Advanced Oxidative Self-Coupling of Benzyl Halides for Commercial Scale Benzyl Benzoate Production
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more sustainable and cost-effective pathways to high-value intermediates. A groundbreaking development in this sector is detailed in Chinese Patent CN108929227B, which discloses a novel method for synthesizing aromatic benzyl esters through the oxidative self-coupling of benzyl halides. This technology represents a paradigm shift from traditional esterification routes, leveraging an N-heterocyclic carbene (NHC) precursor to facilitate the direct transformation of inexpensive benzyl halides into valuable esters like benzyl benzoate. For R&D directors and procurement strategists in the pharmaceutical and fragrance industries, this innovation offers a compelling alternative to legacy processes that rely on costly arylformates. By enabling a one-step synthesis under mild conditions, this patent lays the foundation for a more robust and economically viable supply chain for critical chemical building blocks.
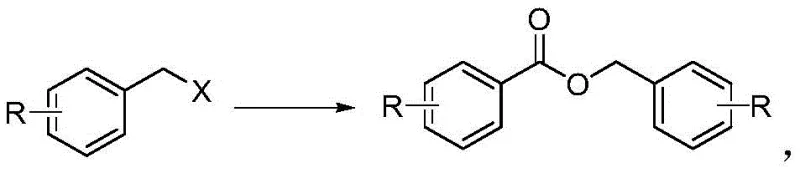
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of aromatic benzyl esters, particularly benzyl benzoate, has relied heavily on two primary methodologies that present significant economic and logistical challenges. The first approach involves the direct reaction of arylformates with benzyl chloride, while the second utilizes an alcohol exchange process reacting methyl or ethyl arylcarboxylates with benzyl alcohol. Both strategies suffer from a fundamental flaw: the reliance on relatively expensive starting materials such as arylformates or alkyl arylcarboxylates. These precursors not only inflate the raw material costs but also introduce additional complexity into the supply chain, as they often require their own multi-step synthesis. Furthermore, traditional esterification processes frequently necessitate harsh reaction conditions, including elevated temperatures and the use of strong acids or bases, which can lead to side reactions, lower atom economy, and increased waste generation. For a procurement manager, these factors translate into higher volatility in pricing and potential bottlenecks in sourcing high-purity precursors.
The Novel Approach
In stark contrast, the methodology described in patent CN108929227B introduces a streamlined, atom-economical route that bypasses the need for pre-formed carboxylic acid derivatives. This novel approach utilizes benzyl halides, which are commodity chemicals available in vast quantities and at a fraction of the cost of arylformates. The core of this innovation lies in the use of 1,3-bis(2,4,6-trimethylphenyl)imidazolium chloride as a catalyst precursor, which generates an active NHC species in situ. This catalyst facilitates the oxidative self-coupling of the benzyl halide in the presence of a base and molecular oxygen from the air. The result is a direct, one-step conversion to the corresponding aromatic benzyl ester. This shift not only drastically simplifies the synthetic route but also aligns perfectly with green chemistry principles by utilizing air as the terminal oxidant and operating under ambient conditions. For manufacturers seeking a reliable aromatic benzyl ester supplier, this technology promises a more stable and cost-efficient production model.
Mechanistic Insights into NHC-Catalyzed Oxidative Coupling
The mechanistic elegance of this transformation lies in the dual role of the catalytic system and the oxidant. The reaction initiates with the deprotonation of the imidazolium salt by a base, such as cesium carbonate, to generate the free N-heterocyclic carbene. This nucleophilic carbene species then interacts with the benzyl halide, likely forming a reactive intermediate that undergoes oxidation. Crucially, the reaction utilizes molecular oxygen from the air atmosphere as the oxidant, which is a significant departure from stoichiometric oxidants that generate heavy metal waste. The oxidation step converts the benzylic carbon into a carbonyl equivalent, which subsequently reacts with another molecule of the benzyl species (or an intermediate thereof) to form the ester linkage. The use of dimethyl sulfoxide (DMSO) as the solvent is particularly critical, as experimental data indicates it provides superior yields compared to other solvents like dichloromethane or acetonitrile. This solvent effect suggests that DMSO may play a role in stabilizing polar intermediates or facilitating the oxygen transfer process, ensuring high conversion rates.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity OLED material or pharmaceutical intermediates. Because the reaction proceeds under mild conditions (20-25°C) and avoids strong acidic or basic hydrolysis conditions typical of traditional esterification, the formation of degradation by-products is minimized. The specificity of the NHC catalyst ensures that the coupling occurs selectively at the benzylic position, reducing the risk of ring substitution or polymerization side reactions. Furthermore, the tolerance of the system to various substituents on the aromatic ring, including electron-withdrawing groups like nitro and halogens, demonstrates the robustness of the catalytic cycle. This broad substrate scope implies that the impurity profile remains consistent and manageable across different derivatives, simplifying downstream purification processes such as silica gel column chromatography or crystallization. For quality control teams, this predictability is invaluable in maintaining stringent purity specifications required for regulated industries.
How to Synthesize Benzyl Benzoate Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and solvent selection to maximize efficiency. The patent outlines a straightforward protocol where benzyl halide is reacted with the imidazolium catalyst and a base in an organic solvent. The choice of base is pivotal; while various bases like potassium carbonate or organic amines were tested, cesium carbonate emerged as the optimal choice, delivering significantly higher yields. Similarly, the solvent must be carefully selected to avoid methanol, which was found to inhibit the reaction completely. Instead, polar aprotic solvents like DMSO are preferred to solubilize the ionic intermediates and facilitate the oxidative process. The following guide summarizes the standardized operational parameters derived from the patent data to ensure reproducible results.
- Combine benzyl halide, 1,3-bis(2,4,6-trimethylphenyl)imidazolium chloride catalyst, and cesium carbonate base in an organic solvent such as DMSO.
- Stir the reaction mixture at ambient temperature (20-25°C) under an air atmosphere for approximately 20 to 24 hours until conversion is complete.
- Quench with water, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography to isolate the pure ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxidative self-coupling technology translates into tangible strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic reduction in raw material costs achieved by substituting expensive arylformates with commodity benzyl halides. Benzyl chloride and its derivatives are produced on a massive global scale for various applications, ensuring a stable supply and competitive pricing. This shift eliminates the dependency on niche ester precursors, thereby insulating the manufacturing process from supply shocks associated with specialized chemical markets. Additionally, the elimination of transition metal catalysts or stoichiometric oxidants simplifies the waste stream, reducing the environmental compliance burden and the costs associated with hazardous waste disposal. These factors collectively contribute to a more resilient and cost-effective manufacturing framework.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the utilization of low-cost starting materials and the minimization of processing steps. By employing benzyl halides, which are significantly cheaper than the arylformates required in conventional methods, the direct material cost per kilogram of product is substantially lowered. Furthermore, the reaction operates at ambient temperature, removing the energy costs associated with heating or cooling large-scale reactors. The use of air as the oxidant eliminates the need for purchasing and handling expensive chemical oxidants, further driving down operational expenditures. This comprehensive cost optimization makes the process highly attractive for large-scale commercial production where margin improvement is critical.
- Enhanced Supply Chain Reliability: Sourcing consistency is a major concern for supply chain leaders, and this method addresses it by relying on widely available feedstocks. Benzyl halides are standard industrial chemicals with established global supply chains, reducing the risk of shortages that can plague more complex intermediates. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supplies, such as steam or chilled water, enhancing overall plant reliability. Moreover, the simplified workflow reduces the number of unit operations required, decreasing the potential for equipment failure or bottlenecks. This reliability ensures a steady flow of high-purity aromatic benzyl esters to downstream customers, strengthening long-term supplier relationships.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the mild nature of this oxidative coupling facilitates a smoother transition from lab to plant. The absence of exothermic hazards associated with strong oxidants or high-temperature reactions allows for safer scale-up in standard stainless steel reactors. From an environmental standpoint, the high atom economy of the self-coupling reaction means that a larger proportion of the starting material ends up in the final product, minimizing waste generation. The avoidance of heavy metal catalysts simplifies the purification process and ensures that the final product meets strict residual metal specifications without extensive treatment. These attributes align with modern sustainability goals and regulatory requirements, positioning the manufacturer as a responsible partner in the green chemistry initiative.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries based on the patent specifications. These questions address key concerns regarding reaction conditions, substrate compatibility, and purification methods. Understanding these details is crucial for R&D directors planning to integrate this method into their existing workflows or for procurement teams assessing the quality implications of the new process. The following insights provide a clear picture of the operational realities and potential advantages of adopting this synthetic route.
Q: What are the primary advantages of this oxidative self-coupling method over traditional esterification?
A: Unlike traditional methods requiring expensive arylformates or alcohol exchange processes, this method utilizes readily available benzyl halides as the sole carbon source, significantly reducing raw material costs and simplifying the supply chain.
Q: Does this process require harsh reaction conditions or inert atmospheres?
A: No, the process operates under mild conditions at ambient temperature (20-25°C) and utilizes air as the oxidant, eliminating the need for expensive inert gas protection or high-energy heating systems.
Q: Is this method applicable to substituted benzyl halides for diverse derivative synthesis?
A: Yes, the patent demonstrates high tolerance for various substituents including nitro, chloro, and different halides (Cl, Br, I), making it a versatile platform for synthesizing a wide range of functionalized aromatic benzyl esters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Benzoate Supplier
The technological breakthroughs detailed in patent CN108929227B highlight the immense potential of oxidative self-coupling for the efficient production of aromatic benzyl esters. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative pathways and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex catalytic reactions under controlled atmospheres, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we are uniquely positioned to deliver high-purity benzyl benzoate and its derivatives to meet the demanding needs of the global pharmaceutical and fragrance markets.
We invite industry partners to explore how this advanced synthesis method can enhance their supply chain efficiency and product quality. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your next project. By collaborating with us, you gain access to cutting-edge chemistry backed by reliable manufacturing capabilities, ensuring a secure and competitive supply of critical chemical intermediates for your business growth.
