Scalable Metal-Free Electrochemical Synthesis of Complex Thiocyanate Intermediates for Pharma
Scalable Metal-Free Electrochemical Synthesis of Complex Thiocyanate Intermediates for Pharma
The landscape of organic synthesis is undergoing a paradigm shift towards sustainable and metal-free methodologies, a trend vividly exemplified by the innovations disclosed in patent CN115386902A. This pivotal intellectual property introduces a groundbreaking diaphragm-free electrochemical method for constructing compounds containing multiple carbon-sulfur (C-S) bonds, a structural motif ubiquitous in high-value pharmaceutical intermediates and agrochemical agents. Unlike traditional approaches that rely heavily on stoichiometric oxidants or expensive transition metal catalysts, this novel technique leverages the precision of constant current electrolysis to drive the formation of thiocyanate derivatives under exceptionally mild conditions. For R&D directors and process chemists seeking to modernize their synthetic portfolios, this technology offers a compelling pathway to access complex heterocyclic scaffolds with superior purity profiles. By utilizing a simple undivided cell setup with carbon and platinum electrodes, the process effectively bypasses the logistical and environmental burdens associated with heavy metal removal, positioning it as a highly attractive candidate for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-SCN bonds has been dominated by methodologies that present significant hurdles for industrial application, particularly regarding safety, cost, and environmental compliance. Traditional synthetic routes frequently depend on transition metal catalysis involving palladium, copper, or iron, which inevitably leads to the contamination of the final product with trace metal residues that are notoriously difficult and expensive to remove to meet stringent pharmacopeial standards. Furthermore, many existing protocols require the use of hazardous stoichiometric oxidants or harsh reaction conditions that pose safety risks during large-scale manufacturing. Photocatalytic methods, while innovative, often suffer from limited photon penetration depths, making them challenging to scale beyond gram quantities without specialized and costly flow reactor equipment. These conventional limitations result in prolonged production lead times, increased waste generation, and a complex purification workflow that erodes profit margins for reliable pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast to these legacy methods, the electrochemical strategy outlined in CN115386902A represents a transformative leap forward by utilizing electrons as the primary reagent to drive the oxidative coupling. This approach eliminates the need for any external chemical oxidants or transition metal catalysts, thereby fundamentally removing the source of heavy metal contamination from the process stream. The reaction is conducted in a diaphragm-free cell using acetonitrile as a solvent, allowing for a simplified reactor design that is inherently easier to scale than divided cells or photo-flow systems. Operating at room temperature with a constant current of 5-15mA, the method ensures a controlled and steady generation of reactive radical species, minimizing side reactions and thermal degradation of sensitive substrates. This gentle yet efficient protocol not only enhances the overall yield and selectivity but also drastically simplifies the downstream processing, offering a clear route for cost reduction in API manufacturing through reduced raw material consumption and waste treatment costs.
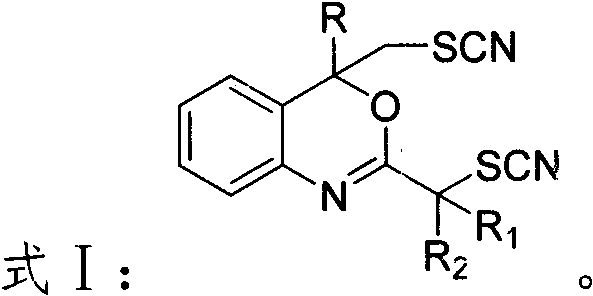
Mechanistic Insights into Electrochemical C-S Bond Construction
The core of this innovation lies in the anodic oxidation of thiocyanate salts to generate reactive thiocyanogen or thiocyanato radical species in situ, which subsequently engage with the electron-rich enaminone substrates. In the absence of a diaphragm, the electrochemical cell facilitates a direct interaction between the anodically generated electrophilic sulfur species and the nucleophilic double bond of the enaminone. The applied constant current ensures a steady flux of electrons, maintaining a low concentration of the highly reactive radical intermediates, which is crucial for controlling selectivity and preventing over-oxidation or polymerization. The presence of trifluoroacetic acid acts as a proton source and electrolyte support, enhancing the conductivity of the acetonitrile medium and facilitating the cyclization steps that lead to the final fused heterocyclic products. This mechanistic pathway avoids the high-energy barriers associated with thermal activation, allowing the reaction to proceed smoothly at ambient temperatures while preserving the integrity of other functional groups on the molecule.
From an impurity control perspective, the metal-free nature of this electrochemical transformation provides a distinct advantage in managing the impurity profile of the final active pharmaceutical ingredient. Since no transition metals are introduced, the rigorous and costly steps typically required for metal scavenging, such as treatment with silica-thiol resins or activated carbon, are rendered unnecessary. The primary by-products are derived from the solvent or minor over-oxidation events, which are generally easier to separate via standard silica gel chromatography compared to metal complexes. The use of a carbon anode and platinum cathode ensures high stability and minimal electrode degradation, further reducing the risk of particulate contamination. This clean reaction profile is particularly beneficial for the synthesis of drug candidates where regulatory limits on elemental impurities are extremely tight, ensuring that the resulting high-purity OLED material or pharmaceutical intermediate meets global quality specifications without extensive remediation.
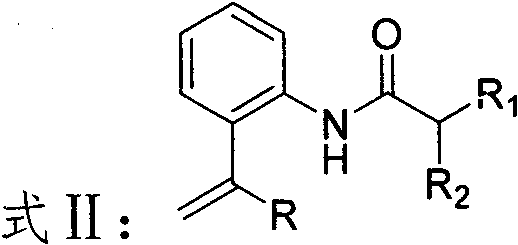
How to Synthesize Thiocyanate Derivatives Efficiently
The practical implementation of this electrochemical methodology is designed to be straightforward and accessible, requiring standard laboratory equipment that can be easily adapted for pilot and commercial plant operations. The process begins with the preparation of the reaction mixture in a dried vessel, where the enaminone substrate is combined with an excess of ammonium thiocyanate and a catalytic amount of trifluoroacetic acid in acetonitrile. This mixture is then subjected to electrolysis using a simple undivided cell configuration, where the precise control of current density becomes the critical parameter for optimizing conversion and selectivity. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, electrode dimensions, and workup procedures necessary to replicate the high yields reported in the patent examples.
- Prepare the reaction vessel by installing a stirring bar in a pre-dried three-necked bottle and adding the enaminone substrate along with ammonium thiocyanate and trifluoroacetic acid.
- Introduce acetonitrile as the solvent system and set up the electrochemical cell using a carbon anode and a platinum cathode without a diaphragm.
- Apply a constant current of 5-15mA at room temperature for 8-12 hours, then concentrate the mixture and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical synthesis route translates into tangible strategic benefits that extend far beyond the laboratory bench. The elimination of expensive transition metal catalysts such as palladium or rhodium directly impacts the bill of materials, resulting in substantial cost savings by removing one of the most volatile and high-cost input categories in fine chemical manufacturing. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and shortens the overall cycle time per batch. This efficiency gain allows for a more responsive supply chain capable of meeting tight delivery schedules without the bottlenecks typically associated with purification-heavy processes. The robustness of the method across a wide range of substrates also means that a single production line can be utilized for multiple products, enhancing asset utilization and flexibility.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free electrochemical process is profound, primarily driven by the removal of precious metal catalysts and the associated purification costs. By relying on electricity as the reagent, the process decouples production costs from the fluctuating market prices of rare earth metals and transition metal salts. Additionally, the mild reaction conditions reduce energy consumption related to heating or cooling, and the use of recoverable solvents like acetonitrile further lowers the operational expenditure. The avoidance of hazardous oxidants also reduces the costs associated with safety handling, storage, and the disposal of toxic chemical waste, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the simplicity and robustness of the reagent profile, which relies on commodity chemicals like ammonium thiocyanate and acetonitrile that are readily available in the global market. Unlike specialized catalysts that may have long lead times or single-source dependencies, the inputs for this electrochemical method are stable and abundant, mitigating the risk of production stoppages due to raw material shortages. The scalability of the undivided cell design means that increasing production volume does not require a complete redesign of the reactor infrastructure, allowing for seamless capacity expansion to meet surging demand from downstream pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this technology aligns perfectly with the industry's push towards greener chemistry and sustainable manufacturing practices. The absence of heavy metal waste streams simplifies effluent treatment and ensures compliance with increasingly strict environmental regulations regarding discharge limits. The process generates minimal hazardous waste, and the potential for solvent recycling creates a closed-loop system that minimizes the environmental footprint. This eco-friendly profile not only reduces liability but also enhances the brand value of the final product, appealing to end-users who prioritize sustainability in their sourcing decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology, based on the detailed experimental data and beneficial effects described in the patent literature. These insights are intended to clarify the operational feasibility and strategic value of adopting this metal-free route for the production of high-value sulfur-containing intermediates. Understanding these nuances is critical for technical teams evaluating the integration of electrochemical flow or batch processes into their existing manufacturing infrastructure.
Q: What are the primary advantages of this electrochemical method over traditional transition metal catalysis?
A: The primary advantage is the complete elimination of transition metal catalysts, which removes the risk of toxic metal residues in the final pharmaceutical intermediate. Additionally, the reaction proceeds under mild room temperature conditions without the need for hazardous external oxidants, significantly simplifying the safety profile and waste treatment protocols.
Q: What are the specific reaction conditions required for this synthesis?
A: The synthesis operates under constant current electrolysis at 5-15mA using a diaphragm-free cell. The reaction utilizes acetonitrile as the solvent with trifluoroacetic acid as an additive, running at room temperature for approximately 8 to 12 hours to ensure complete conversion of the enaminone substrate.
Q: Is this method suitable for complex natural product modification?
A: Yes, the method demonstrates a broad substrate scope that accommodates not only simple synthetic substrates but also complex molecules. The mild, metal-free nature of the electrochemical oxidation makes it particularly compatible with sensitive functional groups often found in natural product derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiocyanate Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in redefining the efficiency and sustainability of pharmaceutical intermediate production. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115386902A are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of thiocyanate derivatives meets the highest international standards. We are committed to leveraging our technical expertise to help you navigate the complexities of metal-free synthesis, delivering high-purity intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to explore how this cutting-edge technology can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecules, demonstrating the economic viability of switching to this electrochemical route. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to bring your next-generation therapeutics to market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
