Revolutionizing Para-Selective C-H Borylation for Commercial Pharmaceutical Intermediate Manufacturing
The landscape of organic synthesis for pharmaceutical intermediates is undergoing a significant transformation driven by the need for precise regiocontrol in C-H functionalization. Patent CN115536681A introduces a groundbreaking methodology for the selective synthesis of para-position pinacol boron ester substituted anisole compounds, addressing a long-standing challenge in the field of transition metal catalysis. Traditionally, the direct borylation of electron-rich arenes like anisoles has been plagued by poor selectivity, often yielding mixtures of ortho and meta isomers that are difficult and costly to separate. This new approach leverages a dual-catalytic system involving a noble metal iridium complex and a specialized organoboron Lewis acid to achieve unprecedented levels of para-selectivity. By fundamentally altering the steric environment around the substrate through non-covalent interactions, this technology enables the efficient production of high-purity building blocks essential for modern drug discovery and development. For R&D directors and procurement specialists, this represents a pivotal shift towards more streamlined, cost-effective synthetic routes that minimize waste and maximize throughput in the manufacturing of complex organic synthons.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, transition metal-catalyzed C-H bond functionalization has relied heavily on the use of directing groups to control regioselectivity. In the context of anisole derivatives, the methoxy group acts as a weak directing group that typically favors activation at the ortho position due to the formation of stable five-membered metallacycle intermediates. This inherent bias severely limits the utility of such reactions for accessing para-substituted products, which are often the desired scaffolds for downstream cross-coupling reactions in API synthesis. Conventional strategies to overcome this limitation often involve multi-step protection and deprotection sequences, or the use of stoichiometric amounts of harsh reagents that generate significant chemical waste. Furthermore, the separation of closely related regioisomers frequently requires extensive chromatographic purification, leading to substantial material loss and increased operational costs. These inefficiencies create bottlenecks in the supply chain, extending lead times and complicating the scale-up process for commercial production of fine chemicals.
The Novel Approach
The methodology disclosed in CN115536681A circumvents these traditional constraints by employing a supramolecular strategy that utilizes non-covalent interactions to dictate reaction outcomes. Instead of relying on covalent coordination to direct the metal catalyst, this process introduces a bulky organoboron catalyst, such as triphenylborane, which interacts with the lone pair electrons on the methoxy oxygen of the anisole substrate. This Lewis acid-base interaction effectively creates a transient, sterically demanding shield around the ortho and meta positions of the aromatic ring. Consequently, the active iridium catalyst species is sterically repelled from these blocked sites and is forced to approach and activate the more accessible remote para C-H bond.  This elegant mechanism allows for the direct installation of pinacol boron ester groups at the para position with high fidelity, eliminating the need for cumbersome protecting group strategies and significantly simplifying the overall synthetic workflow for producing valuable pharmaceutical intermediates.
This elegant mechanism allows for the direct installation of pinacol boron ester groups at the para position with high fidelity, eliminating the need for cumbersome protecting group strategies and significantly simplifying the overall synthetic workflow for producing valuable pharmaceutical intermediates.
Mechanistic Insights into Ir-Catalyzed Remote Para-Selective Borylation
The core of this technological advancement lies in the synergistic interplay between the noble metal catalyst and the organoboron additive. The iridium catalyst, typically generated in situ from precursors like [Ir(COD)OMe]2 and ligands such as 4,4'-di-tert-butylbipyridine (dtbpy), is responsible for the actual C-H bond cleavage and borylation event. However, without the organoboron modifier, the intrinsic electronic and steric properties of the anisole substrate would direct the iridium to the ortho position. The introduction of the trisubstituted organoboron compound changes the physical topology of the substrate. Because the organoboron catalyst possesses an empty p-orbital perpendicular to its sp2 hybridized plane, it acts as a potent Lewis acid. When it complexes with the electron-rich oxygen atom of the methoxy group, the large aryl substituents on the boron atom project outwards, physically occluding the adjacent positions on the benzene ring. 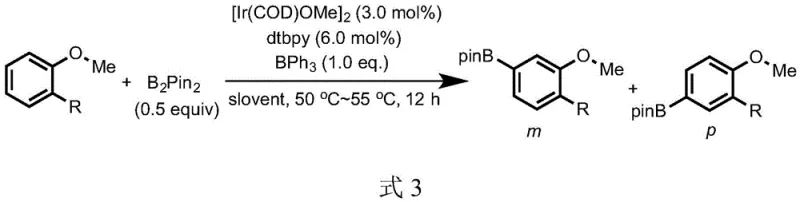 This steric masking effect ensures that the bulky iridium complex can only access the para position, resulting in a meta-to-para isomer ratio that consistently exceeds 15:1 in optimized conditions. This level of control is critical for maintaining high purity specifications required in GMP manufacturing environments.
This steric masking effect ensures that the bulky iridium complex can only access the para position, resulting in a meta-to-para isomer ratio that consistently exceeds 15:1 in optimized conditions. This level of control is critical for maintaining high purity specifications required in GMP manufacturing environments.
From an impurity control perspective, this mechanism offers distinct advantages over traditional electrophilic aromatic substitution or lithiation-borylation sequences. The mild reaction conditions, operating at temperatures between 50°C and 55°C, prevent the thermal degradation of sensitive functional groups that might be present on the substrate. Additionally, the high selectivity minimizes the formation of regioisomeric byproducts, which are often the most difficult impurities to remove during purification. The process tolerates a wide range of substituents, including halogens, esters, amides, and silyl groups, without compromising the para-selectivity. This robustness implies that the impurity profile of the final product is predictable and manageable, reducing the burden on quality control laboratories. For supply chain managers, this translates to more consistent batch-to-batch quality and reduced risk of production delays caused by out-of-specification results, ensuring a reliable flow of high-purity API intermediates to downstream customers.
How to Synthesize Para-Pinacol Boron Ester Efficiently
Implementing this synthesis route requires careful attention to the molar ratios of the catalysts and the choice of solvent to maximize the efficiency of the Lewis acid masking effect. The standard protocol involves charging a dry reaction vessel with the anisole derivative and bis(pinacolato)diboron, followed by the addition of the organoboron catalyst such as BPh3. The noble metal catalyst system is then introduced, and the mixture is heated in a solvent like p-xylene. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by combining the anisole derivative substrate with bis(pinacolato)diboron and the organoboron Lewis acid catalyst in a dry vessel.
- Add the noble metal iridium catalyst system, typically generated from [Ir(COD)OMe]2 and 4,4'-di-tert-butylbipyridine, along with an appropriate organic solvent such as p-xylene.
- Maintain the reaction temperature between 50°C and 55°C for 8 to 24 hours, then isolate the para-substituted product via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced supply security. By eliminating the need for multiple protection and deprotection steps, the overall number of unit operations is decreased, leading to shorter production cycles and lower labor costs. Furthermore, the high selectivity of the reaction reduces the consumption of raw materials that would otherwise be lost in the form of unwanted isomers, thereby improving the overall mass balance of the process. This efficiency gain is particularly valuable in the current economic climate where raw material costs are volatile and margin pressure is high across the fine chemical sector.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions and the use of readily available, stable catalysts significantly lowers the energy footprint and equipment requirements for this synthesis. Traditional lithiation methods often require temperatures below minus 70 degrees Celsius, necessitating specialized cooling infrastructure and high energy consumption. In contrast, this iridium-catalyzed process operates at mild temperatures near 50 degrees Celsius, which can be achieved using standard heating utilities available in most multipurpose chemical plants. Additionally, the removal of expensive and toxic heavy metal scavenging steps, often required for palladium-catalyzed cross-couplings, further reduces the cost of goods sold. The qualitative improvement in process efficiency allows for significant cost savings without compromising the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against various functional groups ensures that a wide range of anisole derivatives can be sourced and processed without extensive custom optimization for each new substrate. This flexibility allows suppliers to maintain a broader inventory of finished goods or respond more rapidly to custom synthesis requests. The use of common solvents like p-xylene and commercially available boron reagents mitigates the risk of supply disruptions associated with exotic or highly regulated chemicals. Consequently, lead times for high-purity building blocks can be reduced, providing downstream drug manufacturers with greater agility in their own production planning and inventory management strategies.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the homogeneous nature of the catalytic system and the absence of hazardous reagents like n-butyllithium. The mild conditions reduce the risk of thermal runaways, making the process inherently safer for large-scale reactors. From an environmental perspective, the high atom economy and selectivity result in less chemical waste generation, aligning with increasingly stringent global regulations on industrial emissions and waste disposal. The ability to produce complex organic synthons with minimal environmental impact enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical companies seeking to meet their corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this para-selective borylation technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners and licensees.
Q: How does this method overcome traditional ortho-selectivity limitations in anisole borylation?
A: Traditional methods rely on directing groups that favor ortho-activation. This patent utilizes a bulky organoboron Lewis acid that coordinates with the methoxy oxygen, sterically blocking the ortho and meta positions, thereby forcing the iridium catalyst to activate the remote para position exclusively.
Q: What are the typical yield and selectivity profiles for this synthesis route?
A: According to the experimental data in CN115536681A, the process achieves total yields ranging from 60% to 82%, with a meta-to-para isomer ratio consistently exceeding 15:1, demonstrating exceptional regioselectivity suitable for industrial applications.
Q: Is this process scalable for commercial production of API intermediates?
A: Yes, the reaction operates under mild thermal conditions (50-55°C) using common organic solvents like p-xylene, avoiding cryogenic requirements or hazardous reagents, which significantly simplifies engineering controls for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Para-Pinacol Boron Ester Supplier
The technical potential of this para-selective borylation route is immense, offering a clear pathway to more efficient and sustainable manufacturing of critical pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the global market. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of pinacol boron ester meets the exacting standards of the international pharmaceutical industry. We understand the complexities of transitioning novel catalytic processes from the bench to the plant and are committed to delivering consistent quality and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced synthetic technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific product portfolio. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us help you secure a competitive advantage in the market with our superior chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
