Advanced Rhodium-Catalyzed Synthesis of O-Alkenyl Aromatic Nitriles for Commercial Scale-up
Introduction to Next-Generation Nitrile Synthesis
The field of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical and material science applications, particularly regarding the efficient construction of functionalized aromatic cores. Patent CN110845363B introduces a groundbreaking methodology for the preparation of o-alkenyl aromatic nitrile compounds, addressing significant bottlenecks in traditional synthetic routes. This technology leverages a sophisticated rhodium-catalyzed oxidative Heck reaction, utilizing aryl imidate derivatives as versatile precursors to access highly valuable nitrile-containing scaffolds. The strategic use of imidates allows for the circumvention of electronic deactivation issues typically associated with direct cyanation, enabling precise regioselective functionalization at the ortho-position. This innovation represents a pivotal shift in how chemists approach the synthesis of complex heterocyclic and carbocyclic systems, offering a streamlined pathway that integrates C-H activation with subsequent functional group transformation.
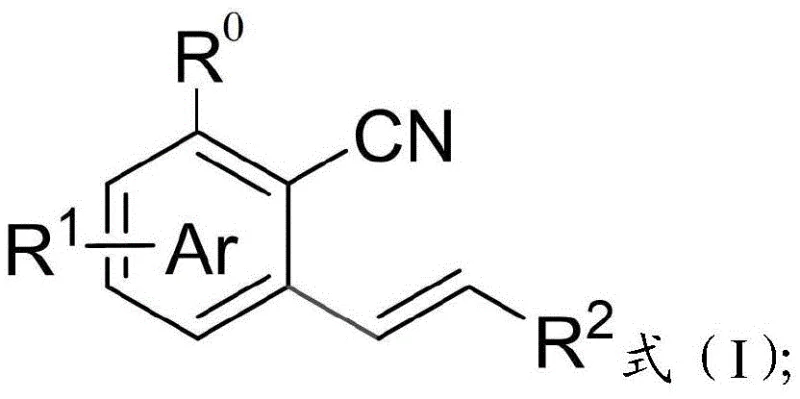
Furthermore, the versatility of this synthetic platform extends beyond simple benzene derivatives to include fused ring systems like naphthalene and heterocycles such as indoles and thiophenes. The ability to introduce both cyano and alkenyl functionalities in a single operational sequence significantly enhances step economy, a critical metric for industrial process development. By establishing a reliable protocol that operates under relatively mild oxidative conditions, this patent provides a robust foundation for the scalable production of intermediates used in the manufacture of active pharmaceutical ingredients (APIs) and advanced functional materials. For procurement and R&D teams alike, understanding the mechanistic underpinnings and practical advantages of this route is essential for evaluating its potential integration into existing supply chains and manufacturing workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of ortho-alkenyl aromatic nitriles has been plagued by inherent chemical challenges stemming from the electronic nature of the cyano group. As a strong electron-withdrawing substituent, the nitrile functionality significantly deactivates the aromatic ring towards electrophilic attack, making direct oxidative Heck reactions on benzonitrile substrates exceptionally difficult and often low-yielding. Conventional strategies frequently rely on the use of ortho-halo substituted aromatic nitriles as starting materials, which necessitates multi-step synthetic sequences involving halogenation followed by palladium-catalyzed cross-coupling. This approach not only increases the overall cost of goods due to the requirement for expensive halogenated precursors but also generates substantial stoichiometric waste. Moreover, the strong coordination ability of the cyano group can poison metal catalysts or lead to undesired linear coordination modes, further complicating the achievement of high ortho-selectivity. These factors collectively result in complex, inefficient processes that are ill-suited for the rapid iteration required in modern drug discovery and process optimization.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes aryl imidate esters as masked nitrile precursors, effectively bypassing the electronic deactivation barrier. The imidate group serves a dual purpose: it acts as a robust directing group to facilitate ortho-C-H activation via coordination to the rhodium center, and it can be subsequently hydrolyzed to reveal the desired cyano functionality. This strategy enables the direct coupling of readily available terminal alkenes with simple aromatic substrates under oxidative conditions. The reaction proceeds with high efficiency and exceptional regioselectivity, driven by the协同 metalation-deprotonation (CMD) mechanism assisted by the imine nitrogen lone pair. By shifting the synthetic logic from pre-functionalized halides to direct C-H bond functionalization, this method drastically simplifies the synthetic route, reduces raw material costs, and minimizes waste generation. This paradigm shift offers a compelling value proposition for manufacturers seeking to optimize their production of high-purity pharmaceutical intermediates.
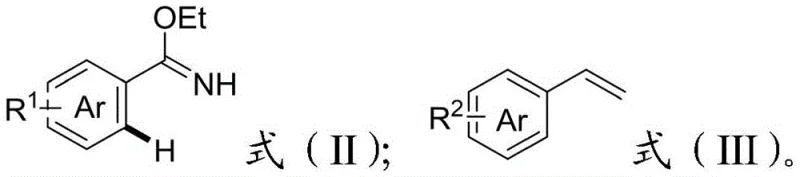
Mechanistic Insights into Rhodium-Catalyzed Oxidative Heck Reaction
The core of this technological advancement lies in the intricate catalytic cycle mediated by trivalent rhodium species, which orchestrates the selective activation of inert carbon-hydrogen bonds. The mechanism initiates with the coordination of the rhodium catalyst to the sp2-hybridized nitrogen atom of the imidate functional group, forming a stable pi-complex that brings the metal center into close proximity with the ortho-C-H bond. Under the promotion of a base such as sodium acetate, the system undergoes a concerted metalation-deprotonation (CMD) process, cleaving the C-H bond to generate a five-membered rhodacycle intermediate. This organometallic species is the key determinant of the reaction's high regioselectivity, ensuring that functionalization occurs exclusively at the position adjacent to the directing group. Subsequently, the terminal alkene substrate coordinates to the metal center and undergoes migratory insertion into the rhodium-carbon bond, extending the carbon framework. The cycle concludes with beta-hydride elimination to release the alkenylated product and regenerate the active catalyst species, often facilitated by a copper oxidant and oxygen atmosphere to re-oxidize the reduced metal center.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or non-directed coupling methods. The rigid geometry of the metallacycle intermediate imposes strict steric constraints that favor the formation of the thermodynamically stable E-isomer of the alkene product, minimizing the formation of Z-isomer impurities that are difficult to separate. Additionally, the use of imidates prevents the competitive coordination of the final nitrile product to the catalyst, which could otherwise lead to over-reaction or catalyst deactivation. The compatibility of this system with a wide range of functional groups, including halogens, esters, and trifluoromethyl groups, ensures that the impurity profile remains clean and manageable. For R&D directors, this level of mechanistic predictability translates to higher confidence in process robustness and easier purification protocols, ultimately leading to superior product quality and consistency in commercial batches.
How to Synthesize O-Alkenyl Aromatic Nitriles Efficiently
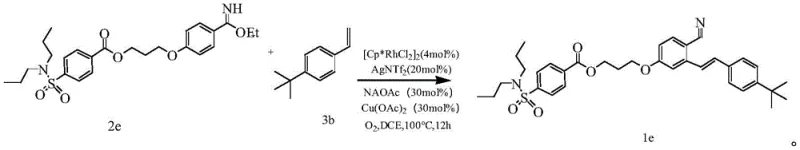
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and selectivity. The general procedure involves dissolving the aryl imidate derivative and the terminal alkene in an inert solvent such as 1,2-dichloroethane, creating a homogeneous reaction mixture. A catalytic amount of a rhodium dimer, specifically dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer, is introduced along with a silver salt additive like silver bis(trifluoromethanesulfonyl)imide to enhance catalyst electrophilicity. Sodium acetate serves as the base to facilitate the C-H cleavage step, while copper acetate acts as the terminal oxidant in conjunction with an oxygen atmosphere. The reaction is typically heated to 100°C for approximately 12 hours to ensure complete conversion. Following the reaction, standard workup procedures involving filtration through diatomaceous earth and purification via silica gel column chromatography yield the target o-alkenyl aromatic nitrile compounds with high purity. The detailed standardized synthesis steps are outlined in the guide below.
- Dissolve aryl imidate derivative and terminal alkene in an inert solvent like 1,2-dichloroethane under an oxygen atmosphere.
- Add rhodium catalyst dimer, silver salt additive, sodium acetate base, and copper oxidant to the reaction mixture.
- Heat the reaction at 100°C for 12 hours, then purify the resulting o-alkenyl nitrile via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed C-H activation technology presents significant opportunities for cost optimization and supply security. The primary economic driver is the elimination of expensive and often supply-constrained ortho-halo aromatic nitriles from the bill of materials. By utilizing simpler, commodity-grade aryl imidates and terminal alkenes, manufacturers can achieve substantial cost reduction in pharmaceutical intermediate manufacturing without compromising on quality. The streamlined nature of the process, which combines C-H activation and alkenylation into a single pot, reduces the number of unit operations required, thereby lowering energy consumption and labor costs associated with isolation and purification steps. Furthermore, the high atom economy of the oxidative coupling reaction minimizes waste disposal costs, aligning with increasingly stringent environmental regulations and sustainability goals. These factors collectively contribute to a more resilient and cost-effective supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The transition from multi-step halide coupling to direct C-H functionalization removes the need for pre-functionalized starting materials, which are often subject to volatile pricing and limited supplier availability. This simplification of the synthetic route directly lowers the cost of goods sold (COGS) by reducing raw material expenses and processing time. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, reducing the burden on downstream purification processes and increasing overall material throughput. The ability to use oxygen as a terminal oxidant further enhances the economic viability by replacing costly stoichiometric oxidants with a cheap and abundant reagent.
- Enhanced Supply Chain Reliability: Relying on broadly available building blocks such as styrenes and benzamidine derivatives mitigates the risk of supply chain disruptions caused by specialized reagent shortages. The robustness of the catalytic system across a wide substrate scope means that a single manufacturing platform can be adapted to produce a diverse library of intermediates, enhancing flexibility. This adaptability is crucial for maintaining continuity of supply for key drug substances, especially when dealing with complex molecules that require late-stage modifications. The method's compatibility with various functional groups ensures that supply chains remain agile and capable of accommodating structural changes during drug development without requiring complete process redevelopment.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to scale-up, operating at moderate temperatures and pressures that are compatible with standard stainless steel reactor infrastructure. The use of less hazardous solvents and the potential for aerobic oxidation pathways support greener manufacturing practices, reducing the environmental footprint of production facilities. This alignment with green chemistry principles not only facilitates regulatory approval but also enhances the corporate social responsibility profile of the manufacturing partner. Scalability is further supported by the high turnover numbers of the rhodium catalyst, ensuring that precious metal usage remains economically feasible even at large production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this o-alkenyl aromatic nitrile synthesis technology. These insights are derived directly from the experimental data and scope limitations defined in the patent literature, providing a realistic overview of what can be achieved in a production environment. Understanding these nuances is vital for project managers evaluating the feasibility of integrating this chemistry into their current portfolio. The answers reflect the balance between synthetic elegance and practical manufacturability that defines modern process chemistry.
Q: Why are aryl imidates used instead of direct benzonitriles in this synthesis?
A: The cyano group is a strong electron-withdrawing group that deactivates the aromatic ring towards electrophilic metalation. Aryl imidates serve as effective precursors that can be converted to nitriles post-reaction, while the imine nitrogen assists in coordinating the metal catalyst for ortho-C-H activation.
Q: What is the stereoselectivity of the resulting alkene products?
A: The reaction demonstrates high stereoselectivity, predominantly yielding the E-configuration isomers of the polysubstituted diarylethene compounds, which is crucial for downstream biological activity and material properties.
Q: Can this method be applied to complex drug molecules?
A: Yes, the protocol exhibits excellent functional group tolerance and has been successfully demonstrated on complex pharmaceutical scaffolds such as probenecid, enabling late-stage functionalization without protecting group manipulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Alkenyl Aromatic Nitrile Supplier
As the global demand for complex pharmaceutical intermediates continues to rise, partnering with a technically proficient CDMO is essential for translating innovative patent technologies into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our team of expert chemists is well-versed in transition metal catalysis and C-H activation methodologies, allowing us to troubleshoot and optimize reactions like the rhodium-catalyzed oxidative Heck coupling described in CN110845363B. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity o-alkenyl aromatic nitriles meets the exacting standards required by top-tier pharmaceutical companies.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this more efficient methodology. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you leverage this cutting-edge chemistry to accelerate your drug development timelines and secure a competitive advantage in the marketplace through superior supply chain performance.
