Advanced Pd-Catalyzed Synthesis of Isocoumarin Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for heterocyclic scaffolds, particularly isocoumarins, which serve as vital cores for anticancer and anti-inflammatory agents. Patent CN108329289B introduces a transformative methodology for synthesizing these valuable drug intermediates, specifically targeting the conversion of epoxy-nitrile precursors into 3-arylisocoumarins. This technical breakthrough leverages a specialized palladium-catalyzed cyclization strategy that operates under relatively mild acidic conditions, distinguishing itself from conventional base-mediated protocols. For R&D directors and process chemists, this patent represents a significant opportunity to streamline the production of complex fused heterocycles while maintaining high purity standards essential for regulatory compliance. The method's reliance on accessible starting materials and a defined catalytic system suggests strong potential for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isocoumarin skeleton has relied heavily on copper-catalyzed coupling reactions or multi-step sequences involving harsh reagents. Prior art, such as methods utilizing copper salts with strong bases, often suffers from limited functional group tolerance and the generation of significant metallic waste. These traditional routes frequently require stringent anhydrous conditions and elevated temperatures that can degrade sensitive substituents on the aromatic rings. Furthermore, the removal of residual copper from the final API intermediate poses a persistent challenge for supply chain managers, necessitating expensive scavenging resins and additional purification steps that inflate overall manufacturing costs. The variability in yield across different substrates in these older methods also complicates scale-up efforts, leading to inconsistent batch quality.
The Novel Approach
In stark contrast, the methodology disclosed in CN108329289B utilizes a palladium-catalyzed intramolecular cyclization that proceeds efficiently in a mixed aqueous-organic solvent system. By employing specific epoxy-nitrile precursors, the reaction achieves ring closure through a mechanism that tolerates a wide range of electronic environments on the aryl rings. The use of methanesulfonic acid as a promoter, rather than a strong base, fundamentally alters the reaction landscape, minimizing side reactions such as polymerization or hydrolysis that plague basic conditions. This approach not only simplifies the operational protocol but also enhances the safety profile of the manufacturing process by avoiding pyrophoric reagents. The ability to utilize a 1,4-dioxane and water mixture further underscores the method's practicality, as it balances solubility requirements with environmental considerations.
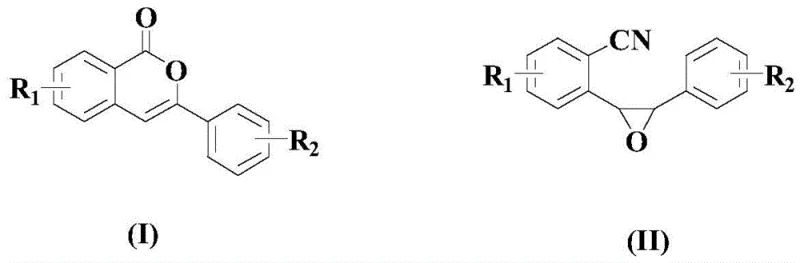
The visual representation of this transformation highlights the elegance of converting the strained epoxide ring directly into the lactone functionality of the isocoumarin core. This direct cyclization eliminates the need for intermediate isolation steps that are common in linear syntheses, thereby reducing the overall process mass intensity. For procurement teams, this efficiency translates into reduced solvent consumption and lower waste disposal fees, contributing to substantial cost reduction in pharmaceutical manufacturing. The robustness of this catalytic system across various substituted substrates ensures that supply chain continuity can be maintained even when specific raw material grades vary slightly, providing a buffer against market volatility.
Mechanistic Insights into Pd-Catalyzed Cyclization
The mechanistic pathway likely involves the initial coordination of the palladium species to the nitrile or the epoxide oxygen, facilitating ring opening and subsequent nucleophilic attack by the carbonyl oxygen or an internal nucleophile generated in situ. The presence of the acidic additive plays a crucial role in activating the epoxide towards nucleophilic attack while stabilizing the cationic palladium intermediates. This delicate balance between Lewis acidity and transition metal catalysis is key to the high yields observed, often exceeding 85% in optimized examples. Understanding this mechanism allows process chemists to fine-tune reaction parameters such as temperature and catalyst loading to maximize throughput without compromising product integrity. The specific requirement for Pd(OAc)2 over other palladium sources suggests that the acetate ligand may participate in the proton transfer steps or assist in the regeneration of the active catalytic species.
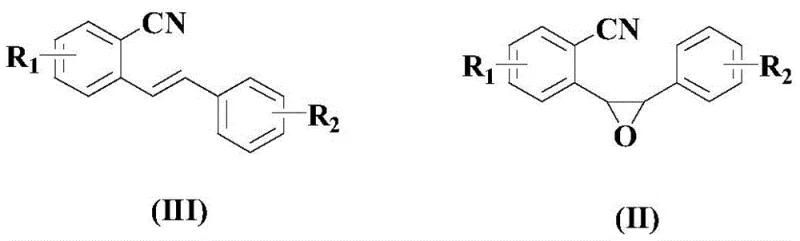
Prior to the cyclization event, the synthesis of the epoxy-nitrile precursor (Formula II) from the corresponding chalcone derivative (Formula III) is achieved via epoxidation using m-CPBA. This upstream step is equally critical, as the stereochemistry and purity of the epoxide directly influence the outcome of the cyclization. The use of m-CPBA ensures high selectivity for the alkene oxidation without affecting the nitrile group, preserving the functionality required for the subsequent palladium step. This two-step sequence from readily available chalcones offers a modular approach to library synthesis, enabling rapid access to diverse isocoumarin analogs for biological screening. The compatibility of this precursor synthesis with the downstream cyclization creates a seamless telescoped process potential, further enhancing the economic viability of the route for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Arylisocoumarin Efficiently
To implement this synthesis effectively, operators must adhere to strict control over the solvent composition and acid concentration, as deviations can lead to complete reaction failure. The process begins with the preparation of the epoxy-nitrile substrate, followed by its dissolution in the specific 1,4-dioxane and water mixture. The addition of the palladium catalyst and methanesulfonic acid must be performed under controlled conditions to ensure homogeneous mixing before heating. Detailed standardized synthetic steps see the guide below for precise stoichiometric ratios and temperature profiles that guarantee reproducible results. Adhering to these parameters is essential for maintaining the high purity specifications required for downstream drug substance manufacturing.
- Prepare the epoxy-nitrile precursor (Formula II) by reacting a chalcone derivative (Formula III) with m-CPBA in dichloromethane at 20-40°C.
- Conduct the cyclization reaction by heating the precursor (Formula II) with a palladium catalyst (e.g., Pd(OAc)2) and methanesulfonic acid in a 1,4-dioxane/water mixture at 60-120°C.
- Perform post-reaction workup involving extraction with ethyl acetate, washing with bicarbonate and brine, drying, and purification via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology addresses several critical pain points associated with the sourcing of heterocyclic building blocks. The shift towards a palladium-catalyzed acidic protocol reduces the dependency on specialized base-stable equipment and minimizes the corrosion risks associated with strong alkaline processes. This operational simplicity allows for the utilization of standard glass-lined reactors, which are widely available in contract manufacturing organizations, thereby enhancing supply chain reliability. Moreover, the high atom economy of the cyclization step means that less raw material is wasted as byproducts, directly impacting the cost of goods sold. For procurement managers, the ability to source high-purity intermediates with a consistent impurity profile reduces the burden on quality control laboratories and accelerates batch release times.
- Cost Reduction in Manufacturing: The elimination of multiple protection-deprotection steps and the use of a single catalytic system for ring closure significantly lowers the operational expenditure. By avoiding the need for expensive ligand systems or exotic reagents often found in alternative cross-coupling methods, the overall material cost is optimized. The simplified workup procedure, which relies on standard aqueous washes and crystallization or chromatography, reduces solvent usage and energy consumption during distillation. These cumulative efficiencies drive down the unit cost of the intermediate, making it a more attractive option for large-scale production campaigns without sacrificing quality.
- Enhanced Supply Chain Reliability: The starting materials, specifically the chalcone derivatives and m-CPBA, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions means that minor fluctuations in raw material quality do not necessarily derail the production schedule, ensuring consistent delivery timelines. This resilience is crucial for maintaining the continuity of supply for downstream API manufacturers who operate on tight just-in-time schedules. Furthermore, the scalability of the process from gram to kilogram scales has been demonstrated, providing confidence for long-term supply agreements.
- Scalability and Environmental Compliance: The use of a water-containing solvent system aligns with green chemistry principles by reducing the volume of volatile organic compounds emitted during the process. The acidic nature of the waste stream is easier to neutralize and treat compared to heavy metal-laden basic waste from copper-catalyzed processes. This environmental advantage simplifies regulatory compliance and reduces the costs associated with waste disposal and environmental monitoring. As sustainability metrics become increasingly important in vendor selection, this process offers a competitive edge for suppliers aiming to meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these nuances is vital for process engineers looking to adapt this technology for their specific manufacturing contexts. The answers reflect the critical success factors identified during the development of this methodology.
Q: Why is Palladium Acetate preferred over other Pd catalysts for this synthesis?
A: Experimental data indicates that Palladium Acetate (Pd(OAc)2) provides significantly higher yields (over 85%) compared to other palladium sources like PdCl2 or Pd(PPh3)4, which often result in sharp yield reductions due to ligand effects or solubility issues.
Q: What is the critical role of the solvent system in this reaction?
A: The mixture of 1,4-dioxane and water is essential; using single-component organic solvents or other aqueous mixtures leads to negligible product formation, suggesting a specific solvent effect is required for the catalytic cycle to proceed efficiently.
Q: How does this method compare to traditional copper-catalyzed routes?
A: Unlike traditional copper-catalyzed methods that often require strong bases and harsh conditions, this Pd-catalyzed approach utilizes acidic additives and milder temperatures, offering better functional group tolerance and simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylisocoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN108329289B for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the plant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of isocoumarin intermediate meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of palladium catalysis and deliver products with consistent quality profiles.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to optimize your manufacturing process and secure a reliable supply of high-value pharmaceutical intermediates.
