Scalable Visible Light Synthesis of Trans-Olefins for Pharmaceutical Intermediates
Scalable Visible Light Synthesis of Trans-Olefins for Pharmaceutical Intermediates
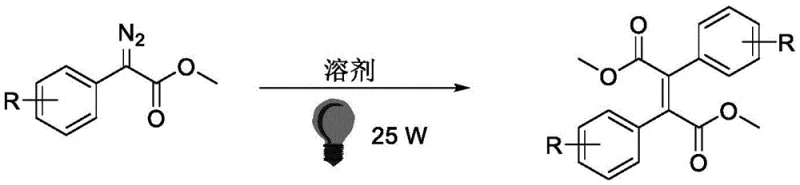
The landscape of organic synthesis is undergoing a transformative shift towards greener, more efficient methodologies, particularly in the production of complex pharmaceutical intermediates. A groundbreaking development in this field is detailed in patent CN115368238A, which discloses a novel visible light-induced method for synthesizing trans-olefins from α-aryl diazoacetic acid methyl esters. This technology represents a significant departure from traditional thermal or metal-catalyzed pathways, offering a streamlined route to tetra-substituted trans-olefin derivatives. By leveraging the energy of blue LED irradiation at room temperature, this process achieves exceptional stereoselectivity without the need for external photocatalysts. For R&D directors and procurement strategists, this innovation signals a new era of cost-effective and environmentally compliant manufacturing for high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted olefins via diazo compounds has relied heavily on transition metal catalysis or harsh thermal conditions. Conventional protocols often necessitate the use of expensive noble metal photocatalysts such as Ru(bpy)3 2+, Ir(ppy)3, or Eosin Y to facilitate the decomposition of diazo precursors into reactive carbenes. These metal-based systems introduce significant logistical and financial burdens, including the high cost of the catalysts themselves and the complex downstream processing required to remove trace metal residues to meet stringent pharmaceutical purity standards. Furthermore, existing metal-free photochemical methods have struggled with poor stereoselectivity, often yielding E/Z ratios as low as 20:1, which complicates purification and reduces overall process efficiency. The reliance on thermal activation also poses safety risks due to the potential instability of diazo compounds at elevated temperatures.
The Novel Approach
In stark contrast, the methodology described in patent CN115368238A eliminates the need for any external photocatalyst or transition metal promoter. By utilizing direct irradiation with a blue LED lamp (450-460 nm), the α-aryl diazoacetic acid methyl ester absorbs photons directly to reach an excited state, triggering the release of nitrogen and the formation of a reactive free carbene intermediate. This catalyst-free approach not only drastically reduces raw material costs but also simplifies the reaction workup, as there are no metal complexes to quench or extract. The process operates under remarkably mild conditions—specifically at room temperature in dichloromethane—which enhances operational safety and energy efficiency. Most critically, this visible light-induced dimerization delivers superior stereocontrol, consistently producing the desired trans-olefin (E-isomer) with an E/Z ratio exceeding 100:1, thereby setting a new benchmark for selectivity in this chemical class.
Mechanistic Insights into Visible Light-Induced Carbene Dimerization
The mechanistic elegance of this transformation lies in the direct photo-excitation of the substrate. Upon exposure to blue light, the α-aryl diazoacetic acid methyl ester undergoes a homolytic cleavage of the carbon-nitrogen bond, expelling a molecule of nitrogen gas to generate a highly electrophilic free carbene species. Unlike metal-carbenoids which are stabilized by coordination to a metal center, this free carbene is transient and highly reactive. The electron-withdrawing nature of the adjacent ester group stabilizes the carbene sufficiently to allow for a controlled intermolecular reaction. A second molecule of the starting diazo compound, acting as a nucleophile through its negatively polarized carbon atom, attacks this free carbene intermediate. This nucleophilic addition forms a new carbon-carbon bond, followed by the subsequent extrusion of a second nitrogen molecule, ultimately yielding the thermodynamically stable E-configured tetra-substituted olefin. This pathway avoids the steric and electronic constraints often imposed by bulky ligand spheres in metal catalysis, allowing for a more direct and selective bond formation.
From an impurity control perspective, the absence of metal catalysts fundamentally alters the impurity profile of the final product. In traditional metal-catalyzed processes, side reactions often include metal-mediated C-H insertions or cyclopropanations, leading to complex mixtures that are difficult to separate. In this photocatalyst-free system, the primary byproduct is nitrogen gas, which harmlessly vents from the reaction vessel. The high stereoselectivity (E/Z > 100:1) indicates that the reaction trajectory is strongly biased towards the trans-configuration, likely due to steric repulsion between the bulky aryl-ester groups in the transition state favoring the anti-arrangement. This inherent selectivity minimizes the formation of the Z-isomer, reducing the burden on purification teams and ensuring a cleaner crude product profile that is easier to bring to specification.
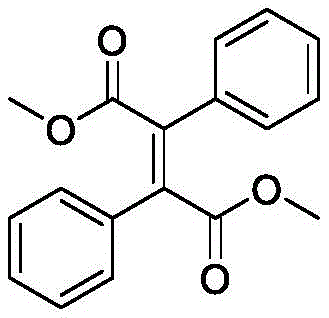
How to Synthesize Dimethyl 2,3-Diphenyl Fumarate Efficiently
The practical implementation of this synthesis is designed for ease of adoption in both laboratory and pilot-scale settings. The protocol utilizes standard glassware and commercially available light sources, removing the barrier to entry for specialized photochemical reactors. The process begins by dissolving the α-aryl diazoacetic acid methyl ester substrate in dichloromethane, a common and effective solvent for this transformation. The reaction mixture is then subjected to blue LED irradiation at ambient temperature with continuous magnetic stirring. Monitoring via TLC typically indicates completion within one hour, after which the solvent is removed under reduced pressure. The resulting crude solid is purified via standard silica gel column chromatography to afford the pure trans-olefin derivative. For detailed standardized operating procedures and specific stoichiometric ratios, please refer to the guide below.
- Dissolve alpha-aryl diazoacetic acid methyl ester in dichloromethane within a glass or quartz reaction vessel.
- Irradiate the reaction mixture with a blue LED lamp (450-460 nm) at room temperature with magnetic stirring for approximately 1 hour.
- Remove the solvent under reduced pressure and purify the crude residue via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this visible light-induced synthesis offers compelling economic and logistical advantages over legacy technologies. The most immediate impact is the elimination of costly noble metal catalysts, which directly translates to a significant reduction in the Bill of Materials (BOM) cost. Furthermore, the removal of metal catalysts obviates the need for expensive scavenging resins or complex extraction protocols designed to lower heavy metal content to ppm levels, thereby streamlining the manufacturing workflow and reducing waste disposal costs. The mild reaction conditions also imply lower energy consumption compared to thermal processes that require heating or cooling cycles, contributing to a smaller carbon footprint and lower utility expenses. These factors combined create a robust business case for switching to this newer methodology for large-scale production.
- Cost Reduction in Manufacturing: The catalyst-free nature of this process removes the dependency on volatile and expensive precious metals like Ruthenium or Iridium, which are subject to significant market price fluctuations. By relying solely on electrical energy from LEDs and inexpensive organic solvents, manufacturers can achieve substantial cost savings in raw material procurement. Additionally, the simplified workup procedure reduces labor hours and consumable usage associated with metal removal steps, further driving down the cost of goods sold (COGS) for these high-value intermediates.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can sometimes be a bottleneck due to geopolitical supply constraints or mining limitations. This new method relies on readily available organic starting materials and standard LED equipment, which are abundant and easily sourced globally. This diversification of supply inputs mitigates the risk of production delays caused by catalyst shortages. Moreover, the stability of the reagents at room temperature simplifies storage and transportation logistics, reducing the need for specialized cold-chain infrastructure.
- Scalability and Environmental Compliance: The operational simplicity of this photochemical reaction makes it highly amenable to scale-up. The use of flow chemistry or large batch reactors equipped with LED arrays allows for seamless transition from gram to kilogram scales without re-optimizing complex catalytic parameters. From an environmental standpoint, the process generates nitrogen gas as the only stoichiometric byproduct, aligning with green chemistry principles. The absence of heavy metal waste simplifies regulatory compliance and wastewater treatment, facilitating smoother audits and faster time-to-market for pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method operates under catalyst-free conditions. It utilizes direct visible light irradiation to activate the diazo compound, eliminating the need for costly ruthenium or iridium photocatalysts and avoiding heavy metal contamination in the final product.
Q: What is the stereoselectivity of the resulting olefins?
A: The process demonstrates exceptional stereoselectivity, consistently yielding the E-isomer (trans-olefin) with an E/Z ratio greater than 100:1. This high selectivity simplifies downstream purification and ensures consistent quality for pharmaceutical applications.
Q: What are the typical reaction conditions required?
A: The reaction proceeds under mild conditions at room temperature using dichloromethane as the solvent. It requires only a standard blue LED light source (450-460 nm) and completes within approximately 1 hour, making it energy-efficient and operationally simple.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Olefins Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free visible light technology for the production of high-purity pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, including the precise control of E/Z isomeric ratios required for advanced drug synthesis. We are committed to delivering consistent quality and supply continuity for complex fine chemicals.
We invite forward-thinking R&D and procurement teams to collaborate with us on leveraging this efficient synthesis route for your next project. By partnering with us, you gain access to our technical expertise in process optimization and a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation for your trans-olefin intermediate needs.
