively bypasses the limitations of traditional nucleophilic substitutions, offering a streamlined pathway that is highly attractive for the development of reliable pharmaceutical intermediate suppliers. Furthermore, the versatility of this reaction allows for the incorporation of diverse functional groups, making it a powerful tool for medicinal chemists aiming to explore structure-activity relationships rapidly.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1-aminoisoquinoline skeleton has relied heavily on classical nucleophilic substitution reactions at the C-1 position, driven by the partial positive charge induced by the adjacent nitrogen atom. While conceptually straightforward, these traditional pathways suffer from significant drawbacks that hinder their utility in modern process chemistry. The preparation of the necessary starting materials is often laborious and time-consuming, requiring multi-step sequences that erode overall yield and increase production costs. Moreover, the harsh conditions frequently associated with these nucleophilic attacks can lead to poor functional group tolerance, limiting the diversity of analogs that can be synthesized. In recent years, transition metal-catalyzed C-H activation has emerged as a promising alternative; however, existing methods predominantly utilize substituted alkynes as coupling partners. This reliance on alkynes presents a critical bottleneck, as it often fails to produce the widely desired 3,4-unsubstituted 1-aminoisoquinoline derivatives, thereby restricting the chemical space accessible to researchers and complicating the supply chain for specific API intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN113735770B employs vinylene carbonate as a superior two-carbon donor, effectively replacing hazardous and less versatile alkynes. This innovative strategy utilizes a 4-phenyl oxadiazolone precursor, where the oxadiazolone ring serves a dual purpose: it acts as a directing group for C-H activation and simultaneously protects the sensitive primary amino functionality. Under the influence of a rhodium catalyst, the system undergoes a seamless cyclization to form the isoquinoline ring, followed by the spontaneous extrusion of carbon dioxide to unveil the free amine. This approach not only simplifies the synthetic operation to a single pot but also dramatically enhances safety profiles by avoiding unstable acetylene derivatives. The result is a highly efficient, atom-economical process that delivers high-purity 1-aminoisoquinoline products with exceptional ease of purification, setting a new standard for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this transformative synthesis lies in the sophisticated interplay between the rhodium catalyst and the oxadiazolone substrate. The mechanism initiates with the coordination of the rhodium center to the nitrogen or oxygen atoms within the oxadiazolone ring, which directs the metal to the proximal ortho-C-H bond on the phenyl ring. This directed C-H activation step generates a stable rhodacycle intermediate, a crucial species that dictates the regioselectivity of the entire transformation. Subsequently, the vinylene carbonate inserts into the rhodium-carbon bond, facilitated by the unique electronic properties of the cyclic carbonate which acts as an alkyne equivalent. This insertion event is followed by a reductive elimination or similar cyclization step that constructs the fused isoquinoline framework. The elegance of this mechanism is further highlighted by the role of the silver salt additives, which likely assist in halide abstraction to generate the active cationic rhodium species, thereby accelerating the catalytic turnover and ensuring high conversion rates even with sterically demanding substrates.
Beyond the formation of the carbon skeleton, the management of the amino protecting group is pivotal to the success of this route. The oxadiazolone ring is ingeniously designed to be labile under the reaction conditions, acting as a "mask" for the primary amine. Once the cyclization is complete, the thermodynamic drive to release carbon dioxide triggers the fragmentation of the oxadiazole moiety. This decarboxylation step is spontaneous and irreversible, cleanly exposing the reactive primary aryl amino group without the need for additional deprotection reagents or harsh acidic/basic workups. This intrinsic self-immolative feature significantly reduces the generation of chemical waste and simplifies the downstream processing. For R&D directors focused on impurity control, this mechanism offers a distinct advantage by minimizing side reactions associated with external deprotection steps, thereby ensuring a cleaner impurity profile and facilitating easier compliance with stringent regulatory standards for drug substances.
How to Synthesize 1-Aminoisoquinoline Efficiently
Implementing this rhodium-catalyzed protocol requires careful attention to reaction parameters to maximize yield and reproducibility. The process typically involves charging a sealed vessel with the 4-aryl oxadiazolone substrate, vinylene carbonate, a selected rhodium catalyst such as dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer, and a silver salt oxidant. An organic solvent system, potentially containing water or trifluoroethanol, is added along with a carboxylic acid additive like pivalic acid to promote the C-H activation step. The detailed standardized synthesis steps, including precise molar ratios and specific temperature ramps optimized for different substrates, are outlined in the guide below to ensure successful replication in your laboratory.
- Prepare the reaction mixture by combining 4-aryl oxadiazolone, vinylene carbonate, a rhodium catalyst (e.g., [Cp*RhCl2]2), a silver salt additive, and a carboxylic acid additive in a suitable solvent system.
- Heat the sealed reaction vessel to a temperature between 100°C and 140°C under an inert or air atmosphere, maintaining stirring for a duration of 8 to 24 hours to ensure complete conversion.
- Upon completion, remove the solvent under reduced pressure and purify the resulting crude residue via flash column chromatography using ethyl acetate and petroleum ether to isolate the high-purity 1-aminoisoquinoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The shift from complex multi-step nucleophilic substitutions to a direct catalytic cyclization fundamentally alters the cost structure of producing 1-aminoisoquinoline derivatives. By eliminating the need for pre-functionalized alkyne reagents and cumbersome protection-deprotection sequences, the process drastically reduces the consumption of raw materials and solvents. This simplification directly correlates to substantial cost savings in manufacturing, as fewer unit operations are required to reach the final API intermediate. Furthermore, the use of vinylene carbonate, a stable and commercially abundant reagent, mitigates supply risks associated with specialized or hazardous alkynes, ensuring a more resilient and continuous supply chain for critical pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic impact of this synthesis route is profound, primarily driven by the high atom economy and the reduction in processing steps. Traditional methods often incur high costs due to the purchase of expensive substituted alkynes and the disposal of stoichiometric byproducts generated during nucleophilic substitutions. In contrast, this rhodium-catalyzed method utilizes vinylene carbonate, which is not only cheaper but also reacts with high efficiency, minimizing waste. The elimination of separate deprotection steps further lowers operational expenditures by reducing energy consumption and labor hours. Consequently, manufacturers can achieve a significantly lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche reagents that have limited suppliers or long lead times. This new methodology addresses this vulnerability by utilizing vinylene carbonate and simple oxadiazolone precursors, both of which are readily available from bulk chemical suppliers. The robustness of the reaction conditions, which tolerate air and moisture better than many sensitive organometallic processes, also reduces the risk of batch failures due to environmental factors. This reliability ensures that production schedules can be met consistently, reducing lead time for high-purity intermediates and preventing costly delays in downstream drug development programs. The ability to source materials locally and store them safely adds an extra layer of security to the procurement strategy.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden complexities, but this protocol is designed with scalability in mind. The reaction operates at moderate temperatures and uses common organic solvents, making it compatible with existing stainless steel reactor infrastructure. The simplified workup, involving basic solvent evaporation and chromatography, avoids the generation of heavy metal waste streams often associated with other transition metal catalyses, as the rhodium loading is low and the catalyst can potentially be recovered. This aligns with increasingly strict environmental regulations, reducing the burden of waste treatment and enhancing the sustainability profile of the manufacturing process. The ease of scale-up facilitates the commercial production of complex heterocycles, ensuring that supply can meet demand as clinical programs advance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology can be integrated into existing workflows. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: What are the primary advantages of using vinylene carbonate over substituted alkynes in this synthesis?
A: Vinylene carbonate serves as a safer, more stable, and easily stored two-carbon donor compared to traditional substituted alkynes. It allows for the efficient synthesis of 3,4-unsubstituted 1-aminoisoquinoline compounds, which are difficult to access via conventional alkyne-based C-H activation methods, while simultaneously improving atom economy.
Q: How does the oxadiazolone directing group facilitate the reaction mechanism?
A: The oxadiazolone moiety acts as a weak nitrogen-oxygen bond shield for the strong coordinating primary aryl amino group. This protection prevents catalyst poisoning and directs the rhodium catalyst to the specific C-H bond for activation. Following cyclization, the oxadiazole ring spontaneously extrudes carbon dioxide to reveal the active primary amino group.
Q: Is this synthetic route scalable for industrial production of API intermediates?
A: Yes, the method is highly feasible for industrial scale-up due to its simple operational steps, broad substrate scope, and straightforward post-treatment procedures. The use of commercially available reagents and the elimination of complex nucleophilic substitution steps significantly enhance its viability for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aminoisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN113735770B for accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from gram-scale research to industrial manufacturing is seamless. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that validate every batch against the highest international standards. We understand that the consistency of your API intermediates is critical to the success of your final drug product, and our state-of-the-art facilities are equipped to handle the specific requirements of rhodium-catalyzed processes with precision and safety.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to our deep expertise in process optimization and supply chain management. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in delivering high-quality 1-aminoisoquinoline derivatives efficiently and reliably.
