Advanced One-Pot Dehydration Strategy for High-Purity 2-Alkylthiobenzonitrile Intermediates
Advanced One-Pot Dehydration Strategy for High-Purity 2-Alkylthiobenzonitrile Intermediates
The landscape of fine chemical manufacturing is constantly evolving towards safer, more efficient, and environmentally benign processes, a shift clearly exemplified by the technological breakthroughs detailed in patent CN113816882B. This intellectual property introduces a sophisticated methodology for the preparation of 2-alkylthiobenzonitriles, a class of versatile intermediates pivotal in the synthesis of complex pharmaceutical agents and agrochemicals. The core innovation lies in the strategic replacement of hazardous traditional dehydrating agents with a mild yet potent catalytic system comprising Selectfluor, sodium iodide, and acetic acid. By operating in acetonitrile at elevated temperatures, this novel route achieves superior conversion rates while circumventing the severe handling risks associated with phosphorus-based reagents. For R&D directors and process chemists, this represents a significant leap forward in reaction design, offering a streamlined pathway that maintains high atom economy and operational simplicity without compromising on product quality or safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of nitriles from primary amides has heavily relied on the use of phosphorus oxychloride (POCl3) in conjunction with polar aprotic solvents like DMF. While chemically effective in certain contexts, this legacy approach presents profound challenges for modern supply chain and EHS (Environment, Health, and Safety) management. POCl3 is notoriously corrosive, volatile, and reacts violently with moisture, necessitating specialized storage infrastructure and rigorous containment protocols that drive up operational expenditures. Furthermore, the disposal of phosphorus-containing waste streams imposes a heavy burden on wastewater treatment facilities, often requiring complex neutralization steps to meet environmental compliance regulations. From a yield perspective, comparative data indicates that these traditional methods often struggle to exceed 27% isolated yield for sterically hindered or electronically sensitive substrates like 2-ethylthiobenzamide, leading to significant material loss and increased cost of goods sold (COGS).
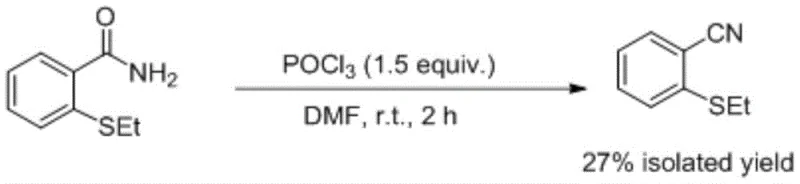
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a synergistic combination of Selectfluor (1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate)), sodium iodide, and acetic acid to effect dehydration under much milder and safer conditions. This system operates effectively in standard acetonitrile solvent at 120 °C, eliminating the need for strictly anhydrous conditions that are mandatory for POCl3 reactions. The transition to this reagent set not only mitigates the acute toxicity risks for plant operators but also simplifies the downstream processing workflow. By avoiding the generation of corrosive phosphoric acid byproducts, the workup procedure is drastically simplified to mere concentration and chromatographic purification. This shift allows manufacturers to utilize standard glass-lined or stainless steel reactors without the need for exotic corrosion-resistant alloys, thereby reducing capital expenditure and enhancing the overall economic viability of producing high-purity 2-alkylthiobenzonitriles on a commercial scale.
Mechanistic Insights into Selectfluor-Mediated Intramolecular Cyclization
The success of this transformation is deeply rooted in the unique mechanistic pathway facilitated by the ortho-alkylthio substituent, which acts as an internal neighboring group participant. Upon exposure to the oxidative conditions generated by Selectfluor and iodide ions, the amide nitrogen undergoes activation, prompting an intramolecular nucleophilic attack by the sulfur atom. This leads to the formation of a transient N,S-cyclic cationic intermediate, which subsequently isomerizes to a more stable N,O-cyclic cationic species. The presence of iodide anions is critical at this stage, as they facilitate the ring-opening and subsequent elimination steps that generate a cyanosulfoxide intermediate. Finally, this intermediate is reduced in situ by the HI/AcOH system generated during the reaction to furnish the final nitrile product. This intricate cascade highlights the importance of the specific reagent stoichiometry, where sodium iodide serves not merely as a salt but as a crucial redox mediator and nucleophile.

Understanding this mechanism is vital for impurity control and process optimization, particularly when scaling up for commercial production. The reliance on the ortho-thio group means that unsubstituted benzamides fail to react under these conditions, as evidenced by control experiments showing 0% conversion for simple benzamide. This specificity ensures that if the starting material contains meta- or para-isomers, they are less likely to interfere or form difficult-to-separate byproducts, thereby enhancing the purity profile of the final API intermediate. Furthermore, the reaction conditions are robust enough to tolerate various functional groups on the alkyl chain, including esters and nitriles, expanding the scope of accessible chemical space. For procurement teams, this mechanistic robustness translates to a wider range of acceptable raw material specifications, reducing the risk of batch rejection due to minor feedstock variations.
How to Synthesize 2-Alkylthiobenzonitrile Efficiently
The practical execution of this synthesis is designed for ease of operation, making it highly attractive for contract development and manufacturing organizations (CDMOs) looking to optimize their service offerings. The protocol involves a straightforward one-pot procedure where all reagents are charged sequentially into a pressure-resistant vessel, minimizing the need for complex addition funnels or inert atmosphere gloveboxes. The reaction proceeds under vigorous stirring to ensure homogeneous heat transfer and mass transport, which is critical for maintaining the 120 °C setpoint over the 24-hour duration. Following the reaction, the workup is remarkably simple, involving solvent removal followed by standard purification techniques. For a detailed breakdown of the precise operational parameters and safety checks required for implementation, please refer to the standardized synthesis guide below.
- Charge a pressure-resistant sealed tube with acetonitrile solvent, 2-alkylthiobenzamide substrate, sodium iodide (3.0 equiv.), acetic acid (1.0 equiv.), and Selectfluor (1.0 equiv.).
- Heat the reaction mixture to 120 °C and stir vigorously for 24 hours to ensure complete conversion via the intramolecular cyclization mechanism.
- Upon completion, concentrate the reaction mixture under reduced pressure and purify the crude residue via column chromatography to isolate the target nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond simple yield improvements. The primary benefit lies in the drastic simplification of the supply chain for raw materials; Selectfluor, sodium iodide, and acetic acid are commodity chemicals with stable global supply networks, unlike specialized or highly regulated toxic reagents. This availability ensures business continuity and reduces the risk of production stoppages due to raw material shortages. Additionally, the elimination of POCl3 removes the logistical complexities associated with transporting and storing hazardous corrosive liquids, which often incur premium shipping rates and require specialized containment vessels. These factors collectively contribute to a more resilient and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The economic impact of switching to this method is driven by multiple factors, primarily the reduction in waste treatment costs and the elimination of expensive corrosion-resistant equipment maintenance. By avoiding the use of phosphorus oxychloride, facilities can significantly lower their expenditure on neutralization agents and hazardous waste disposal fees, which are often calculated based on toxicity and volume. Furthermore, the higher isolated yields observed with this method mean that less starting material is required to produce the same amount of final product, directly lowering the variable cost per kilogram. The ability to use technical grade acetonitrile without rigorous drying also contributes to substantial cost savings in solvent procurement and preparation.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for downstream drug manufacturers, and this process enhances stability by utilizing reagents with long shelf lives and broad availability. Sodium iodide and acetic acid are produced in massive quantities globally, insulating the supply chain from the volatility seen with niche specialty reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in ambient humidity or temperature during setup, reducing the likelihood of batch failures. This consistency allows supply chain planners to forecast production timelines with greater accuracy, ensuring that delivery commitments to pharmaceutical clients are met without delay.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden bottlenecks related to heat management and effluent treatment, but this methodology is inherently scalable due to its homogeneous nature and moderate thermal requirements. The absence of highly exothermic quenching steps, which are typical when destroying excess POCl3 with water, simplifies the engineering controls needed for large-scale reactors. From an environmental perspective, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product) and eliminating persistent phosphorus pollutants. This compliance facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled answers to common inquiries regarding its implementation and scope. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production portfolios or for designing new synthetic pathways for complex molecules.
Q: Why is the ortho-thio substituent critical for this dehydration reaction?
A: The ortho-thio group acts as an internal nucleophilic assistant, facilitating the formation of a cyclic cationic intermediate that lowers the activation energy for dehydration compared to unsubstituted benzamides.
Q: How does this method improve safety over traditional POCl3 protocols?
A: This protocol eliminates the need for phosphorus oxychloride (POCl3), a highly corrosive and volatile toxic reagent, replacing it with stable solid reagents like Selectfluor and sodium iodide.
Q: What is the typical yield range for various alkyl substituents?
A: The method demonstrates robust versatility, achieving isolated yields ranging from 52% to 75% across diverse substrates including methyl, ethyl, benzyl, and functionalized alkyl chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkylthiobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative methods like the Selectfluor-mediated dehydration can be seamlessly transferred to industrial scale. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-alkylthiobenzonitrile meets the exacting standards required for pharmaceutical applications. Our commitment to quality and safety makes us the ideal partner for companies seeking to secure their supply of critical fine chemical intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance metrics.
