Advanced Chiral Ionic Thiourea Catalysts for High-Efficiency Asymmetric Synthesis
Advanced Chiral Ionic Thiourea Catalysts for High-Efficiency Asymmetric Synthesis
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of multifunctional ionic systems that merge the benefits of organocatalysis with the unique properties of ionic liquids. Patent CN101143862B discloses a groundbreaking class of ionic compounds containing chiral amine-thiourea or urea structures, designed to overcome the limitations of traditional neutral catalysts. These novel materials function not merely as hydrogen-bond donors but as complex ionic environments that can stabilize transition states through electrostatic interactions while simultaneously activating substrates via dual hydrogen bonding. This dual-activation mode represents a paradigm shift in how we approach stereocontrol in carbon-carbon bond-forming reactions, offering a robust platform for the synthesis of high-value pharmaceutical intermediates and fine chemicals with exceptional optical purity.
At the core of this innovation is the strategic combination of a chiral cation, typically derived from imidazolium or pyridinium scaffolds linked to pyrrolidine units, with a chiral anion featuring thiourea or urea moieties. This architecture ensures that both the cation and anion contribute to the chiral environment, creating a synergistic effect that enhances enantioselectivity beyond what is achievable with single-component catalysts. The ability to tune the steric and electronic properties of both ions allows for precise optimization of reaction outcomes, making this technology highly versatile for diverse synthetic challenges. Furthermore, the ionic nature of these compounds imparts unique solubility profiles, enabling their use in greener solvent systems and potentially allowing for catalyst recycling, a critical factor for sustainable industrial processes.
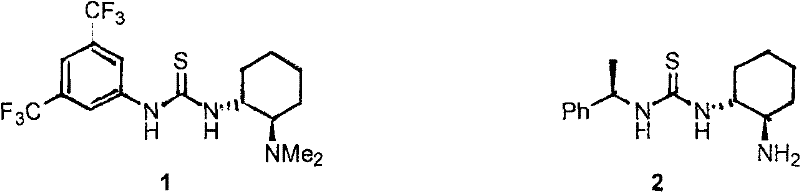
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional asymmetric organocatalysis has largely relied on neutral small-molecule catalysts, such as the well-known Takemoto or Jacobsen thioureas. While effective, these neutral species often suffer from significant drawbacks when translated to industrial settings, particularly regarding recovery and reuse. Since they are soluble in standard organic solvents, separating the catalyst from the product usually requires chromatographic purification or extensive washing, which increases waste generation and operational costs. Additionally, neutral catalysts can sometimes exhibit limited solubility in environmentally benign solvents like water, restricting the ability to run reactions in greener media. The lack of an ionic charge also means they cannot leverage electrostatic stabilization effects, which could otherwise lower activation energies and improve reaction rates in polar transition states.
The Novel Approach
The ionic strategy presented in this patent addresses these deficiencies by converting the catalytic motif into a salt form. By incorporating a charged imidazolium or pyridinium headgroup, the catalyst gains solubility in polar and aqueous mixtures, opening the door to biphasic catalysis or simple precipitation methods for recovery. The patent data demonstrates that these ionic catalysts maintain, and in some cases exceed, the enantioselectivity of their neutral counterparts. For instance, in asymmetric Michael reactions, specific embodiments of these ionic compounds achieved yields of 97% with enantiomeric excess (ee) values reaching 98%, outperforming many literature precedents. This approach effectively merges the high selectivity of organocatalysis with the process advantages of ionic liquids, providing a compelling solution for cost reduction in pharmaceutical intermediate manufacturing.
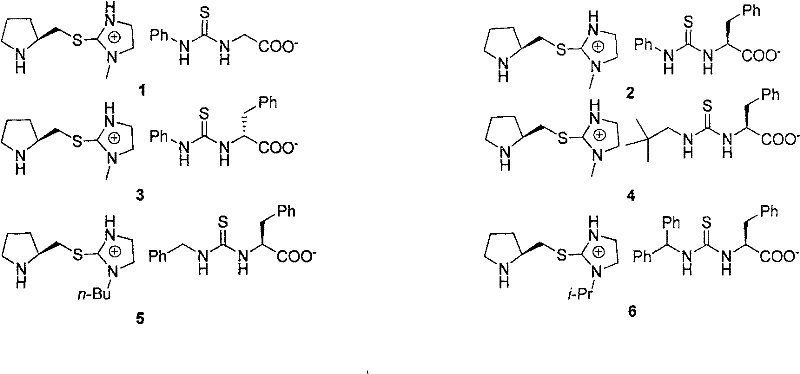
Mechanistic Insights into Dual-Activation Ionic Catalysis
The superior performance of these chiral ionic compounds stems from a sophisticated dual-activation mechanism that leverages the distinct roles of the cation and anion. The thiourea or urea moiety on the anion acts as a potent hydrogen-bond donor, activating electrophilic substrates such as nitroolefins or aldehydes by lowering the energy of their LUMO. Simultaneously, the chiral amine component, often a pyrrolidine ring attached to the cation, can form enamines or iminium ions with carbonyl nucleophiles, thereby activating them for attack. The proximity of the positive charge on the imidazolium ring further stabilizes the developing negative charge in the transition state, effectively organizing the reactants within a tight chiral pocket. This organized ionic environment minimizes non-productive background reactions and ensures that the nucleophilic attack occurs from a specific facial trajectory, resulting in high stereocontrol.
Impurity control is another critical aspect where this ionic design excels. The strong electrostatic interactions between the cation and anion prevent the dissociation of the catalytic species, reducing the likelihood of catalyst decomposition or the formation of inactive aggregates that often plague neutral systems. Moreover, the specific substitution patterns on the aromatic rings of the thiourea anion, such as the 3,5-bis(trifluoromethyl)phenyl groups mentioned in the patent, enhance the acidity of the N-H protons, strengthening the hydrogen bonding network. This increased acidity translates to higher turnover frequencies and allows the reaction to proceed efficiently even at lower catalyst loadings. The result is a cleaner reaction profile with fewer side products, simplifying downstream purification and ensuring the high-purity standards required for API synthesis.
How to Synthesize Chiral Ionic Thiourea Compounds Efficiently
The preparation of these advanced catalysts is remarkably straightforward, relying on a simple metathesis reaction that avoids harsh conditions or complex multi-step sequences. The process involves mixing a chiral halide salt (the cation precursor) with a chiral carboxylate salt (the anion precursor) in a suitable solvent system. The reaction proceeds spontaneously at room temperature, driven by the precipitation of the inorganic byproduct (typically sodium bromide) or simply by the stability of the resulting ionic pair. This simplicity is a major advantage for scale-up, as it eliminates the need for specialized equipment or hazardous reagents. Detailed standardized synthesis steps for specific analogues are provided in the guide below, ensuring reproducibility across different batches.
- Dissolve the chiral imidazolium or pyridinium bromide salt and the chiral thiourea/urea sodium salt in a mixture of organic solvent and water.
- Stir the reaction mixture at room temperature for 24 to 72 hours to ensure complete ion exchange and formation of the target ionic compound.
- Remove the solvent under reduced pressure, dissolve the solid residue in chloroform or dichloromethane, filter to remove inorganic salts, and evaporate to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic catalyst technology offers tangible benefits that extend beyond mere chemical performance. The ability to utilize green solvents such as water-acetone mixtures significantly reduces the environmental footprint of the manufacturing process, aligning with increasingly stringent global regulations on volatile organic compound (VOC) emissions. This shift away from chlorinated solvents not only lowers disposal costs but also mitigates supply risks associated with regulated chemicals. Furthermore, the high efficiency of these catalysts means that lower quantities are required to achieve the same output, directly reducing the raw material cost per kilogram of the final product. The robustness of the ionic structure also suggests improved shelf-life and stability during transport, reducing the risk of degradation before use.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals and the ability to operate under mild conditions drastically simplify the production workflow. By avoiding cryogenic temperatures and inert atmospheres, energy consumption is significantly lowered, and the requirement for specialized reactor linings is removed. The potential for catalyst recycling due to the ionic nature of the material further amplifies these savings, as the effective cost of the catalyst per batch decreases over time. This creates a more predictable and optimized cost structure for long-term production campaigns.
- Enhanced Supply Chain Reliability: The starting materials for these catalysts, such as substituted imidazoles and amino acids, are commercially available commodity chemicals with stable supply chains. Unlike exotic ligands that rely on single-source suppliers, the modular nature of this ionic design allows for flexible sourcing of precursors. This diversification reduces the risk of supply disruptions and ensures continuity of operations. Additionally, the simplified synthesis route shortens the lead time for catalyst production, enabling faster response to fluctuating market demands for chiral intermediates.
- Scalability and Environmental Compliance: The use of aqueous solvent systems facilitates easier heat management and safety control during scale-up, as water has a high heat capacity and is non-flammable. This inherent safety margin allows for larger batch sizes without proportional increases in risk, supporting the commercial scale-up of complex pharmaceutical intermediates. Moreover, the reduced generation of hazardous waste simplifies compliance with environmental permits, accelerating the approval process for new manufacturing sites and ensuring long-term operational viability in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these chiral ionic catalysts in industrial settings. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for process evaluation. Understanding these details is crucial for R&D teams assessing the feasibility of integrating this technology into existing synthetic routes.
Q: What are the primary advantages of these ionic catalysts over traditional small molecules?
A: Unlike traditional neutral thiourea catalysts, these ionic variants offer enhanced solubility in green solvents like water and polyethylene glycol, facilitating easier recovery and potential recyclability while maintaining high enantioselectivity.
Q: Which asymmetric reactions are best suited for this catalyst library?
A: The patent specifically highlights exceptional performance in Michael additions, Aldol reactions, Baylis-Hillman reactions, and tandem Michael-Aldol sequences, often achieving ee values exceeding 90%.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the preparation involves simple salt metathesis at room temperature without requiring cryogenic conditions or expensive transition metals, making it highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ionic Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the ionic thiourea catalyst technology described in CN101143862B to your specific process needs, ensuring that the high enantioselectivity observed in the lab is maintained at the plant scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of catalyst meets the exacting standards required for GMP manufacturing of active pharmaceutical ingredients.
We invite you to collaborate with us to unlock the full potential of this innovative catalytic system for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current synthesis route. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our chiral ionic compounds can drive efficiency and quality in your manufacturing operations.
