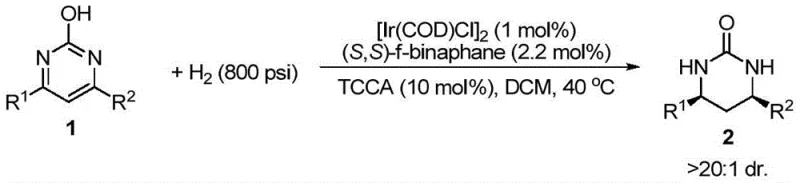
Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Commercial Chiral Cyclic Urea Production
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral scaffolds, particularly cyclic ureas which serve as critical motifs in numerous bioactive molecules. A groundbreaking approach detailed in patent CN109824601B introduces a highly efficient iridium-catalyzed asymmetric hydrogenation strategy for converting 2-hydroxypyrimidine compounds into valuable chiral cyclic ureas. This technology addresses the longstanding challenge of hydrogenating aromatic heterocycles, which typically require harsh conditions that compromise stereocontrol. By leveraging the unique tautomerization properties of 2-hydroxypyrimidines, this method achieves exceptional enantiomeric excess values reaching up to 96% under relatively mild conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this innovation represents a significant leap forward in process efficiency and product quality.

The synthesis of chiral amines and ureas via asymmetric hydrogenation of aromatic heterocycles has historically been fraught with difficulties due to the inherent stability of the aromatic ring. Conventional methods often struggle to balance reactivity with stereoselectivity, frequently necessitating extreme pressures or temperatures that degrade product integrity. Furthermore, the presence of heteroatoms like nitrogen and oxygen in these substrates can lead to catalyst poisoning, resulting in low turnover numbers and inconsistent batch quality. Previous attempts using ruthenium or palladium catalysts have shown promise but were often limited by narrow substrate scopes or mediocre enantioselectivity, failing to meet the rigorous purity standards required for modern drug development. These limitations underscore the urgent need for a more versatile and robust catalytic system capable of handling diverse substitution patterns without sacrificing yield or optical purity.
The novel approach presented in this patent overcomes these barriers by utilizing a specialized iridium catalytic system paired with chiral bisphosphine ligands. The key breakthrough lies in the strategic selection of 2-hydroxypyrimidine substrates, which exist in a keto-enol equilibrium that effectively reduces aromatic character, rendering the ring more susceptible to hydrogenation. This subtle electronic modulation allows the iridium complex to engage the substrate efficiently at moderate temperatures ranging from 25 to 80 degrees Celsius. The use of additives such as trichloroisocyanuric acid further enhances the catalytic cycle, ensuring high conversion rates and superior diastereoselectivity. This methodology not only simplifies the synthetic route but also provides a scalable pathway for producing high-purity pharmaceutical intermediates with consistent stereochemical outcomes.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The success of this transformation relies heavily on the interplay between the iridium metal center and the chiral ligand environment. The catalytic cycle initiates with the formation of an active iridium-hydride species upon exposure to hydrogen gas. The chiral ligand, such as (S,S)-f-Binaphane or (R)-SegPhos, creates a sterically defined pocket that dictates the facial selectivity of the hydrogen addition to the pyrimidine ring. As the substrate coordinates to the metal center, the reduced aromaticity of the keto-tautomer facilitates the insertion of the hydride species. This step is critical as it establishes the stereocenters at the 4 and 6 positions of the resulting tetrahydropyrimidinone ring. The precise geometry of the ligand ensures that hydrogen delivery occurs from a specific face, thereby locking in the desired absolute configuration with high fidelity.
Impurity control is another vital aspect of this mechanistic design, particularly for applications requiring stringent regulatory compliance. The mild reaction conditions minimize the formation of side products such as over-reduced species or decomposition byproducts that often plague high-temperature hydrogenations. Additionally, the choice of solvent and additive plays a crucial role in suppressing non-selective background reactions. For instance, the use of dichloromethane or ethanol mixtures optimizes the solubility of both the catalyst and the substrate while maintaining the stability of the active catalytic species. This careful tuning of the reaction medium ensures that the final product profile is clean, significantly reducing the burden on downstream purification processes and enhancing the overall economic viability of the manufacturing process.
How to Synthesize Chiral Cyclic Urea Efficiently
Implementing this synthesis requires careful attention to catalyst preparation and reaction parameters to maximize yield and enantioselectivity. The process begins with the in situ generation of the active catalyst by mixing the iridium precursor with the chiral ligand in an inert atmosphere. This mixture is then introduced to the substrate solution containing the necessary halogenated additive. The reaction is conducted in a pressurized vessel under a hydrogen atmosphere, where temperature and pressure are tightly controlled to maintain optimal catalytic activity. Detailed standardized synthesis steps for this procedure are outlined in the guide below, providing a clear roadmap for laboratory and pilot-scale execution.
- Prepare the catalyst by mixing an iridium metal precursor, such as [Ir(COD)Cl]2, with a chiral bisphosphine ligand like (S,S)-f-Binaphane in an organic solvent at room temperature.
- Combine the prepared catalyst solution with the 2-hydroxypyrimidine substrate and an additive like trichloroisocyanuric acid (TCCA) in a hydrogenation vessel.
- Pressurize the reactor with hydrogen gas (400-800 psi) and stir at 25-80°C for 12-24 hours to achieve high conversion and enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this iridium-catalyzed technology offers substantial benefits for supply chain stability and cost management in fine chemical manufacturing. The ability to operate under milder conditions compared to traditional high-energy processes translates directly into reduced operational expenditures and lower safety risks. Furthermore, the high selectivity of the reaction minimizes waste generation and simplifies purification, leading to significant improvements in overall process mass intensity. For procurement managers, this means a more predictable supply of high-quality intermediates with reduced variability between batches, ensuring consistent raw material quality for downstream API synthesis.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the use of commercially available catalyst precursors significantly lower the barrier to entry for production. By avoiding expensive proprietary reagents and complex multi-step sequences, manufacturers can achieve substantial cost savings. The high atom economy of the hydrogenation reaction ensures that raw materials are utilized efficiently, reducing the cost per kilogram of the final product and enhancing profit margins for large-scale operations.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against various substituents on the pyrimidine ring ensures that supply chains are less vulnerable to raw material fluctuations. Since the method tolerates a wide range of functional groups, suppliers can source diverse starting materials without needing to re-optimize the entire process for each new derivative. This flexibility allows for rapid adaptation to changing market demands and ensures continuous availability of critical chiral building blocks for pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard high-pressure hydrogenation equipment found in most modern chemical facilities. The mild temperatures and absence of toxic heavy metal residues in the final product simplify waste treatment and disposal protocols. This alignment with green chemistry principles not only reduces environmental impact but also streamlines regulatory approvals, facilitating faster time-to-market for new drug candidates relying on these chiral cyclic urea scaffolds.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this method into their existing workflows. The answers provided are derived directly from the experimental data and specifications outlined in the patent documentation, ensuring accuracy and relevance for technical decision-making.
Q: What are the key advantages of this iridium-catalyzed method over traditional ruthenium or palladium systems?
A: Unlike traditional ruthenium or palladium systems which often suffer from poor enantioselectivity or limited substrate scope, this iridium-catalyzed method achieves up to 96% ee and works effectively on a broad range of 2-hydroxypyrimidine derivatives under mild conditions.
Q: How does the tautomerization of the substrate facilitate the hydrogenation reaction?
A: The introduction of a hydroxyl group at the 2-position of the pyrimidine ring creates a keto-enol tautomerization equilibrium. In protic solvents, the keto form predominates, which significantly reduces the aromaticity of the ring, thereby lowering the energy barrier for hydrogenation and improving reactivity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes commercially available catalysts and standard high-pressure hydrogenation equipment. The mild reaction temperatures (25-80°C) and robust catalytic system make it highly scalable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cyclic Urea Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of iridium-catalyzed hydrogenations, including high-pressure reactors and rigorous QC labs capable of verifying stringent purity specifications. We understand that consistency is paramount in the pharmaceutical supply chain, and our dedicated process chemistry team ensures that every batch meets the highest international standards for enantiomeric excess and chemical purity.
We invite global partners to collaborate with us to leverage this cutting-edge technology for their drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable synthetic strategy available in the market today.
