Advanced Organocatalytic Synthesis of Chiral Trifluoromethyl Maleimide Derivatives for Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, particularly those incorporating fluorine atoms which are known to enhance metabolic stability and bioavailability. Patent CN111548298B introduces a groundbreaking approach to synthesizing chiral trifluoromethyl-substituted maleimide derivatives, a class of compounds with significant potential in drug discovery. This technology leverages an asymmetric non-conjugated Michael addition coupled with a spontaneous 1,3-hydrogen migration, enabling the direct formation of the maleimide core in a single operational step. By utilizing chiral organic catalysts, specifically thiourea derivatives, the process achieves exceptional stereocontrol without the need for toxic transition metals. This innovation addresses a critical gap in the current synthetic landscape, providing a reliable pathway to enrich chiral drug candidate libraries with fluorinated motifs that were previously difficult to access efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this development, the synthesis of chiral maleimides from 2-alkenyl succinimides was severely constrained by substrate specificity. Existing literature, such as reports in Chem. Eur. J. (2010) and J. Org. Chem. (2012), demonstrated that the crucial 1,3-hydrogen migration required to aromatize the ring to a maleimide could only proceed when the alkenyl terminus was unsubstituted. When aromatic or bulky groups were present at this position, the reaction would stall at the succinimide stage, failing to generate the desired maleimide architecture. This limitation drastically restricted the chemical diversity accessible to medicinal chemists, forcing them to rely on multi-step sequences or harsh conditions that often compromised yield and optical purity. Furthermore, traditional methods frequently lacked the ability to introduce trifluoromethyl groups directly, necessitating additional functionalization steps that increased cost and environmental burden.
The Novel Approach
The methodology disclosed in CN111548298B overcomes these historical barriers by employing a specialized chiral organocatalytic system that tolerates a wide range of substituents at the alkenyl terminus. As illustrated in the general reaction scheme below, the process successfully accommodates aryl, heteroaryl, and condensed aryl groups, thereby vastly expanding the scope of accessible derivatives. 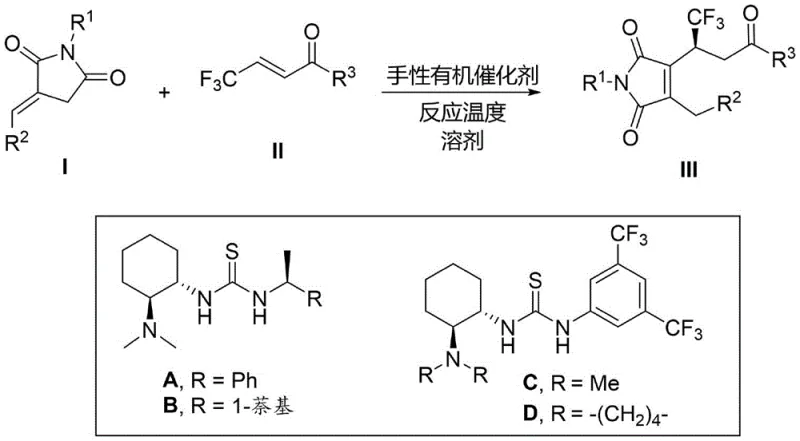 The reaction proceeds under mild conditions, typically between 0-50°C, utilizing common organic solvents like dichloromethane. This novel approach not only simplifies the synthetic route by combining the addition and migration into a one-pot procedure but also ensures high stereoselectivity, with enantiomeric excess values reaching up to 98% in optimized examples. This represents a significant leap forward for cost reduction in pharmaceutical intermediates manufacturing, as it eliminates the need for protecting group manipulations or separate oxidation steps often required in legacy routes.
The reaction proceeds under mild conditions, typically between 0-50°C, utilizing common organic solvents like dichloromethane. This novel approach not only simplifies the synthetic route by combining the addition and migration into a one-pot procedure but also ensures high stereoselectivity, with enantiomeric excess values reaching up to 98% in optimized examples. This represents a significant leap forward for cost reduction in pharmaceutical intermediates manufacturing, as it eliminates the need for protecting group manipulations or separate oxidation steps often required in legacy routes.
Mechanistic Insights into Asymmetric Non-Conjugated Michael Addition
The core of this transformation lies in the precise activation of the substrates by the chiral thiourea catalyst. The catalyst, derived from cyclohexanediamine, functions as a bifunctional activator. It simultaneously activates the beta-trifluoromethyl enone electrophile through hydrogen bonding interactions with the thiourea moiety while organizing the 2-arylalkenyl succinimide nucleophile. This dual activation lowers the energy barrier for the asymmetric non-conjugated Michael addition, ensuring that the new carbon-carbon bond is formed with high facial selectivity. Following the initial addition, the intermediate undergoes a spontaneous 1,3-hydrogen migration. This rearrangement is driven by the thermodynamic stability gained upon forming the conjugated maleimide double bond system. The presence of the trifluoromethyl group adjacent to the newly formed stereocenter further influences the electronic environment, stabilizing the transition state and locking in the chiral information imparted by the catalyst.
Impurity control is inherently managed by the high specificity of the organocatalyst. Unlike metal-catalyzed reactions which can suffer from background racemic reactions or metal-induced side products, this organic system relies on well-defined hydrogen-bonding networks. The patent data indicates that optimizing the catalyst structure (varying R groups on the thiourea scaffold) allows for fine-tuning of the steric environment, effectively suppressing the formation of diastereomers. For instance, specific catalyst variants like Catalyst A or D are selected based on the electronic nature of the R2 substituent on the succinimide. This tunability ensures that even with diverse substrates, the impurity profile remains clean, facilitating easier downstream purification and reducing the load on quality control laboratories during commercial production.
How to Synthesize Chiral Trifluoromethyl Maleimide Efficiently
To implement this synthesis effectively, one must adhere to the optimized parameters regarding solvent choice and stoichiometry. The patent highlights dichloromethane as the preferred solvent due to its ability to balance solubility and reaction rate, although toluene and ethyl acetate are viable alternatives. The molar ratio of the succinimide substrate to the trifluoromethyl enone is critical; a ratio of approximately 2:1 is recommended to drive the reaction to completion without excessive waste of the valuable fluorinated reagent. The reaction time typically spans 1 to 3 days at temperatures around 30°C, allowing sufficient time for the slow 1,3-hydrogen migration step to occur fully. Below is the standardized protocol framework derived from the patent examples, illustrating the practical execution of this chemistry.
- Dissolve the chiral organic catalyst and 2-arylalkenyl succinimide substrate in an organic solvent such as dichloromethane.
- Add beta-trifluoromethyl enone to the mixture under stirring and maintain the reaction temperature between 0-50°C for 1 to 3 days.
- Upon completion, separate and purify the resulting chiral maleimide derivative using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic route offers substantial strategic benefits beyond mere chemical elegance. The elimination of transition metal catalysts is a primary driver for cost optimization, as it removes the expensive and time-consuming heavy metal scavenging steps typically required to meet regulatory limits for API intermediates. This simplification of the workup procedure translates directly into reduced processing time and lower consumption of specialized purification resins. Furthermore, the use of readily available starting materials, such as substituted succinimides and commercially sourced trifluoromethyl enones, ensures a stable and resilient supply chain. The robustness of the reaction conditions means that sourcing does not require exotic or highly sensitive reagents that might be subject to geopolitical supply constraints.
- Cost Reduction in Manufacturing: The process utilizes low loading levels of chiral organic catalysts, typically around 10 mol%, which is economically favorable compared to precious metal complexes. Since the catalyst is metal-free, there is no risk of metal contamination in the final product, thereby avoiding the capital expenditure associated with metal removal technologies. The atom economy of the reaction is high, as the 1,3-hydrogen migration occurs intramolecularly without the need for external oxidants or additives, leading to significant waste reduction and lower disposal costs for the manufacturing facility.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are structurally simple and can be sourced from multiple global vendors, mitigating the risk of single-source dependency. The reaction's tolerance to various functional groups means that a single platform technology can be used to generate a wide library of derivatives, allowing for flexible production scheduling based on market demand. The mild reaction temperatures (0-50°C) reduce energy consumption compared to high-temperature processes, contributing to more predictable operational expenditures and reducing the thermal load on plant infrastructure.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is straightforward due to the absence of exothermic hazards often associated with metal-catalyzed couplings. The solvents used, such as dichloromethane or ethyl acetate, are standard in the fine chemical industry and have established recovery and recycling protocols. By avoiding heavy metals, the process aligns better with increasingly stringent environmental regulations regarding wastewater discharge, simplifying the permitting process for new manufacturing lines and enhancing the overall sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for R&D and procurement planning. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing production workflows.
Q: What distinguishes this synthesis method from conventional approaches for maleimides?
A: Unlike conventional methods which are limited to terminal alkenyl groups, this patented process allows for diverse aromatic substituents at the alkenyl terminus, significantly expanding substrate universality while maintaining high stereoselectivity through a unique 1,3-hydrogen migration mechanism.
Q: What type of catalyst is utilized in this trifluoromethyl maleimide preparation?
A: The process utilizes chiral organic catalysts derived from cyclohexanediamine thiourea. These metal-free catalysts facilitate the asymmetric non-conjugated Michael addition, eliminating the need for expensive transition metals and simplifying downstream purification.
Q: What are the optimal reaction conditions for scaling this synthesis?
A: The reaction operates under mild conditions, preferably at 30°C in solvents like dichloromethane. The molar ratio of substrate to enone is optimized around 2:1 to maximize yield and minimize reagent waste, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Maleimide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology for the development of next-generation fluorinated pharmaceuticals. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements essential for chiral intermediates. We are committed to delivering high-purity chiral maleimide derivatives that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this synthesis route can optimize your specific project requirements. By engaging with us, you can request a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the economic advantages of this metal-free approach. Please contact our technical procurement team today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you secure a competitive edge through superior chemical innovation and supply chain reliability.
